To evaluate incorporation of the hospital pharmacist to the routine activity of an intensive care unit (ICU).
DesignA prospective observational study was carried out to evaluate the impact of pharmacist interventions, made by a pharmacist temporarily assigned to the ICU, upon medical prescriptions.
SettingA medical and surgical ICU with 21 beds.
PatientsPatients with at least one ICU stay were included, while patients with admission and discharge in periods when the pharmacist was not present were excluded.
InterventionsThe interventions were made after daily review of the prescriptions, and were communicated verbally or in writing to the supervising physician.
Main variablesNumber of interventions, therapeutic group of the drugs involved, type of intervention and degree of acceptance.
ResultsA total of 194 interventions were made in 62 patients. The majority were related to safety aspects (33%) and the optimization of therapy (32%). The most frequent interventions were the administration of drugs via the nasogastric tube (19%) and pharmacokinetic monitoring (14.4%). The most frequently involved groups of drugs were anti-infectious agents (33%) and digestive system medications (27%). A total of 56.2% of the interventions were made verbally, and 80% were accepted.
ConclusionsPharmacist adscription to an ICU and the implementation of interventions on prescriptions have allowed improvement of safety and the optimization of pharmacotherapy in more than 50% of the patients. The high rate of acceptance of these interventions would support the implementation of such programs in critical care units.
Evaluar la incorporación de un farmacéutico de hospital a la actividad habitual de una unidad de cuidados intensivos (UCI).
DiseñoEstudio observacional prospectivo para evaluar las intervenciones farmacéuticas, realizadas por un farmacéutico adscrito temporalmente a una UCI, sobre las prescripciones médicas.
ÁmbitoUCI médico-quirúrgica con 21 camas.
PacientesSe incluyeron los pacientes con al menos una estancia en UCI y se excluyeron los pacientes con ingreso y alta en periodos en los que no se contó con la presencia física del farmacéutico.
IntervencionesSe realizaban tras la revisión diaria de las prescripciones y se comunicaban de forma verbal o escrita al médico responsable.
Variables principalesIntervenciones realizadas, grupo terapéutico de los medicamentos implicados, forma de realización de la intervención y el grado de aceptación.
ResultadosSe realizaron 194 intervenciones en 62 pacientes. La mayoría estaban relacionadas con aspectos de seguridad (33%) y optimización de la terapia (32%). Las más frecuentes se dirigieron a la administración de medicamentos por sonda nasogástrica (19%) y a la monitorización farmacocinética (14,4%). Los grupos de medicamentos más implicados fueron los antiinfecciosos en un 33% de los casos y los del aparato digestivo en un 27%. El 56,2% de las intervenciones se realizaron verbalmente y el 80% fueron aceptadas.
ConclusionesLa adscripción de un farmacéutico a una UCI y la realización de intervenciones sobre las prescripciones han permitido mejorar la seguridad y optimizar la farmacoterapia en más de la mitad de los pacientes. La alta tasa de aceptación de las mismas apoyaría la implementación de este tipo de programas en las unidades de críticos.
Patients admitted to the intensive care unit (ICU) are at a high risk of suffering medication error because of the nature or critical condition of their illness, polymedication, the use of high risk drugs, and the increased frequency of changes in drug treatment.1–3 A number of studies suggest that the daily participation of a clinical pharmacist in the ICU can result in increased prescription effectiveness and efficiency, and moreover may reduce the number of medication errors.1,3–7
The most common activities of pharmacists in the ICU comprise the provision of drug information, the detection of drug-related problems (DRPs), and the identification of interactions – these activities representing between 3% and over 90% of their interventions.5,8 The literature indicates that most pharmaceutical interventions, between 80% and 100%, are accepted by the physicians.8–10
With the purpose of defining the clinical practice and activities of intensive care pharmacists, the American College of Clinical Pharmacy and the Society of Critical Care Medicine produced a document11,12 specifying the fundamental, desirable and optimum activities of pharmacists in intensive care. The activities recommended by these organizations focused on drug treatment safety and optimization.
According to the mentioned document, the fundamental activities are: (1) evaluation of the pharmacotherapeutic regimens (doses, interactions, drug allergies, etc.); (2) identification and prevention of DRPs and medication errors; (3) pharmacokinetic monitoring; (4) nutritional care; (5) the supplying of drug information; (6) pharmacotherapeutic recommendations; and (7) implementation of measures to curb drug costs. All these fundamental activities seek to ensure safe pharmaceutical care. The desirable activities in turn focus on the development of clinical functions as a member of the multidisciplinary team during the patient monitoring and follow-up sessions, and participation in the development of protocols. Lastly, the optimum activities are the expression of an integrated and specialized model dedicated to research and training, with the purpose of optimizing the pharmacotherapeutic outcomes.
The Departments of Hospital Pharmacy and Intensive Care Medicine of our center designed a pharmaceutical care plan for critical patients to be implemented by a pharmaceutical resident in training (PRT) during his or her clinical rotation in the ICU in the course of the last year of the resident training program and under the supervision of the specialists in both departments.
The present study evaluates the adscription of a full time dedication pharmacist to the ICU team, conducting pharmaceutical interventions (PIs), with the aim of determining whether this type of initiative can offer added value to the optimization of drug therapy and safety in drug use among the critical patients of our unit.
Patients and methodsA prospective observational study was carried out to assess the impact of the adscription of a pharmacist to the ICU clinical team upon pharmacotherapeutic prescription practice. The impact was measured on the basis of the number and types of PIs made.
The study was carried out during the months of February and March 2016, in the course of the rotation of the PRT, who checked the medical prescriptions on the working days of the week. As a result, not all the patient stays are covered by the study. We included all patients with at least one ICU stay (except those admitted to the ICU in the afternoon and discharged on that same afternoon). Patients with admission and discharge on weekends or holidays were excluded.
The ICU of our third level university hospital is a medical-surgical unit with 21 admission beds (polyvalent, polytraumatized, neurocritical, coronary and postsurgical patients). The unit registers 1050–1080 admissions a year, with a mean stay of 5.7 days, and occupation rate of 85–90% and a mortality rate of 10–12.4%. The unit operates with electronic prescriptions and case histories, and a specialist from the Department of Hospital Pharmacy usually performs the pharmaceutical care of critical patients on a part time basis. Among other functions, his or her activities include participation in the clinical session of the Department of Intensive Care Medicine one day a week. The PRT participates in the daily patient clinical review session and then remains in the unit during the morning shift (from 8:00 a.m. to 3:00 p.m.) to conduct patient pharmacotherapeutic review and resolve any possible pharmacotherapeutic consultations.
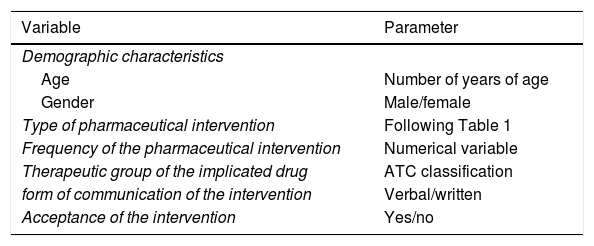
The interventions were classified into 5 types: optimization of therapy, safety, medication reconciliation, drug information, and prophylaxis. Each intervention in turn was structured into different categories (Table 1). A form was developed for patient data collection, with registry of the following variables (Table 2): patient demographic characteristics (age and gender), the type of pharmaceutical intervention and its frequency, the therapeutic group of the drug implied in the intervention based on the Anatomical Therapeutic Chemical (ATC) classification. The latter classifies drugs into therapeutic groups using an alphanumerical code developed by the World Health Organization (WHO), and which contemplates the system or organ upon which the drug acts, the pharmacological effect and the chemical structure of the drug. The customized form also recorded the type of communication (written or verbal) of the intervention, and its acceptance (yes or no) on the part of the clinician.
Types of pharmaceutical interventions.
| Optimization of therapy |
| Dose adjustment |
| Dose adjustment in renal failure |
| Changes in treatment |
| Pharmacokinetic monitoring |
| Parenteral nutrition |
| Safety |
| Interactions |
| Drug allergies |
| Prescription error |
| NGT drug administration |
| Maximum doses |
| Pharmacotherapeutic reconciliation |
| Drug omission |
| Drug information |
| Professionals: active or passivea |
| Reporting of ADRs |
| Prophylaxis |
| Ulcer |
| Deep venous thrombosis |
| Laxatives |
ADRs: adverse drug reactions; NGT: nasogastric tube.
Study variables.
| Variable | Parameter |
|---|---|
| Demographic characteristics | |
| Age | Number of years of age |
| Gender | Male/female |
| Type of pharmaceutical intervention | Following Table 1 |
| Frequency of the pharmaceutical intervention | Numerical variable |
| Therapeutic group of the implicated drug | ATC classification |
| form of communication of the intervention | Verbal/written |
| Acceptance of the intervention | Yes/no |
ATC: anatomical therapeutic chemical.
A descriptive statistical analysis was made of the study variables, with the calculation of measures of central tendency and dispersion for quantitative variables and percentages for qualitative variables.
ResultsThe PRT remained in the unit for a total of 34 days during the months of February and March 2016, from 8:00 a.m. to 3:00 p.m., Monday to Friday. During that study period, a total of 165 patient admissions were recorded, with a mean stay of 6.88 days and a mortality rate of 12.1%. A total of 96 patients were included in the study. Males predominated (71.8%), and the overall mean age was 61.03 years (standard deviation [SD] 13.8; range: 18–84).
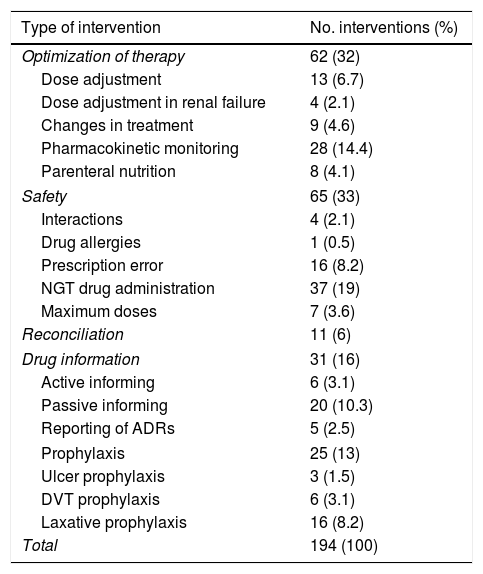
A total of 194 interventions were carried out, affecting 64.6% of the patients included in the study (i.e., practically two-thirds of the total), with an average of 5.4 interventions a day, and of two interventions per patient (SD 2.6; range: 0–13). Most of the interventions (Table 3) were related to safety (33%) and to treatment optimization (32%), followed at a distance by drug information (16%) and pharmacotherapeutic prophylaxis (13%). There were few drug reconciliation interventions (6%).
Types of interventions carried out.
| Type of intervention | No. interventions (%) |
|---|---|
| Optimization of therapy | 62 (32) |
| Dose adjustment | 13 (6.7) |
| Dose adjustment in renal failure | 4 (2.1) |
| Changes in treatment | 9 (4.6) |
| Pharmacokinetic monitoring | 28 (14.4) |
| Parenteral nutrition | 8 (4.1) |
| Safety | 65 (33) |
| Interactions | 4 (2.1) |
| Drug allergies | 1 (0.5) |
| Prescription error | 16 (8.2) |
| NGT drug administration | 37 (19) |
| Maximum doses | 7 (3.6) |
| Reconciliation | 11 (6) |
| Drug information | 31 (16) |
| Active informing | 6 (3.1) |
| Passive informing | 20 (10.3) |
| Reporting of ADRs | 5 (2.5) |
| Prophylaxis | 25 (13) |
| Ulcer prophylaxis | 3 (1.5) |
| DVT prophylaxis | 6 (3.1) |
| Laxative prophylaxis | 16 (8.2) |
| Total | 194 (100) |
ADRs: adverse drug reactions; NGT: nasogastric tube; DVT: deep venous thrombosis.
The most common interventions (Table 3) were referred to drug administration via the nasogastric tube (19%), pharmacokinetic monitoring (14.4%) and passive informing (10.3%). Prescription errors (8.2%), interventions for the prevention of constipation (8.2%), and dose adjustment (6.7%) were relatively frequent. The least common interventions were referred to aspects as important as drug allergies (0.5%), pharmacological prevention of stress ulcers (1.5%), clinically relevant drug interactions (2.1%), and dose adjustment in the context of renal failure (2.1%).
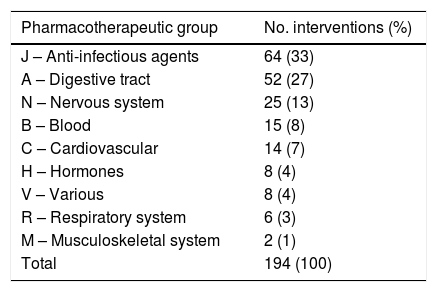
The drugs most often implicated in the interventions (Table 4), classified according to the ATC system, were anti-infectious agents – group J (33%), drugs of the digestive tract – group A (27%), and drugs acting upon the nervous system – group N (13%). The least frequently implicated drugs were those acting upon the musculoskeletal system – group M (1%), drugs with actions upon the respiratory system – group R (3%), and various – group V (4%).
Interventions according to pharmacotherapeutic group.
| Pharmacotherapeutic group | No. interventions (%) |
|---|---|
| J – Anti-infectious agents | 64 (33) |
| A – Digestive tract | 52 (27) |
| N – Nervous system | 25 (13) |
| B – Blood | 15 (8) |
| C – Cardiovascular | 14 (7) |
| H – Hormones | 8 (4) |
| V – Various | 8 (4) |
| R – Respiratory system | 6 (3) |
| M – Musculoskeletal system | 2 (1) |
| Total | 194 (100) |
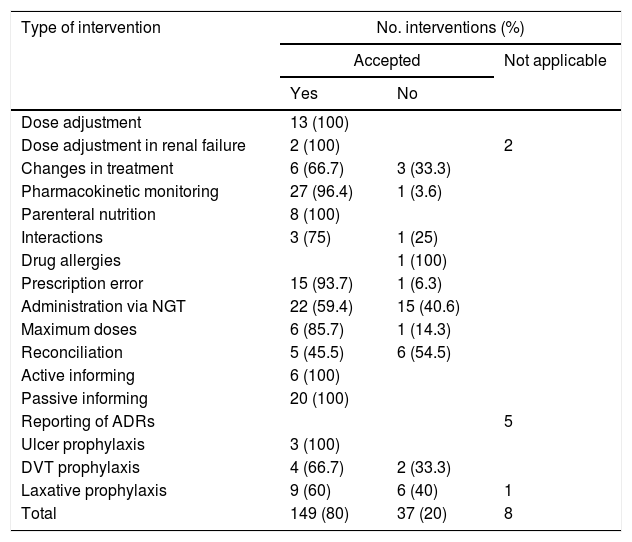
A total of 56.2% of the interventions were carried out verbally, and 80% of them were accepted by the clinician. The distribution of acceptance according to the type of intervention is shown in Table 5. Among the proportionately less accepted interventions, mention must be made of those referred to administration via the nasogastric tube, prophylaxis with laxatives (in patients receiving high-dose opiates and/or admitted due to acute coronary syndrome), and the omission of some medication after pharmacotherapeutic reconciliation (detection of unjustified discrepancies on comparing the regular home medication of the patient against the medication prescribed upon hospital admission). In contrast, the most widely accepted interventions were referred to pharmacokinetic monitoring and drug dose adjustment.
Intervention outcomes according to category.
| Type of intervention | No. interventions (%) | ||
|---|---|---|---|
| Accepted | Not applicable | ||
| Yes | No | ||
| Dose adjustment | 13 (100) | ||
| Dose adjustment in renal failure | 2 (100) | 2 | |
| Changes in treatment | 6 (66.7) | 3 (33.3) | |
| Pharmacokinetic monitoring | 27 (96.4) | 1 (3.6) | |
| Parenteral nutrition | 8 (100) | ||
| Interactions | 3 (75) | 1 (25) | |
| Drug allergies | 1 (100) | ||
| Prescription error | 15 (93.7) | 1 (6.3) | |
| Administration via NGT | 22 (59.4) | 15 (40.6) | |
| Maximum doses | 6 (85.7) | 1 (14.3) | |
| Reconciliation | 5 (45.5) | 6 (54.5) | |
| Active informing | 6 (100) | ||
| Passive informing | 20 (100) | ||
| Reporting of ADRs | 5 | ||
| Ulcer prophylaxis | 3 (100) | ||
| DVT prophylaxis | 4 (66.7) | 2 (33.3) | |
| Laxative prophylaxis | 9 (60) | 6 (40) | 1 |
| Total | 149 (80) | 37 (20) | 8 |
ADRs: adverse drug reactions; NGT: nasogastric tube; DVT: deep venous thrombosis.
The present study was carried out to globally assess the full time presence of a pharmacist in the ICU of a Spanish hospital, taking into account that such a presence is not common in our setting – though it has been widely implemented in other countries, as evidenced by the many studies that have examined the impact of the adscription of a pharmacist to the ICU. The published studies differ in terms of design and pharmacist dedication time. In this regard, mention should be made of interventional studies with reference periods and interventional periods,1,4,6 with full dedication of the pharmacist as part of the ICU clinical team,4,6 or with only part time dedication.1,3 Although some are controlled studies,2,5,8 many are limited to addressing only partial aspects. Most publications have focused on the impact of the prevention of medication errors,1,4,6 while others are more specifically referred to sedation and analgesia,7 antimicrobial treatment7 or the detection of interactions.13 Some studies have moreover evaluated whether the adscription of a pharmacist to the ICU and the introduction of interventions for optimization and the prevention of errors have an impact in terms of cost reductions.1,4,5,7
Klopotowska et al.1 carried out a study on the adscription of a hospital pharmacist to the ICU, with the purpose of evaluating its impact upon safety in the use of medicines. The authors found the incidence of prescription error during the intervention period to be significantly lower than during the reference period: 62.5 per 1000 patients/day versus 190.5 per 1000 patients/day, respectively (p<0.001). In the study published by Leape et al.,4 the activities of the pharmacists included prescription clarification and correction, the provision of drug information, the recommendation of alternative therapies and the identification of interactions. The authors recorded a 66% decrease in DRP rate following intervention of the pharmacist. Ho et al.14 in turn related patient complexity to the presence of PI, and found increased complexity patients to be those with the greatest number of PIs. Results similar to our own were also obtained by Johansen et al.3 and Hunfeld et al.,15 who found 53.4% and the 66% of their patients to have some PI, with an average of 1.8 and 2.1 interventions/patient respectively.
In our case, the adscription of a pharmacist to the ICU was on a full time basis and forming part of the ICU team. However, it covered only a short period of time, patient stays of less than one day or spanning only a weekend were excluded, with no reference period, and taking advantage of the rotation of a PRT in the last year of residency. The activities and PIs carried out by the PRT during the study were consistent with those defined as fundamental activities of pharmacists in the ICU established in the document11,12 of the American College of Clinical Pharmacy and the Society of Critical Care Medicine.
Most of the PIs focused on treatment safety and optimization, in concordance with the data reported by Shulman et al.,9 where the two predominant interventions were related to the improvement and optimization of pharmacotherapeutic effectiveness and safety, and accounted for 73.8% of all the interventions (versus 65% in our study).
The interventions referred to dose omission error, inadequate dosing, maximum doses, treatment duplication and dose adjustments in renal failure have been the most common interventions in the majority of studies.1,3,4,6,10,15,16 In our case, the predominant PI was referred to drug administration through the nasogastric tube. This may be explained by the cost optimization programs in force and the protocols referred to the reduction of infectious complications by avoiding intravenous dosing in critical patients. This type of intervention has been reported in two studies15,16 and with a frequency of only 13% and 5.9%, respectively. In both studies the intervention included recommendations on administration via the nasogastric tube and on the compatibility of intravenous drug mixtures.
Pharmacokinetic monitoring was one of the PIs compiled by Johansen et al.,3 with a frequency of 7%, which is half that recorded in our series (14.4%). This difference could be explained by the fact that in our context (a unit largely focused on infections and neurocritical patients) it is of special interest to monitor the levels of nephrotoxic antibiotics and antiepileptic drugs – these being the drugs we most widely monitor, with 739 determinations in 2016.
Providing drug information is one of the main activities of the pharmacist in the ICU.8 Johansen et al.3 reported that most of the consultations they received took place while the pharmacist was present in the ICU – thus underscoring the importance of his or her physical presence in the unit.
The pharmacotherapeutic groups most often associated with PI in almost all the studies1,3,6,9,10,15 are anti-infectious agents – group J, drugs acting upon the nervous system – group N, drugs of the digestive tract – group A, blood and hematopoietic organs – group B, and cardiovascular drugs – group C. The distribution was similar in our case, though with the particularity of an important presence of drugs of the digestive tract – group A, which were represented to a greater extent than in other studies (between 9% and 27% versus 27% in our case). We attribute this to the existence of two types of interventions specifically referred to drugs belonging to this pharmacotherapeutic group: ulcer prophylaxis and the prevention of constipation.
The degree of acceptance of the pharmacist interventions was 80%, which is similar to that reported by other studies,1,3,6,9,10,13 with acceptance rates of between 74% and 90%. A question that may be raised here is whether this agreement between pharmacist and intensivist can also be extrapolated to those units in which no such studies are made, i.e., whether the mere fact that such studies are made might imply closeness and confidence between the departments from the start, with concern about drug utilization, which would imply a situation of advantage in the relationship. We are unable to know this, however.
Most of the interventions that were not accepted were referred to administration via the nasogastric tube. This situation resulted in a clinical session imparted by the PRT in the ICU referred to key aspects that could modify the effectiveness of drugs when manipulated for administration through the nasogastric tube. A document in turn was produced with recommendations on nasogastric tube administration of the most widely used drugs, and a survey was made to assess regular practice in the preparation/administration of drugs through the tube and the level of knowledge among the nursing staff in the ICU. As an opportunity for improvement, the multidisciplinary (physicians, pharmacists and nurses) development of a guide on drug administration via the nasogastric tube was proposed, with specific recommendations for each drug, and making the guide available to all the healthcare staff in our center.
With regard to the intervention referred to prophylaxis with laxatives – the second intervention with the highest non-acceptance rate and included in one of the leading pharmacotherapeutic groups in our series (drugs of the digestive tract – group A) – prior to the start of the study and following a literature review,17–19 we established two types of patients amenable to receiving prophylaxis against constipation: patients diagnosed with acute coronary syndrome and patients receiving high-dose opiates. The moderate acceptance of the PI suggests the need to reach consensus and/or establish a protocol referred to prophylaxis with laxatives in the unit.
Our study has a number of limitations (study conduction problems and biases in the methodology employed) that may be associated with the circumstances described in our study:
- 1.
The time period covered is too short, considering the importance of seasonality in critical illness. In this regard, a new study will probably be needed to quantify the clinical impact of the presence of the pharmacist in the ICU or the long-term outcomes of that presence.
- 2.
The exclusion of stays lasting less than a day, and of patients only admitted over the weekend, may have special implications in terms of demographic particulars, the type and severity of disease, and acuteness of the disorder.
- 3.
The lack of a reference period reduces significance of the effectiveness and/or efficacy of the activity of the PRT, since we are unable to establish comparisons. In any case, comparisons would involve historical periods, which would be exposed to biases generated by other intervening variables that are difficult to control.
- 4.
The lack of control precluded assessment of the impact upon hard variables such as mortality, stay, or cost reductions.
On the other hand, consideration is required of the impact for the ICU of the fact that the pharmacist was a PRT rather than a staff specialist. Furthermore, it must be added that the prior presence of a staff specialist, with part time dedication but with physical presence activity in the ICU and participation in the clinical sessions, could have reduced the number of potential interventions compared with units lacking such a presence.
ConclusionsThe adscription of a pharmacist to the ICU, and the performance of PIs, has improved safety and optimized pharmacotherapy in over one-half of the patients admitted to our ICU. The physical presence of the pharmacist generates synergies between the two departments, allowing the detection of opportunities for improvement and the definition of consensus-based protocols aimed at improving patient safety and the effectiveness and efficiency of drug treatments. This, and the high interventions acceptance rate, could justify the implementation of programs of this kind in ICUs.
AuthorshipThe authors of the manuscript: María Teresa Franco-Sereno, Raúl Pérez-Serrano, Ramón Ortiz Díaz-Miguel, María Carmen Espinosa-González, Hasania Abdel-Hadi Álvarez, Alfonso Ambrós-Checa and Marta Rodríguez-Martínez have participated in conduction of the study, and in the drafting of the manuscript.
The study was designed by Raúl Pérez-Serrano and Alfonso Ambrós-Checa. Study data development and compilation were carried out by María Teresa Franco-Sereno.
Supervision and monitoring during conduction of the study were carried out by: Raúl Pérez-Serrano, Ramón Ortiz Díaz-Miguel, María Carmen Espinosa-González, Hasania Abdel-Hadi Álvarez, Alfonso Ambrós-Checa and Marta Rodríguez-Martínez.
All the authors have participated in drafting of the manuscript and have approved the final version of the article.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Franco Sereno MT, Pérez Serrano R, Ortiz Díaz-Miguel R, Espinosa González MC, Abdel-Hadi Álvarez H, Ambrós Checa A, et al. Adscripción del farmacéutico a cuidados intensivos: generando sinergias. Med Intensiva. 2018;42:534–540.