Traumatic spinal cord injury requires a multidisciplinary approach both for specialized treatment of the acute phase and for dealing with the secondary complications. A suspicion or diagnosis of spinal cord injury is the first step for a correct management.
A review is made of the prehospital management and characteristics of the acute phase of spinal cord injury. Respiratory monitoring for early selective intubation, proper identification and treatment of neurogenic shock are essential for the prevention of secondary spinal cord injury. The use of corticosteroids is currently not a standard practice in neuroprotective treatment, and hemodynamic monitoring and early surgical decompression constitute the cornerstones of adequate management.
Traumatic spinal cord injury usually occurs as part of multiple trauma, and this can make diagnosis difficult. Neurological examination and correct selection of radiological exams prevent delayed diagnosis of spinal cord injuries, and help to establish the prognosis.
La lesión medular traumática precisa una acción multidisciplinar, tanto para el tratamiento especializado de la fase aguda como para las complicaciones secundarias. La sospecha y/o el diagnóstico de una lesión medular es el primer paso para establecer un tratamiento correcto.
En esta revisión se aborda el manejo en la fase prehospitalaria y los aspectos característicos de la fase aguda. La monitorización respiratoria para una intubación selectiva precoz, la identificación y el tratamiento correcto del shock neurogénico son fundamentales para la prevención del daño medular secundario. En la actualidad el uso de esteroides no es un estándar en el tratamiento neuroprotector, siendo el control hemodinámico y la descompresión quirúrgica precoz pilares fundamentales.
La lesión medular traumática generalmente se presenta en el contexto de un politraumatismo que puede dificultar el diagnóstico. El examen neurológico y la selección adecuada de pruebas radiológicas evitan que pase desapercibida la lesión medular y otras lesiones asociadas, y ayudan a establecer el pronóstico.
Traumatic spinal cord injury (SI) is a devastating neurological condition. The management of SI demands the contribution of important healthcare resources, since coordinated and multidisciplinary action is required not only for highly specialized care in the acute phase but also for the associated secondary complications that arise over the long term.1
The worldwide incidence of traumatic SI varies greatly: although a global incidence of 2.3 cases/100,000 inhabitants was estimated in 2007,2 the figures found in the literature cover a broad range. In the concrete case of Spain, the few existing epidemiological studies offer global figures of between 0.8 and 2.3 cases/100,000 inhabitants.3,4
Traumatic SI can give rise to a range of neurological problems, including motor and sensory function loss, intestinal and bladder dysfunction, spasticity, neuropathic pain and autonomic dysreflexia.
Acute traumatic SI involves primary and secondary injury mechanisms. The primary mechanism is related to the initial mechanical damage caused by local deformation and energy transformation within the spinal cord at the time of injury, and this damage is irreversible. The secondary mechanisms intervene after the initial traumatic event and lead to tissue destruction during the first hours after injury. These secondary mechanisms include processes such as ischemia, axonal degeneration, vascular dysfunction, oxidative stress, excitotoxicity, demyelination and inflammation leading to cell death, and are potentially avoidable and/or reversible. This concept is crucial for the development of protective strategies aimed at improving the prognosis of patients with acute traumatic SI.
Pre-hospital care and transferDamage to the spine and spinal cord is generally associated to high energy traumatisms such a traffic accidents and falls from a height. However, in elderly people or individuals with pre-existing spinal disorders, SI may be caused by minor traumatisms. Initial assessment at the site of the accident is carried out using the usual ABCDE (Airway, Breathing, Circulation, Disability, Exposure) sequence. Protection of the spine is important, though adequate management of the airway, bleeding control and other forms of critical care are also crucial.
Basic neurological exploration, assessing patient capacity to mobilize all four extremities (including feet and hands) contributes to the diagnosis, particularly if early intubation is needed. The presence of certain clinical signs can cause us to suspect acute SI (Table 1).
Signs suggestive of spinal cord injury.
| Weakness or paralysis of the extremities |
| Sensitivity alterations of the trunk or extremities |
| Speech difficulty (hypophonia) |
| Abdominal breathing |
| Hypotension and paradoxical bradycardia |
| Flexed elbows position |
| Spinal pain or deformity |
| Paresthesias. Electric discharge sensation |
| Absence of pain in the presence of predictably painful injuries |
| Priapism |
The current recommendation in reference to the immobilization of trauma patients is to apply selective immobilization, identifying those individuals that may benefit from this measure.5–7 The devices used for immobilization are associated to complications such as increased intracranial pressure, difficulties accessing the airway, restricted lung function, pain, agitation, pressure ulcers and the prolongation of patient transfer time. The use of a cervical collar does not completely limit mobility of the cervical spine. When vertebral damage is suspected, the entire spinal column is to be immobilized, since the presence of additional non-contiguous vertebral injury occurs in up to 20% of the cases.8 Although strong supporting evidence is lacking9,10 and negative effects may even occur, the recommendation to immobilize patients with suspected vertebral-spinal cord injury is based on anatomical, mechanical and clinical considerations—the aim being to prevent the development or worsening of SI in the presence of unstable vertebral damage.11
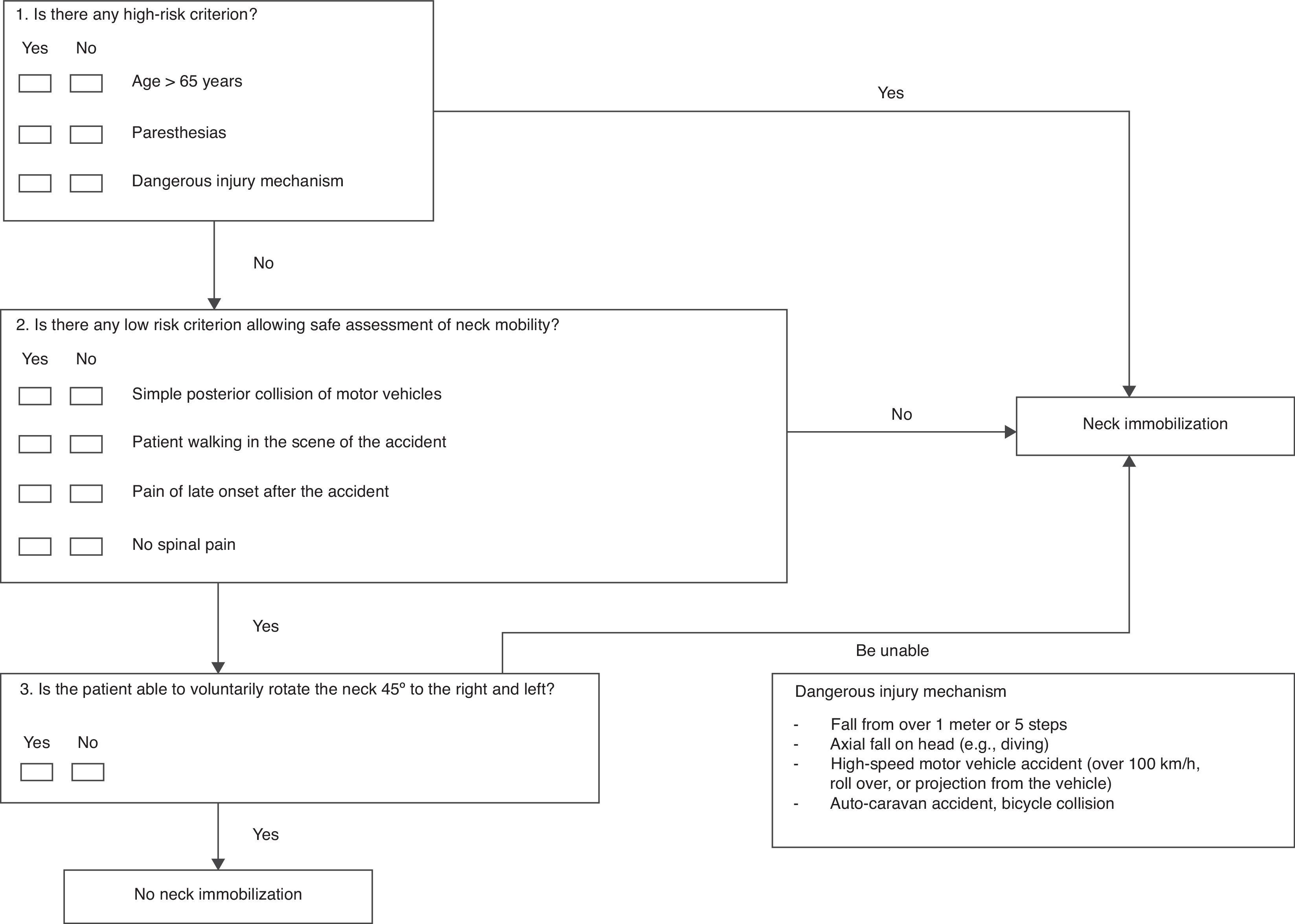
Immobilization is not indicated in patients with penetrating trauma in the absence of neurological symptoms.6,7,12 Algorithms have been developed in the case of closed trauma patients, in an attempt to avoid indiscriminate immobilization.13–16 The most widely used criteria in the out-hospital emergency care setting are those proposed by the National Emergency X-Radiography Utilization Study (NEXUS)14 and the Canadian C-Spine Rule (CCSR).15 The NEXUS establishes 5 low risk criteria which when met could discard cervical damage (Table 2). The CCSR combines high and low risk criteria and the capacity to rotate the head 45°. In this regard, the patient is without risk and therefore will not need immobilization if one low risk criterion is met and the patient can voluntarily rotate the head 45°17 (Table 3). These or similar immobilization algorithms have been incorporated to emergency team intervention protocols throughout the world.5,17–21 The National Institute for Health and Care Excellence (NICE) guide on the assessment and management of spinal injuries favors the CCSR criteria in evaluating the cervical spine, though its immobilization recommendations combine NEXUS and CCSR criteria. Although it is agreed that a selective immobilization algorithm should be applied, there is no consensus regarding which algorithm is best. In the United Kingdom, the Faculty of Pre-Hospital Care drafted a consensus document suggesting that an algorithm similar to that proposed by the NEXUS might be appropriate, though this remains to be confirmed.6 There is controversy in the literature regarding inclusion of the injury mechanism as a risk criterion; whether different algorithms should be used in conscious and unconscious patients; and regarding which patients are amenable to auto-rescue measures.6,22,23
The usual immobilization technique involves the use of a spinal table with straps and head fixation, with the use of a cervical collar.7,24 However, this practice—a standard for many emergency care teams—is changing. The immobilization methods may be different during rescue, retrieval and transport, and can be modified according to the clinical condition of the patient, placing priority on the ABC approach. In the management of trauma patients, spinal protection maneuvering should be the norm until clinical evaluation can be made. Immobilization devices may be difficult to fit in patients with certain characteristics (short neck or spinal deformities) or in individuals that fail to cooperate or are restless, and fitting moreover takes time. Under these circumstances, and when “rapid rescue” is needed, manual immobilization can be maintained,6,21 followed by fitting of a cervical collar of adequate size, if indicated. Mobilization is to be carried out trying to preserve the vertebral axis, applying linear traction upon the cervical spine and avoiding deformities of the spine and movements of the head and neck. The High Arm In Endangered Spine (HAINES) or modified HAINES technique is currently more widely recommended than the classical log-roll procedure, since it causes less mobilization of the spine.25 The long spinal table, Kendricks rescue device and scissor-type stretcher can also be used during rescue. The National Association of EMS Physicians and the American College of Surgeons Committee on Trauma limit use of the spinal table to certain situations,23 regarding it as a “rescue” device, rather than a transport device.6 For transport purposes, the vacuum mattress combined with a cervical collar affords similar or better immobilization compared with the spinal table, and is more comfortable.21,26 Transport can also be safely made using the ambulance stretcher, correctly fastening the patient with straps together with the cervical collar.23 In children, the disproportion between the head and rest of the body may require the addition of cushioning in order to preserve body posture and avoid neurological deterioration. The same applies to individuals with spinal kyphosis. Mobilization and transfer should be limited as far as possible. Inter-exchanging immobilization means are therefore needed between the pre-hospital and in-hospital settings.
The suspicion or confirmation of SI, together with safe transfer to hospital, constitutes the first step in correct patient management. In the event of life-threatening situations, the patient should be taken to the nearest hospital center, followed by referral to a center specialized in SI as soon as possible (preferably within the first 24h). The choice of transport will depend on the available means, patient stability and the distance to the hospital center.27 Transport by air does not necessarily have to be used, though it may be recommended for distances of over 80km.28
ABC and resuscitationOnce in the hospital setting we must continue with the immobilization and life support measures, with the conduction of radiological evaluations and the indication of specific treatments. The management of life-threatening situations takes priority over any radiological studies.
As in all trauma cases, acute management of the patient with SI requires preservation of the airway, breathing and circulation.
Airway and breathingContinuous monitoring of respiratory failure is required in all patients with cervical SI.29 Effective respiratory muscle function is lacking in the case of C1–C2 injuries. Lesions at C3–C4 level in turn produce bilateral phrenic nerve paralysis, and ventilation becomes dependent upon the accessory muscles. Consequently, patients with complete motor damage above C5 almost invariably require ventilatory support. Tetraplegic patients with adequate ventilation at the expense of important respiratory labor must be intubated and placed under mechanical ventilation without delay. Injuries below C5 cause paralysis of the intercostal and abdominal muscles, and most affected individuals will require ventilatory support.
The loss of respiratory capacity results from exhaustion of the ventilatory muscles, ascending spinal cord edema or bleeding, the accumulation of secretions, atelectasis, other associated traumatisms, or other patient conditions. Lesions below T5 usually do not produce respiratory failure of neuromuscular origin.
Lung edema secondary to traumatic SI may be cardiogenic (due to high catecholamine and beta-endorphin levels) and/or non-cardiogenic (pulmonary hyperpermeability). Echocardiographic studies and the new cardiopulmonary assessment devices may be of help in determining the predominant mechanism. Pulmonary thromboembolism and pneumonia are complications that tend to manifest after the first 24h.
Endotracheal intubation may be particularly difficult in patients with cervical SI. Intubation is sometimes needed before the lesion and is location are confirmed. Consequently, patients requiring to be intubated following trauma are to be managed as cervical SI cases, ensuring preservation of the airway with as little movement of the cervical spine as possible. Emergency intubation in the case of suspected or confirmed cervical SI must be carried out with a rapid induction sequence (to reduce coughing and spontaneous movements), with linear manual traction stabilization of the spine.24 Traction must be done carefully in order to avoid distraction—particularly in lesions affecting the occipitocervical junction. In patients with cervical immobilization, use of the Airtraq device reduces the risk of failure.30 There is a lack of evidence on the usefulness of other intubation devices.
If securing the airway and ventilatory support are not initially required, the need for intubation should be monitored by means of PCO2 and spirometry with the measurement of vital capacity (which shows excellent correlation to other lung function tests) and peak inspiratory pressure (PImax).31,32 This offers a simple and global evaluation of inspiratory muscle strength, and constitutes one of the best point of care markers. In general terms, PImax estimates the strength of the inspiratory muscles (diaphragm), being recorded under residual volume and with maximum inspiratory effort. Ventilatory failure most often occurs beyond the fourth day after trauma. This is relevant for monitoring and for the decision to maintain postoperative intubation in patients undergoing early surgery – particularly those subjected to neck surgery.
Circulation: prevention and treatment of hypotensionAdequate and early resuscitation with fluids is a priority concern in the treatment of hypotension, in order to maintain adequate perfusion and avoid secondary damage to the central nervous system.33 The most appropriate resuscitation target and optimum mean blood pressure (MBP) value for maintaining spinal cord perfusion are not clear. Non-controlled studies using fluids and vasopressor drugs to secure an MBP of 85mmHg during a minimum of 7 days in patients with acute SI have reported favorable results.34–36 Many studies underscore that hypotension must be identified, seeking the cause and starting resuscitation with fluids. Further studies are needed to define the ideal MBP and the role of fluids and drug support in this regard.
The initial base defect or lactate concentration can be used to assess shock and the need for resuscitation with fluids.37
Neurogenic shock is common in patients with acute SI above level T6.38 However, before assuming that SI is the origin of hypotension, other causes of hemodynamic instability in trauma patients should be discarded, such as bleeding, pneumothorax, myocardial damage, cardiac tamponade, sepsis related to abdominal injuries, adrenal gland failure, etc. The physical examination and symptoms are scantly sensitive in patients with SI; thoracoabdominopelvic imaging studies are therefore required in order to rule out other possible causes of hypotension.
Neurogenic shock is secondary to sympathetic denervation, which leads to arterial vasodilatation and blood sequestration in the venous compartment, together with interruption of cardiac sympathetic innervation (T1–T4) with unopposed vagal nerve activity that promotes bradycardia and reduces cardiac contractility. Neurogenic shock is therefore characterized by a drop in arterial pressure and systemic vascular resistances, with a variable heart rate response. Spinal injury above T6 should be suspected in all polytraumatized patients with hypotension and bradycardia, particularly in the absence of signs of peripheral vasoconstriction.
Experimental studies have shown that hypotension and hypovolemia are deleterious for the damaged spinal cord, contributing to hypoperfusion and perpetuating the secondary damage. Higher lesion levels are correlated to more severe hypotension. It is essential to restore intravascular volume and, if necessary, start vasopressor drug treatment.36 Bradycardia-dysrhythmias can lead to hypotension and asystolia – this being more frequent in the first two weeks after injury. Hypoxia, laryngeal or tracheal manipulation maneuvers, and hypothermia, worsen bradycardia. Atropine must be at hand whenever airway manipulation is intended in patients with cervical SI. The need for chronotropic drugs (atropine, adrenalin, noradrenalin) or phosphodiesterase inhibitors (aminophylline, theophylline) is more frequent in patients with cervical SI. The β2 receptors are an attractive target for modulating vagal nerve activity and thus raising the heart rate in the absence of significant risks.39 A cardiac pacemaker to deal with bradycardia is reserved for those patients with symptomatic bradycardia that fails to respond to drug treatment.
TemperatureThe autonomic nervous system is interrupted in cases of injury above T6, giving rise to alterations in thermoregulation due to the incapacity of the hypothalamus to control temperature due to the loss of vasomotor control. The affected patients can present hypothermia as well as incapacity to dissipate body heat.40 Temperature monitoring is essential during management of the acute phase.
Diagnosis of acute traumatic spinal cord injuryExplorationThe immediate effects of complete SI include loss of movement and sensitivity below the level of the lesion. Furthermore, the patient may suffer neurogenic shock, flaccid paralysis of the bladder and intestine with urinary retention and paralytic ileus, and alterations of all systems below the level of the lesion. Initial phase spinal shock is also observed, characterized by a loss of reflex activity and flaccidity below the level of the lesion. This period usually lasts days or weeks, and is subsequently followed by spasticity.
The clinical diagnosis starts with the basic neurological examination. Loss of consciousness and the mechanism of injury are to be evaluated as far as possible. If the patient is conscious, detailed motor and sensory assessment is indicated. However, such evaluation is often not possible in polytraumatized patients, which moreover may be sedated or intubated. In these circumstances, it is therefore advisable to manage the patient as if vertebral-spinal cord injury were effectively present. Table 1 describes the alarm signs that may cause us to suspect the presence of such damage at initial exploration.
The neurological exploration is made according to the International Standards for the Neurological Classification of Spinal Cord Injury (http://asia-spinalinjury.org/wp-content/uploads/2016/02/International_Stds_Diagram_Worksheet.pdf) of the American Spinal Injury Association (ASIA) and the International Spinal Cord Society.41 This system describes the level and extent of the lesion, based on a systematic evaluation of sensory and motor function. In addition to being of prognostic value, this assessment based on the ASIA scale is of use in monitoring the patient neurological course. This evaluation has been protocolized to be carried out upon patient admission, after 72h, after one month and at discharge, and also in certain situations in which worsening of SI may be suspected. Table 4 offers basic definitions of terms used in assessing SI.
Definitions of basic terms and ASIA classification.
| Tetraplegia | Loss of motor and/or sensory function in the cervical segments of the spinal cord secondary to damage of the neural elements within the spinal canal. It causes functional disorders of the arms, trunk, legs and pelvic organs. |
| Paraplegia | Loss of motor and/or sensory function in the thoracic, lumbar or sacral segments of the spinal cord secondary to damage of the neural elements within the spinal canal. It causes functional disorders of the trunk, legs and pelvic organs. |
| Dermatome | Skin area innervated by the sensory axons of each segmental nerve (root). |
| Myotome | Muscle innervated by the motor axons of each segmental nerve (root). |
| Motor level | The most caudad key muscle group with a muscle balance of 3/5 or more, provided the muscle balance of the higher positioned muscles is 5/5 |
| Sensory level | The most caudad dermatome with normal light tactile and pain sensitivity on both sides of the body. |
| Neurological level | The most caudad level where motor and sensory function are normal. |
| Skeletal level | The level of greatest vertebral damage as evidenced by the radiological studies. |
| Motor index | Sum of the motor function scores (maximum 100). |
| Sensory index | Sum of the scores of each dermatome; 28 dermatomes are evaluated on each side of the body, with a total of 112 for pain and 112 for tactile sensation. |
| Complete lesion | Absence of motor and sensory function in the inferior sacral segments (S4–S5). |
| Incomplete lesion | Preservation of motor and/or sensory function below the neurological level, including sacral segments S4–S5. |
| Zone of partial preservation (ZPP) | Referred to dermatomes or myotomes below the neurological level that remain partially innervated; the most caudad segment with some motor or sensory function defines the extent of the zone of partial preservation; only in complete lesions. |
| ASIA classification | |
| A: complete lesion | Absence of motor and sensory function extending to sacral segments S4–S5. |
| B: incomplete sensory | Preservation of sensory function extending to sacral segments S4–S5 and with absence of motor function. |
| C: incomplete motor | Preservation of motor function below the neurological level, and over one-half of the muscles below the neurological level have a muscle balance of under 3. |
| D: incomplete motor | Preservation of motor function below the neurological level, and at least one-half of the muscles below the neurological level have a muscle balance of 3 or more. |
| E: normal | Normal motor and sensory function in all segments. |
In relation to the sensory exploration, we assess 28 dermatomes in each side of the body (C2 to S4–S5, considering the latter as a single dermatome). For each dermatome, we assess sensitivity to pain (needle prick) and superficial tactile sensitivity (cotton friction) based on a three-point scale (0=none; 1=impaired; 2=normal). Deep anal sensitivity is also evaluated by rectal digital testing, registered as either present or absent.
We likewise explore muscle balance in 5 key muscle groups of the upper extremity and in 5 of the lower extremity (in each side of the body). Muscle balance is scored from 0 to 5 according to the Medical Research Council scale. Elbow flexion (C5), carpal extension (C6), elbow extension (C7) and flexion of the fingers (C8) and abductors of the little finger (T1) are evaluated in the upper extremity. Hip flexion (L2), knee extension (L3), ankle dorsiflexion (L4), extension of the big toe (L5) and ankle plantar flexion (S1) are evaluated in the lower extremity. As part of the motor evaluation, we assess voluntary contraction of the external anal sphincter through rectal digital testing, registered as either present or absent.
The sensory level is the most caudad intact dermatome referred to both sensitivities (tact and pain). The motor level in turn is defined by the most caudad key muscle with a muscle score of at least 3, provided the key muscles above that level are intact (muscle score 5). In the regions where there are no dermatomes for evaluation, the motor level is taken to be the same as the sensory level.
The neurological level of the lesion refers to the most caudad segment of the spinal cord in which both motor and sensory function are bilaterally normal. It is the most cephalic of the sensory and motor levels.
ASIA disability scaleSpinal cord injury is generally classified as “complete” or “incomplete”, based on the presence of sacral preservation. The latter refers to the presence of sensory or motor function in the most caudad sacral segments (preservation of light tact or pain sensation in S4–S5, deep anal sensation, or voluntary contraction of the anal sphincter).
A complete lesion is defined as the absence of sacral preservation (no sensory or motor function in segments S4–S5), while an incomplete lesion is defined as the presence of sacral preservation (some preserved sensory or motor function in segments S4–S5).
The ASIA scale comprises 5 grades (Table 4). Grade A corresponds to complete SI; grades B, C and D are incomplete lesions of different grades; and grade E is indicative of normal motor and sensory function (this grade being applied provided the patient has suffered some degree of spinal cord injury that has recovered).
Clinical syndromes- •
Central spinal cord. Central spinal cord injury is the most frequent type of incomplete lesion. It is characterized by greater motor weakness in the upper extremities than in the lower extremities, sphincter alterations and variable degree of sensory involvement. These presentations are more common in patients with previous degenerative changes of the vertebras that suffer hyperextension.42
- •
Brown-Sèquard (cord hemisection). This presentation is characterized by loss of deep sensitivity ipsilateral to the lesion, with contralateral alteration of heat-pain sensitivity. Pure cord hemisection is rare in traumatic SI, combining Brown-Sèquard symptoms and central cord symptoms (Brown-Sèquard plus syndrome).
- •
Anterior spinal cord. This presentation can result from direct damage to the anterior part of the spinal cord due to retropulsion of a bone or disc fragment, or can be caused by damage to the anterior spinal artery. It is characterized by paralysis and alteration of heat-pain sensitivity, with preservation of the posterior cords (light tact sensation and positional sensitivity).
- •
Medullary cone. These are lesions of the sacral spinal cord (cone) and lumbar nerve roots within the neural canal. Medullary cone injuries result in paralysis of the bladder, intestine and lower extremities. Depending on the affected level, these cases can manifest as mixed superior and inferior motor neuron syndromes.
- •
Cauda equina. These are lesions of the lumbosacral roots below the medullary cone, located within the neural canal. The clinical manifestations are similar to those of medullary cone injury, making it sometimes difficult to distinguish between the two presentations.
The indications of radiological exploration in diagnosing traumatic SI depend on the circumstances of trauma, the need to perform tests for diagnosing associated lesions, and the availability of techniques in each individual center.
Clinical protocols contemplated in the Eastern Association of Trauma39 guides allow us to discard cervical spine injuries in asymptomatic patients without performing imaging explorations. The NEXUS14 and CCSR criteria15 are very sensitive in discarding significant cervical injuries without conducting radiological studies. Patients with spinal pain, neurological symptoms, and unconscious trauma patients, require radiological evaluation. In patients with masking associated injuries or short-duration altered consciousness, we can decide to use spinal protection measures until the mentioned injuries are treated or sufficient consciousness is regained to allow adequate clinical assessment. However, if urgency is a priority concern, these individuals must be evaluated under the same criteria as in unconscious polytraumatized patients.43
In patients requiring a radiological study, it is advisable to evaluate the entire spine, since the incidence of multiple vertebral fractures is close to 20%.8 Plain X-rays with anteroposterior and lateral projections of the entire spine were traditionally indicated, together with transoral projection in the cervical spine, though the current tendency is to perform computed axial tomography (CAT) with sagittal and coronal reconstructions.44 Spiral CAT, while more expensive, is more sensitive and specific than conventional radiology44,45: it allows easier identification of fractures that might go undetected on plain X-rays; ensures correct visualization of the occipitocervical and cervicothoracic junctions; affords full visualization of the entire spine with reconstructions allowing improved characterization of canal invasion; and provides information of help in surgical treatment. Plain X-rays studies in flexion and extension are useful for assessing instability secondary to ligamentous damage, but should be avoided in the acute phase of patients with neurological deficits.
Emergency magnetic resonance imaging (MRI) is usually difficult to perform in acute polytraumatized patients. Emergency MRI is indicated in traumatic SI in those cases where the neurological defects cannot be explained by the radiological findings, or when neurological deterioration occurs, with a view to discarding the possible presence of epidural hematoma or other causes indicating emergency surgical treatment. Magnetic resonance imaging should be performed as soon as allowed by the clinical condition of the patient, because it is able to detect damage to soft tissues and ligaments in the setting of neurological damage, and is moreover able to adequately characterize the type of SI involved (contusion, edema, bleeding, cord sectioning). This information in turn can be complemented by clinical evaluation based on the ASIA scale in order to establish the prognosis of SI. In patients with cord sectioning or intramedullary hemorrhage, neurological recovery generally can be expected to be poor or inexistent, while in individuals with spinal cord edema or contusions, greater recovery may be observed.46,47 Furthermore, MRI allows us to evaluate canal involvement, the degree of spinal cord compression, and the length of the lesion – these factors also being important for predicting the severity of the damage and patient recovery.48,49
Evaluation of associated injuriesThe mechanism underlying SI may involve high or low intensity forces. High energy traumatisms are the presentations most often associated to damage at other levels, and in close to 37% of the cases multiple fractures are present.50,51 The type of lesion associated to SI varies according to the level of the lesion—head injuries being more frequent in patients with cervical lesions, while thoracoabdominal injuries are more often seen in patients with SI at dorsal level.52
In patients with suspected or confirmed SI, in the same way as in all trauma cases, tertiary evaluation of the traumatism is required in order to reduce the incidence of undetected lesions, since physical examination can be conditioned by the level of the lesion, due to the existence of sensitivity and motor alterations.53,54 In other words, following initial resuscitation, identification and classification of all the lesions is a priority concern. In this respect, we must repeat the entire physical exploration, evaluate the mechanism of injury, the basal characteristics (age, comorbidity and genetic predisposition), and the level of SI.55 This protocol is completed with laboratory tests, expert review of the imaging studies, and the conduction of complementary studies in the event of new clinical findings. Assessment is time dependent: it is advisable to perform evaluation in the first 24h after trauma and to repeat it once the patient is able to communicate.53,54
The main considerations in clinically evaluating the associated lesions are:
- a.
Document the presence of traumatic brain injury, particularly in cervical injuries, in order to perform a correct neurological evaluation and plan rehabilitation. This should be done early, based on the Glasgow coma scale (GCS).56
- b.
Associated thoracoabdominal injuries are to be considered in all patients with SI, using imaging techniques. The first diagnostic approach in unstable patients is to perform Focused Abdominal Sonography for Trauma (FAST), though the technique of choice is CAT, whenever allowed by the clinical condition of the patient or when ultrasound proves positive.57,58
- c.
In fractures of the extremities-pelvis, the main concern should be to secure early stabilization in order to maintain a good range of mobility and facilitate rehabilitation.59
- d.
In very high energy impacts we must discard aortic damage, and in the event of added cervical luxation/fracture (fundamentally at level C1–C3), we must discard cerebrovascular injuries using AngioCAT/MRI.60,61
Together with tertiary evaluation of the SI, we must quantify severity and estimate the probability of survival based on trauma severity scores, in order to facilitate the decision-making process and reduce morbidity-mortality.62,63 The trauma scales used should assess the anatomical lesion, the physiological changes that occur, and the functional reserve of the patient.64,65
Multiple scores have been developed, grouped according to different aspects (anatomical, physiological and combined scores). In the case of patients with SI, the most widely cited and used instruments are the Injury Severity Score,66 the Trauma Score Revised67 and the TRISS,68 which together with inclusion of the GCS allow complete evaluation – determining which patients may benefit from rehabilitation, and contributing to optimize resource utilization. Furthermore, in trauma patients with SI admitted to the Intensive Care Unit (ICU), the Acute Physiology and Chronic Health Evaluation (APACHE) and Sequential Organ Failure Assessment (SOFA) scales have been validated for establishing the prognosis.69,70
In evaluating the associated lesions in spinal cord patients, adequate tertiary assessment and the definition of a functional prognosis based on the combination of different scales are essential, since no universally accepted score has been established.
NeuroprotectionCurrent management includes surgical decompression, hemodynamic control, and methylprednisolone in selected cases. However, these early treatment measures are associated to only modest functional recovery.
The search for an effective neuroprotective strategy capable of preventing secondary lesions in the context of acute traumatic SI remains a priority concern both in clinical practice and in basic research.
Corticosteroids formed part of the initial therapeutic repertoire. Methylprednisolone is believed to reduce membrane peroxidation and lessen inflammation. It exerts immune modulating effects, inhibiting neutrophil and macrophage infiltration of the spinal cord, which might improve the functional outcomes. Three clinical trials and other non-randomized studies, with important limitations, have been carried out to evaluate the use of methylprednisolone following traumatic SI.71–73 Based on the existing evidence, the generalized administration of high dose methylprednisolone in traumatic SI is not recommended.74,75 The use of the drug must therefore be carefully evaluated on the basis of the patient characteristics and intercurrent disease conditions, in view of its side effects in terms of infections, respiratory alterations, gastrointestinal bleeding and mortality.74,76 Specifically, there is no evidence that the administration of corticosteroids has a beneficial effect in cases of complete traumatic SI. In the event of non-stabilized acute SI—or in the presence of neurological deterioration—the administration of corticosteroids could be started in the first hours, with short dosing schemes and taking the potential side effects into account.77
Current investigations in acute traumatic SI are exploring the use of certain treatments with neuroprotective intent (riluzole, minocycline, G-CSF, FGF-2 and polyethylene glycol), as well as neuroregenerative strategies (chondroitinase ABC, self-assembling peptides and Rho inhibition). Many cell therapies (embryonic stem cells, neural stem cells, induced pluripotent stem cells, mesenchymal stromal cells, Schwann cells, olfactory ensheathing cells and macrophages) have also been found to be promising.78–80
However, due to the many factors that condition lesion progression, restoration of the damaged spinal cord will probably require a number of repair strategies applied in combination.79
Some experimental studies have suggested that cooling attenuates the secondary lesion mechanisms.81 Initial clinical studies involving direct cooling of the spinal cord during surgery have not revealed benefits. However, a recent study investigated the use of hypothermia (33°C), with favorable results.82 In any case, the existing evidence is too weak to recommend systemic hypothermia.83
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Galeiras Vázquez R, Ferreiro Velasco ME, Mourelo Fariña M, Montoto Marqués A, Salvador de la Barrera S. Actualización en lesión medular aguda postraumática. Parte 1. Med Intensiva. 2017;41:237–247.