Acute heart failure (AHF) is the leading cause of consultation and admission in Hospital Emergency Services (HES).1 The usefulness of morphine in treating AHF is unclear.2 The guidelines of the European Society of Cardiology3 recommend the use of this drug in the early phases of the management of patients with AHF, with the purpose of alleviating dyspnea, agitation, anxiety or chest pain. While these guidelines3 warrant the use of opiates in the treatment of AHF, the American Heart Association opines otherwise.4 The primary objective of this study was therefore to determine the impact of early phase morphine upon in-hospital mortality among patients with AHF seen in a HES.
A retrospective analysis was made of 991 patients with AHF seen in the HES of a third-level hospital during the period between November 2013 and December 2015. The study was approved by the local Ethics Committee. The initial treatment decision was exclusively fundamented upon clinical criteria as established by the physician supervising patient care in the HES, based on the recommendations of the European Society of Cardiology.3 Demographic, clinical and laboratory test data were collected. The demographic and other baseline parameters were subjected to a basic descriptive analysis. Continuous variables were reported as the mean±standard deviation (SD) in the presence of a normal distribution, and as the median (percentiles 25 and 75) in the case of a non-normal distribution. In turn, categorical variables were reported as absolute (n) and relative frequencies (%). Quantitative variables were compared using the Student t-test or Mann–Whitney U-test, while associations between qualitative variables were explored using the Pearson chi-squared test or Fisher exact test. Survival analysis was based on a Cox regression model including age, gender and left ventricular ejection fraction, as well as those variables found to be significant (p<0.05) in the univariate analysis. The proportional hazard assumption was tested using Schoenfeld residual analysis. The SPSS® version 20 statistical package (SPSS Inc., Armonk, NY, USA) was used throughout.
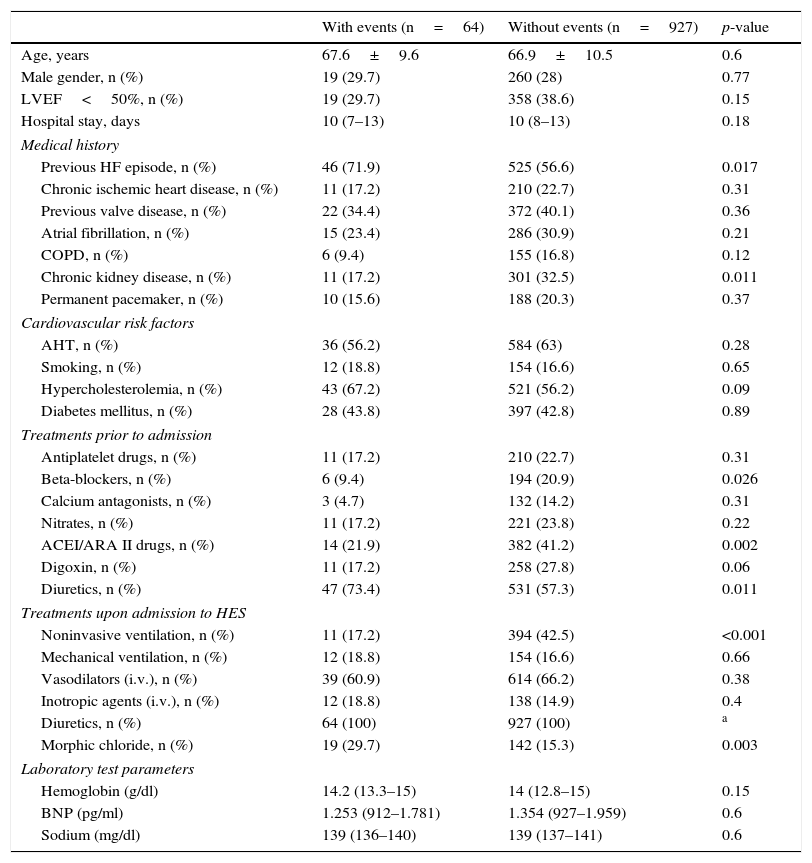
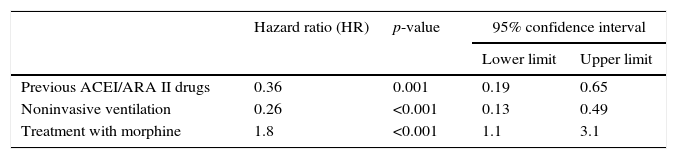
The characteristics of the study population are described in Table 1. Of the 991 patients with AHF, 64 (6.4%) met the primary endpoint (in-hospital mortality). In the comparison of groups, those patients that met the primary endpoint had a greater prevalence of previous heart failure and a lesser prevalence of chronic kidney disease. With regard to home treatment, the patients with events were treated in lesser proportion with beta-blockers and angiotensin converting enzyme inhibitors, and in greater proportion with diuretics. With regard to emergency care, the patients with events received less invasive ventilation and a greater proportion of treatments with intravenous morphine. After adjusting for other covariables (age, gender, previous episode of heart failure, chronic kidney disease, home treatment with beta-blockers and diuretics, left ventricular ejection fraction <50%), the Cox survival analysis identified intravenous morphine as an independent predictor of the primary endpoint (Table 2).
Patient classification according to the appearance of events.
| With events (n=64) | Without events (n=927) | p-value | |
|---|---|---|---|
| Age, years | 67.6±9.6 | 66.9±10.5 | 0.6 |
| Male gender, n (%) | 19 (29.7) | 260 (28) | 0.77 |
| LVEF<50%, n (%) | 19 (29.7) | 358 (38.6) | 0.15 |
| Hospital stay, days | 10 (7–13) | 10 (8–13) | 0.18 |
| Medical history | |||
| Previous HF episode, n (%) | 46 (71.9) | 525 (56.6) | 0.017 |
| Chronic ischemic heart disease, n (%) | 11 (17.2) | 210 (22.7) | 0.31 |
| Previous valve disease, n (%) | 22 (34.4) | 372 (40.1) | 0.36 |
| Atrial fibrillation, n (%) | 15 (23.4) | 286 (30.9) | 0.21 |
| COPD, n (%) | 6 (9.4) | 155 (16.8) | 0.12 |
| Chronic kidney disease, n (%) | 11 (17.2) | 301 (32.5) | 0.011 |
| Permanent pacemaker, n (%) | 10 (15.6) | 188 (20.3) | 0.37 |
| Cardiovascular risk factors | |||
| AHT, n (%) | 36 (56.2) | 584 (63) | 0.28 |
| Smoking, n (%) | 12 (18.8) | 154 (16.6) | 0.65 |
| Hypercholesterolemia, n (%) | 43 (67.2) | 521 (56.2) | 0.09 |
| Diabetes mellitus, n (%) | 28 (43.8) | 397 (42.8) | 0.89 |
| Treatments prior to admission | |||
| Antiplatelet drugs, n (%) | 11 (17.2) | 210 (22.7) | 0.31 |
| Beta-blockers, n (%) | 6 (9.4) | 194 (20.9) | 0.026 |
| Calcium antagonists, n (%) | 3 (4.7) | 132 (14.2) | 0.31 |
| Nitrates, n (%) | 11 (17.2) | 221 (23.8) | 0.22 |
| ACEI/ARA II drugs, n (%) | 14 (21.9) | 382 (41.2) | 0.002 |
| Digoxin, n (%) | 11 (17.2) | 258 (27.8) | 0.06 |
| Diuretics, n (%) | 47 (73.4) | 531 (57.3) | 0.011 |
| Treatments upon admission to HES | |||
| Noninvasive ventilation, n (%) | 11 (17.2) | 394 (42.5) | <0.001 |
| Mechanical ventilation, n (%) | 12 (18.8) | 154 (16.6) | 0.66 |
| Vasodilators (i.v.), n (%) | 39 (60.9) | 614 (66.2) | 0.38 |
| Inotropic agents (i.v.), n (%) | 12 (18.8) | 138 (14.9) | 0.4 |
| Diuretics, n (%) | 64 (100) | 927 (100) | a |
| Morphic chloride, n (%) | 19 (29.7) | 142 (15.3) | 0.003 |
| Laboratory test parameters | |||
| Hemoglobin (g/dl) | 14.2 (13.3–15) | 14 (12.8–15) | 0.15 |
| BNP (pg/ml) | 1.253 (912–1.781) | 1.354 (927–1.959) | 0.6 |
| Sodium (mg/dl) | 139 (136–140) | 139 (137–141) | 0.6 |
BNP: brain natriuretic peptide; COPD: chronic obstructive pulmonary disease; LVEF: left ventricular ejection fraction; AHT: arterial hypertension; HF: heart failure; i.v.: intravenous; HES: Hospital Emergency Service.
Cox survival analysis.
| Hazard ratio (HR) | p-value | 95% confidence interval | ||
|---|---|---|---|---|
| Lower limit | Upper limit | |||
| Previous ACEI/ARA II drugs | 0.36 | 0.001 | 0.19 | 0.65 |
| Noninvasive ventilation | 0.26 | <0.001 | 0.13 | 0.49 |
| Treatment with morphine | 1.8 | <0.001 | 1.1 | 3.1 |
After adjusting for the following variables: age (p=0.57), gender (p=0.68), previous HF (p=0.27), previous treatment with beta-blockers (p=0.1) or diuretics (p=0.33), chronic kidney disease (p=0.39) and ventricular dysfunction (p=0.3).
Our study is original in that it demonstrates for the first time in a Spanish cohort of AHF patients seen in a HES that the use of intravenous morphine in the early phases of AHF is associated to in-hospital mortality. The guidelines of the American Heart Association, published in 2013, do not mention morphine among the therapeutic options in patients with AHF.5 A retrospective analysis of the Acute Decompensated Heart Failure National Registry (ADHERE), with a total of 147,362 patients, showed the administration of morphine in AHF to be an independent predictor of in-hospital mortality (odds ratio [OR]: 4.8; 95% confidence interval [95%CI]: 4.52–5.18; p<0.001).6 Morphine is used in AHF because of its expected anxiolytic and vasodilator effects. However, over the last decade there has been controversy regarding the benefits and risks of this drug in AHF.2 At present, our study supports the need to design a randomized trial in order to establish the effects, and particularly the risks, of using morphine for the treatment of AHF. In this regard, our group will soon start a phase IV clinical trial (EudraCT number 2016-000884-17 – Study MIMO/2016), with the aim of determining whether intravenous morphine in patients with AHF reduces or increases the in-hospital mortality risk.
Financial supportPart of this study has been financed by Internal Projects Call 2016 of the Universidad Europea de Canarias.
Please cite this article as: Dominguez-Rodriguez A, Avanzas P, Burillo-Putze G, Abreu-Gonzalez P. Influencia del tratamiento con morfina en la mortalidad intrahospitalaria de pacientes con insuficiencia cardíaca aguda. Med Intensiva. 2017;41:382–384.