Drug rash with eosinophilia and systemic symptoms (DRESS) occurs in one out of every 1000–10,000 individuals exposed to drugs.1 The precise physiopathological mechanism involved is not clear, though two main theories have been proposed2: (a) the development of an allergic hypersensitivity reaction in which the drug substance acts as a hapten or allergen; and (b) the absence of epoxide hydroxylase–an enzyme that mediates the detoxification of drugs that trigger the immune response. The disorder is often associated to reactivation of viruses belonging to the herpes family (HHV-6, HHV-7, Epstein–Barr and cytomegalovirus), though the relationship of this phenomenon to the etiopathogenesis of DRESS is not known.1–5
The drugs most often associated to this syndrome are the aromatic antiepileptic agents (carbamazepine, phenobarbital) and allopurinol, though many others have also been implicated in DRESS, such as antibiotics, antituberculosis drugs2 (especially rifampicin),3 nonsteroidal antiinflammatory drugs (NSAIDs) and antivirals.
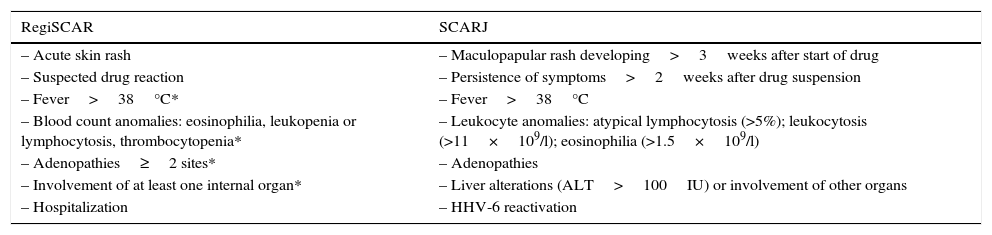
A series of diagnostic criteria for DRESS have been developed over the last decade: the European Registry of Severe Cutaneous Adverse Reactions to Drugs (RegiSCAR)6 and the Japanese Registry of Severe Cutaneous Adverse Reactions to Drugs (SCARJ)7 (Table 1).
Diagnostic criteria of DRESS.
| RegiSCAR | SCARJ |
|---|---|
| – Acute skin rash | – Maculopapular rash developing>3weeks after start of drug |
| – Suspected drug reaction | – Persistence of symptoms>2weeks after drug suspension |
| – Fever>38°C* | – Fever>38°C |
| – Blood count anomalies: eosinophilia, leukopenia or lymphocytosis, thrombocytopenia* | – Leukocyte anomalies: atypical lymphocytosis (>5%); leukocytosis (>11×109/l); eosinophilia (>1.5×109/l) |
| – Adenopathies≥2 sites* | – Adenopathies |
| – Involvement of at least one internal organ* | – Liver alterations (ALT>100IU) or involvement of other organs |
| – Hospitalization | – HHV-6 reactivation |
SCARJ: all 7 criteria are to be met for establishing the diagnosis; RegiSCAR: 3 of the 4 criteria marked by an asterisk (*) are required to establish the diagnosis.
Symptoms onset occurs between 2 and 8 weeks after introduction of the drug.4,8 The initial manifestations comprise fever (90–100%), morbilliform rash (87–90%), erythroderma with mucosal membrane involvement (22%) and generalized adenopathies (75%). An erythematous maculopapular reaction subsequently develops affecting the skin of the face, trunk and extremities, together with facial edema (25%). The third phase is characterized by multisystem involvement–the most frequently affected organ being the liver (50–60%) in the form of hepatomegalia and serum transaminase elevation, followed by hematological disorders (23–50%) in the form of eosinophilia and atypical lymphocytosis, and interstitial nephritis (11%) that may result in acute renal failure. Less frequent manifestations are (interstitial pneumonitis 9%), carditis (pericarditis or myocarditis)8 and even meningitis or encephalitis. The mortality rate associated to DRESS is 10%, and is mainly conditioned by the age of the patient and liver and kidney involvement.1,2,5
The management of DRESS comprises suspension of the suspect drug and the administration of glucocorticoids at a dose of 1–2mg/kg/day. If the condition persists despite such measures, 1g/day pulses of methylprednisolone can be prescribed during three days. In cases that do not respond to the adopted treatments, there have been reports of a good outcome following the administration of systemic immunoglobulins or plasmapheresis.1,2,5
We present the case of a 21-year-old male without known drug allergies who developed cough, fever and right-side pleural effusion. In addition to the pleural effusion, computed tomography revealed right pleural thickening and the presence of mediastinal adenopathies, with no lung lesions. Thoracocentesis yielded a lymphocytic exudate with adenosine deaminase (ADA) elevation. Ziehl-Neelsen staining and pleural fluid culture in Lowënstein medium proved negative, while interferon-gamma in response to Mycobacterium tuberculosis antigens was positive. The Mantoux intradermoreaction test proved positive. Pleural tuberculosis was diagnosed, and treatment was started with Rimstar® (isoniazid, rifampicin, pyrazinamide and ethambutol).
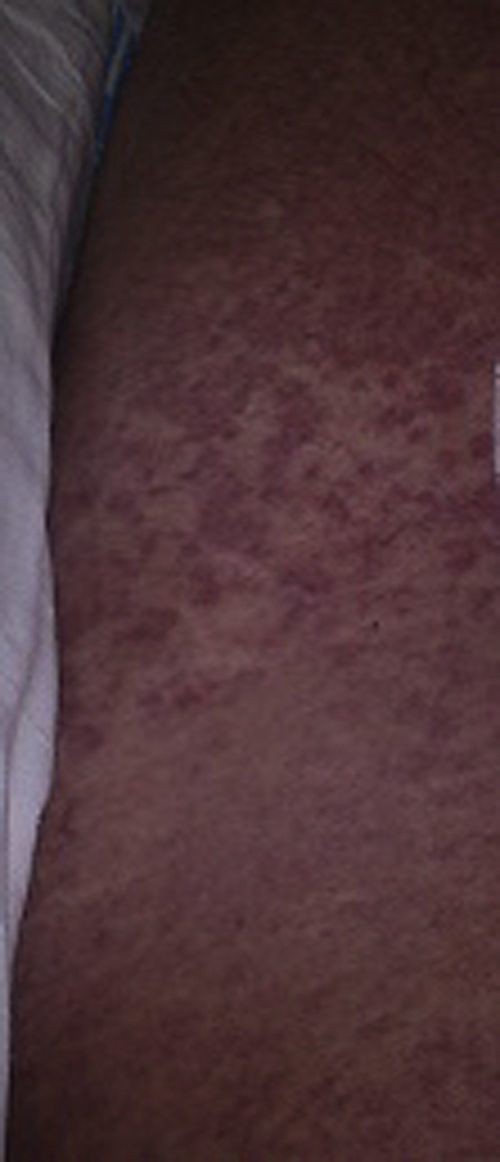
Three weeks later, the patient reported due to the appearance of a confluent maculopapular skin rash affecting 90% of the skin surface, including the soles and palms, but without mucosal membrane involvement (Fig. 1), and accompanied by fever of up to 42°C. The condition progressed to severe acute respiratory failure and distributive shock with lactate concentrations of up to 6mmol/l. Admission to the Intensive Care Unit (ICU) was therefore decided. Intubation and mechanical ventilation proved necessary, as well as volume replacement and vasoactive amine drugs as shock resuscitation measures. A first echocardiographic evaluation revealed preserved ventricular function with mild pericardial effusion. Blood cultures, bronchial aspirate and urine culture were carried out, with negative results. Within 24h the organ dysfunction worsened, with plasma transaminase elevation, coagulopathy, thrombocytopenia, eosinophilia and anuric acute renal failure requiring continuous renal replacement therapy. Abdominal ultrasound revealed important hepatosplenomegalia. The thoracic computed tomography scan showed a generalized bilateral ground-glass pattern. With the suspicion of DRESS, we suspended Rimstar® and started glucocorticoid treatment (1.5mg/kg/day). The skin biopsy revealed marked eosinophilic infiltration, which confirmed the diagnosis. Human herpes virus-6 serological testing was positive. The course proved unfavorable despite the treatment measures, with gradual intensification of the eosinophilia to 7.5×109cells/l and the appearance of myocardial dysfunction with global hypokinesis (left ventricular ejection fraction 45%) and a troponin I concentration of 4ng/ml. Cardiac involvement secondary to eosinophilic infiltration was suspected. In view of the rapid progression of organ dysfunction, with no response to glucocorticoid therapy, and after 5 days of treatment, plasmapheresis was carried out, with poor hemodynamic tolerance. We therefore replaced plasmapheresis with a session of leukapheresis and granulocyte apheresis (the aim being to capture the granulocyte interphase, block the inflammatory response and eliminate the peripheral blood stem cells and avoid eosinophilic proliferation), followed by the administration of hydroxyurea in order to block bone marrow eosinophil proliferation. Following these treatment measures, the eosinophil count decreased rapidly and continuously, with a favorable clinical course allowing withdrawal of all the supportive measures and patient discharge from the ICU within 6 days.
Drug rash with eosinophilia and systemic symptoms (DRESS) is an often underdiagnosed and potentially fatal disorder, with a growing incidence. An early diagnostic suspicion is required in order to eliminate the causal agent as soon as possible and start first line treatment. In the absence of a response to glucocorticoids, second line therapy with immunoglobulins or plasmapheresis is considered. In our case leukapheresis was decided in view of the seriousness of the clinical condition and patient intolerance of plasmapheresis. Apheresis involves the removal of certain cellular components from blood–leukocytes being removed in the case of leukapheresis. This technique is indicated for the management of hyperleukocytosis (hematological malignancies with >100×109leukocytes/l) prior to chemotherapy, with the aim of obtaining CD34 progenitor cells for bone marrow transplantation, among other conditions.9,10 Its use in successfully treating hypereosinophilia has not been reported to date. We therefore decided to describe this potential indication of apheresis, which caused no complications and proved very successful in the context of a patient with extremely serious multiorgan dysfunction.
Financial supportThe authors have received no financial support for carrying out this study.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Martínez de Lagrán I, Marcos P, Batlle M, Alonso E, Plana A, Tomasa T. Leucoaféresis en el tratamiento del síndrome de drug rash with eosinophilia and systemic symptoms. Med Intensiva. 2017;41:191–193.