Limbic encephalitis (LE), described in 1960 by Brierley et al.1 is a rare neurological clinical entity of subacute course that causes neuropsychiatric symptoms.
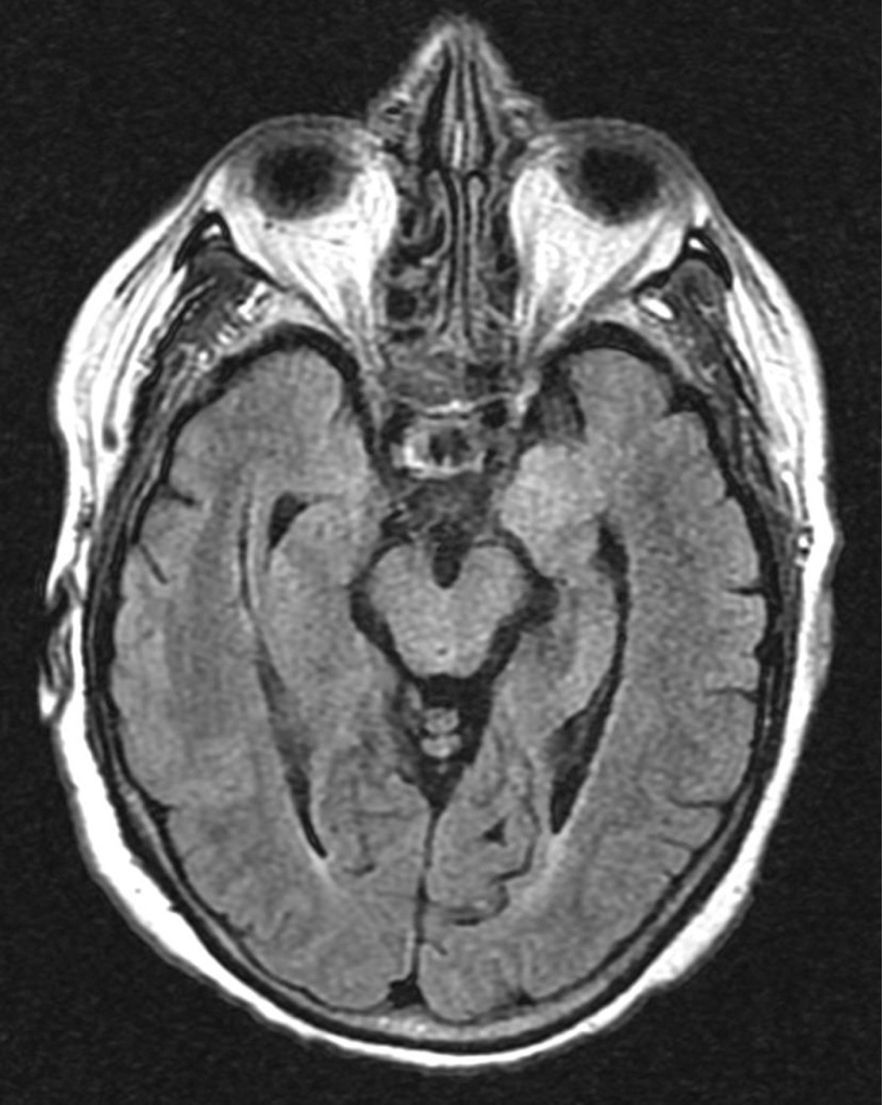
Hereby we present the clinical case of a seventy one year old female with dyslipidemia, hypothyroidism and breast ductal carcinoma with radical exeresis and treated with chemotherapy. She was admitted in the hospital following an event of sepsis due to methicillin-sensitive Staphylococcus aureus (due to reservoir infection). She suffers from progressive disorientation, repetitive talking, and sleepiness with progression towards status epilepticus, which is why she was admitted in the unit of intensive care medicine (ICM). The urgent cranial computed tomography (CT) scan performed did not show any significant findings. At admission her state is post-critical after the administration of intravenous (IV) benzodiazepines and her temperature is 38°C; the rest of the device based-medical examination was normal. The blood sample showed platelet levels of 22×109/l, creatinine levels of 3.5mg/dl, urea levels of 95mg/dl, C-reactive protein (CRP) levels of 15.6mg/dl, the rest being normal. Doctors proceeded with orotracheal intubation and lumbar puncture (LP) followed by secretion of cerebrospinal fluid (CSF) colourless at normal-pressure without cells, glucose levels of 68mg/gl and protein levels of 64mg/dl (plasma proteins 5.2g/dl). Doctors proceeded with sedoanalgesia, fluid therapy, levetiracetam, and broad-spectrum IV antimicrobial treatment including acyclovir. They were isolated in bronchoaspiration and one hemoculture of the extended-spectrum-beta-lactamase-producing Klebsiella pneumonia without condensation in the chest X-ray was collected; both the urine culture and the CSF culture tested negative. CSF with polymerase chain reaction (PCR) for herpes simplex virus (HSV), cytomegalovirus (CMV), varicella zoster virus (VZV) and Epstein–Barr virus (EBV) all tested negative, and the cytology to rule out malignant cells tested negative too. Serologies for human immunodeficiency virus (HIV), hepatitis, HSV, CMV and VZV tested negative for active infections. Elevated CA 125 level (180.3U/ml); remaining tumour markers normal. Negative test results for the anti-DNA antibodies, antinuclear antibodies, c-ANCA and p-ANCA. Thyroid hormones in normal ranges with replacement therapy and negative test results for the antiperoxidase and antithyroglobulin antibodies. One second CT scan with the administration contrast is performed and it rules out structural disease, and also another control LP with secretion of colourless fluid at normal pressure with normal biochemical profile without cells, and negative for malignancy. Given the low level of consciousness, one electroencephalogram (EEG) is performed that is consistent with the effects of sedation and the nuclear magnetic resonance (NMR) of the brain with the following results: in the T2-weighted sequence of the hypocampic region, there is hyperintensity of left predominance, in the diffusion-weighted sequence, there is hyperintensity and a mild decrease of the apparent diffusion coefficient (ADC) in the hypocampic region of the left temporal lobe. After the administration of paramagnetic contrast there is no presence of any uptakes (Figs. 1 and 2A and B).
Given the clinical data and the characteristic neuroimaging obtained, a diagnosis of LE was suggested; onconeuronal antibodies tests in blood and CSF (against NMDA, IA-2, GAD, Hu, Yo, Ri, PNMA2, CV2, antiphysine, titin, recoverin and SOX1) were conducted and they tested negative. Treatment with 1mg/kg/d of IV methylprednisolone was initiated that resulted in a slow but favourable neurological recovery. The patient was discharged from the ICM unit after 35 days, then from the hospital after 20 days, and then administered 1mg/kg/d of oral prednisone for the next three3 months after which she achieved ad integrum neurological recovery.
The clinical manifestations of LE are the subacute2 appearance of several neuropsychiatric alterations including, among others, cognitive impairment, seizures, depression, irritability, hallucinations, and loss of short term memory.3,4 In most cases, the CSF5 shows inflammatory changes with discrete proteinorrachia and/or pleocytosis without pathognomonic findings. The CT scan5 is usually normal, without contrast uptake, while in 70% of the cases, the NMR of the brain5 is asymmetrically hyperintense in both temporal lobes, especially the T2 and the FLAIR-weighted sequences, while remains unenhanced after the administration of contrast. The EEG can show temporal or diffuse slowness, temporal spikes, and temporal or generalized focal seizure activity.
Today we can distinguish three3 types of LE:6 paraneoplastic, viral and idiopathic (non-paraneoplastic autoimmune).
Our patient shows clinical data associated with additional tests that are highly suggestive of LE. In the context of immunosuppression and since the most common cause of LE is viral, we ruled out a diagnosis of viral LE since the CRP in the CSF tested negative for HSV, CMV, VZV, and EBV (sensitivity and specificity over 95%).7 Differential diagnosis was conducted with other causes of encephalopathy with affectation of the temporal lobe such as mesial sclerosis, ischaemic stroke, cerebral neoplasm, Hashimoto encephalopathy and lupus encephalopathy.
Taking these data into consideration, the most probable clinical diagnosis is paraneoplastic LE8 (PLE). This is a rare entity (occurring in <1% of patients with cancer) and in between 60 and 70% of the cases, the neurological clinical manifestations can occur even years before the identification of the neoplasm; the tumours more commonly associated with PLE are small cell lung carcinomas (50%), testicular cancers (20%), and breast cancers (8%).
The diagnosis of this entity requires meeting the four4 following criteria:9 a compatible clinical picture, absence of neuro-oncological complications, <4 years-interval between symptom onset and the diagnosis of the tumour, and at least, one of the following: (a) inflammatory changes in the CSF with a negative cytology, (b) one NMR of the brain with changes in the temporal lobe, (c) the identification of onconeuronal antibodies6,10 in blood or the CSF–present in between 50 and 60% of cases whose specificity is over 90%.
Our patient meets all diagnostic criteria except for testing positive for onconeuronal antibodies.
The LE is a rare and misdiagnosed clinical entity that should be taken into consideration when conducting the differential diagnosis of confusional state of subacute onset. In medical literature there are very few cases of PLE reported, and it is an exceptional diagnosis after the neoplastic process.
Management of PLE is based on symptom control and the initiation of oncospecific and/or immunomodulating therapy. Up to 50% of the patients do not respond to first line immunomodulating therapies (corticoids, IV immunoglobulins or plasma exchange–alone or combined). In the management of refractory cases, the administration of second-line therapies (rituximab and cyclophosphamide – alone or combined)10 enables full recoveries or with minimal deficits in 80% of the cases.
Please cite this article as: Sosa-Torres B, Prieto-Bragado MJ, Domínguez-Quintero LÁ, Fernández-Arroyo P, Blasco-Ciscar E, Cantó-Pérez R. Encefalitis límbica: la gran desconocida. Med Intensiva. 2017;41:319–321.



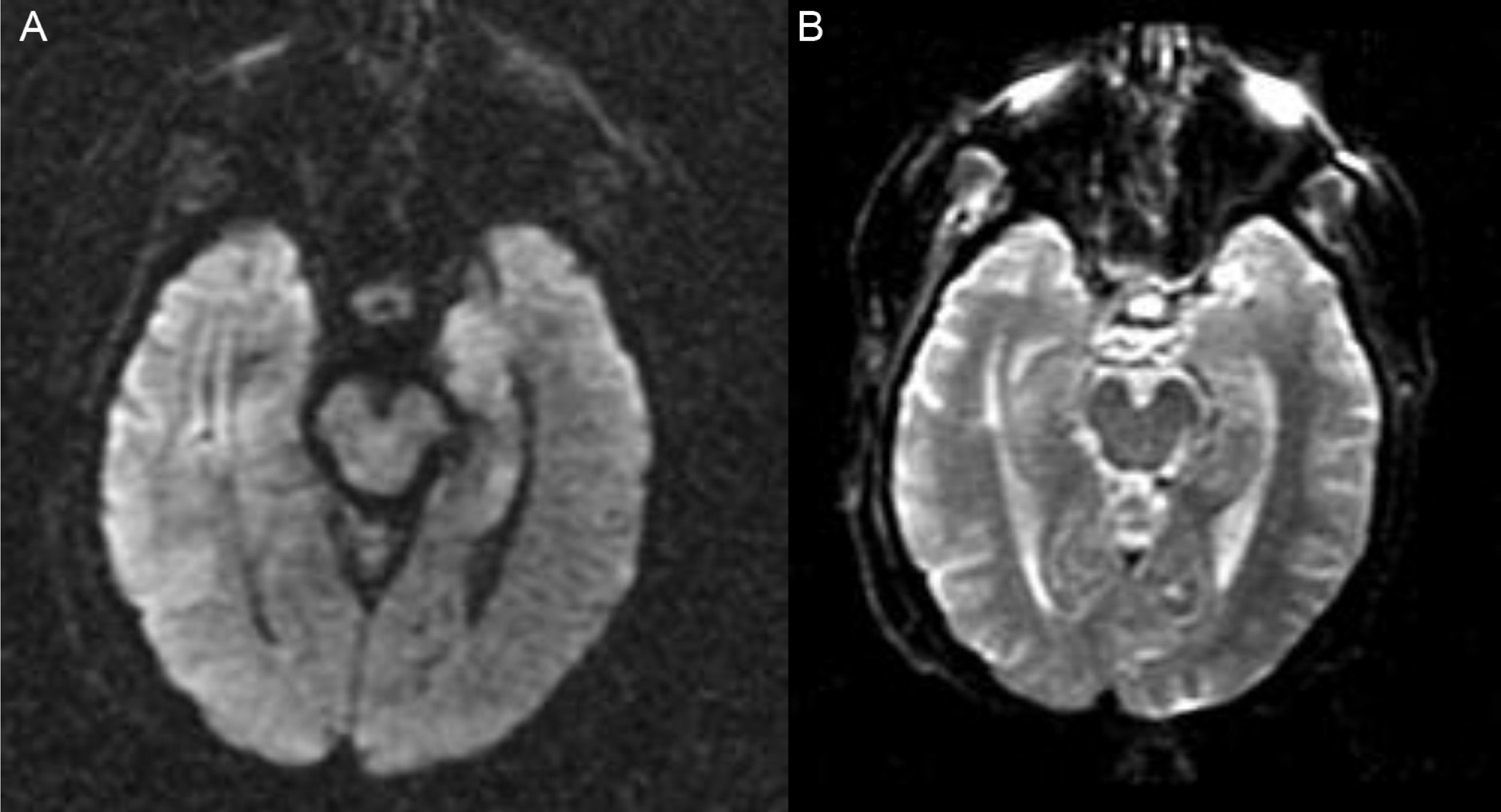
![NMR of the brain ([A]: diffusion study, and [B]: ADC coefficient). The diffusion study shows hyperintensity with discrete decrease of the ADC coefficient in the hypocampic region of the left temporal lobe. NMR of the brain ([A]: diffusion study, and [B]: ADC coefficient). The diffusion study shows hyperintensity with discrete decrease of the ADC coefficient in the hypocampic region of the left temporal lobe.](https://static.elsevier.es/multimedia/21735727/0000004100000005/v1_201706100124/S2173572717300917/v1_201706100124/en/main.assets/thumbnail/gr2.jpeg?xkr=1dZuESKpnCAWr3yCSGZ24A==)

