Takotsubo syndrome is a type of myocardiopathy characterized by transient left ventricular dysfunction associated to electrocardiographic changes similar to those of acute myocardial infarction, though without evidence of coronary disease, and with scantly altered myocardial enzyme levels. The syndrome is also known as transient apical dysfunction, “apical ballooning” or stress-induced myocardiopathy, since it generally affects postmenopausal women following some stressing event. The diagnostic criteria of Takotsubo syndrome are: acute electrocardiographic anomalies such as ST-segment elevation or T-wave inversion; the absence of obstructive coronary disease; akinesia or transient dyskinesia of the left ventricle; and the absence of traumatic brain injury, intracranial hemorrhage, pheochromocytoma, myocarditis or hypertrophic myocardiopathy.1 The incidence and prevalence of the syndrome are not known, and few cases have been reported in patients subjected to cardiovascular surgery. We present the case of a woman operated upon due to an atrial myxoma, who in the postoperative period developed cardiogenic shock secondary to Takotsubo syndrome.
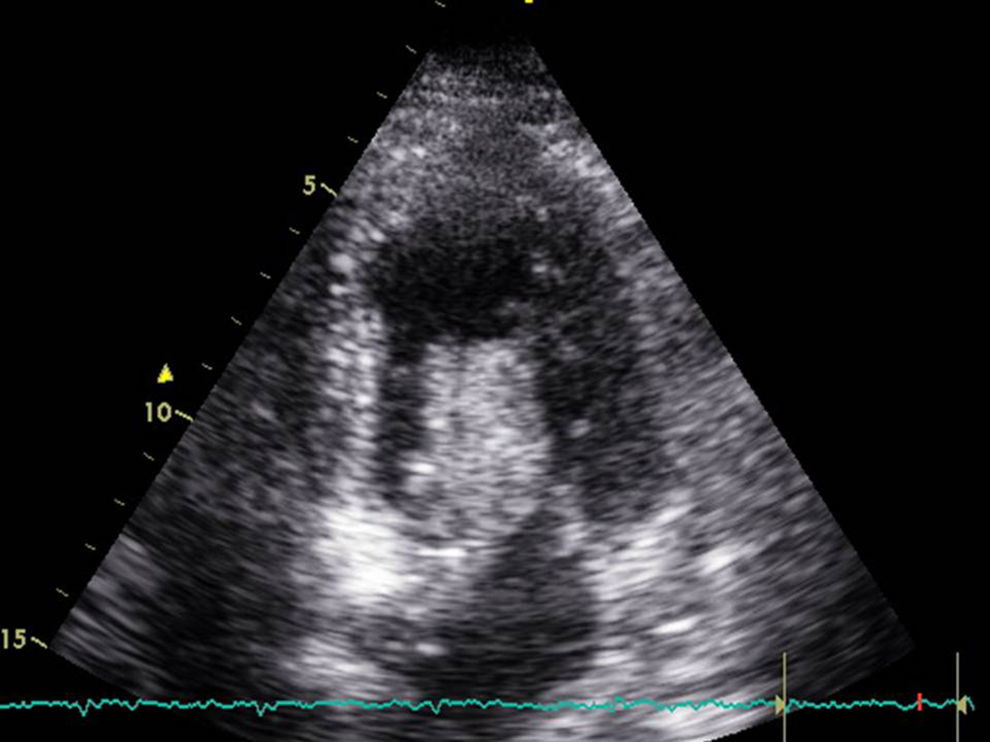
A 68-year-old woman with arterial hypertension and non-insulin dependent diabetes reported to the emergency service due to clinically manifest heart failure for the last two weeks. Upon admission she presented resting dyspnea and palpitations. Rapid atrial fibrillation was detected, requiring pharmacological cardioversion. The echocardiographic study (Fig. 1) revealed a large left atrial mass (1.8×7cm in size) protruding toward the left ventricle and causing secondary mitral valve stenosis and atrial dilatation, with a preserved left ventricular ejection fraction (LVEF). Preoperative coronary angiography showed no lesions.
Surgery was performed four days later, with trans-septal removal of the myxoma. There were no relevant incidents other than a tendency toward hypoxemia. The clamping and extracorporeal circulation times were 52 and 83min, respectively. Intraoperative echocardiography confirmed the absence of any remnant myxoma tissue, with a normal LVEF. The patient remained stable and subjected to mechanical ventilation during her first hours of stay in the Intensive Care Unit (ICU) due to the aforementioned hypoxemia, with FiO2=0.6 and PEEP=10–12cmH2O. Eight hours after surgery she suffered a sudden hypertensive crisis (systolic blood pressure >220mmHg) followed by hypotension and cardiac arrest with pulseless electrical activity (electromechanical dissociation). Resuscitation maneuvering restored the pulse within a few minutes. The patient was in cardiogenic shock refractory to high-dose vasoactive medication (adrenalin, dobutamine, noradrenalin); as a result, venous-arterial extracorporeal membrane oxygenation (V-A ECMO) was therefore finally decided.
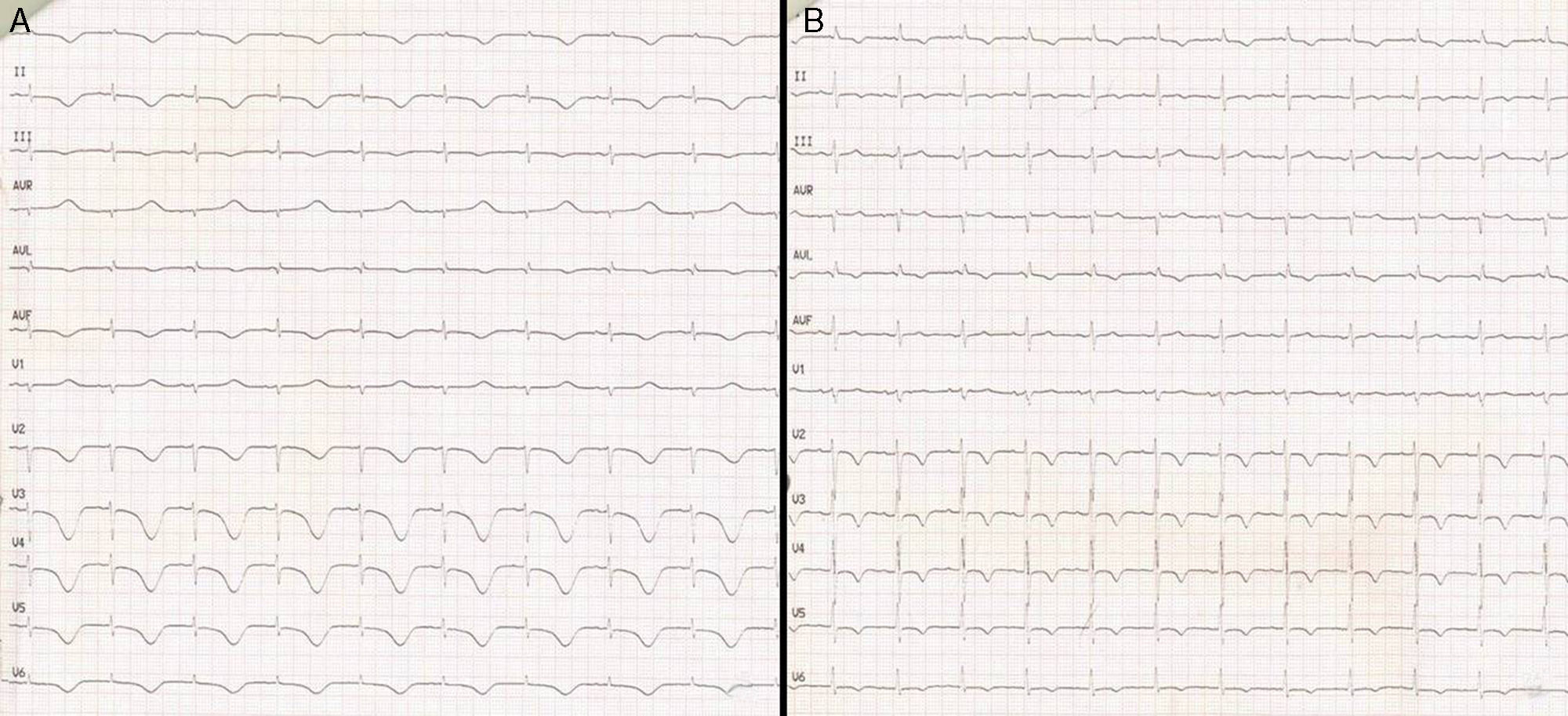
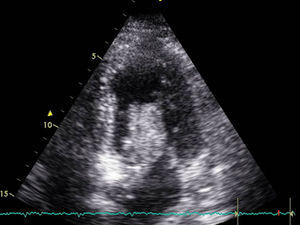
Transthoracic echocardiography revealed severe left ventricular dysfunction (LVEF<20%), hyperdynamic basal segments and akinesia of the middle and apical segments–these findings being typical of Takotsubo syndrome. There were no significant cardiac enzyme elevations over the following days (taking surgery into account), with a maximum ultra-sensitive troponin I concentration of 4000pg/ml (normal 2–15.6pg/ml). In contrast, the electrocardiographic tracing showed significant changes with respect to the previous recordings, with a long QTc (680ms) and the appearance of inverted T-waves on precordial leads during several days (Fig. 2). The electrolyte profile and rest of the laboratory test parameters were normal.
Venous-arterial ECMO was maintained for four days, allowing the withdrawal of vasoactive drug support, and followed by a favorable clinical course. The echocardiographic studies revealed gradual improvement of ventricular function. The patient was extubated a few days later and moved to the hospital ward, where she remained until discharge in good functional condition. The last echocardiographic exploration showed slight anterior septal and inferior hypokinesia, with globally preserved systolic function.
In this case the diagnosis of Takotsubo syndrome was established from the echocardiographic findings, the absence of previous coronary lesions, scant alteration of the cardiac enzyme levels despite the ventricular dysfunction and surgery, and the precordial electrocardiographic tracings (inverted T-waves and long QT syndrome, with normal electrolyte levels). There has been an increase in the number of diagnoses of this syndrome in recent years,2 also in cardiovascular surgery patients,3–5 though Takotsubo syndrome has not been previously described in the postoperative period of atrial myxoma resection.
The etiology and physiopathology of Takotsubo syndrome have not been fully established, though a number of mechanisms have been proposed, such as coronary vasospasm, coronary microvascular functional anomalies, and particularly catecholamine-mediated cardiotoxicity. Probably because of this, the main risk factor is considered to be stress (both physical and mental), which is present in different diagnostic tests and surgical procedures–especially those of an emergent or urgent nature.6 Cardiac surgery and extracorporeal circulation generate important stress, and may intrinsically account for the appearance of Takotsubo syndrome, particularly if the epidemiological profile of the patient and the echocardiographic and electrocardiographic features are consistent with the diagnostic criteria, as in our case. The differential diagnosis includes coronary embolism or poor myocardial protection. The typical echocardiographic findings, with scant enzyme alterations, brief surgery without incidents, and previous hemodynamic stability with a normal first electrocardiographic tracing allow us to reasonably discard both of the aforementioned disorders. In recent years, evidence has been gained suggesting that magnetic resonance imaging may be useful in establishing a differential diagnosis with other disorders such as myocarditis or coronary embolism,7 though this technique could not be used in our case.
Of note is the appearance of a long QT interval on the electrocardiographic tracing. The association between Takotsubo myocardiopathy and transient prolongation of the QT interval has been documented,8,9 being more frequent in patients with some form of heart disease, previous long QT syndrome, or genetic alterations. In Takotsubo syndrome, electrocardiographic normalization occurs approximately two months after the episode. Although malignant ventricular arrhythmias, sudden death or cardiac block are infrequent, there have been reports of such situations. Increased QT prolongation (QTc>500ms) has been identified as a risk factor for complications of this kind; close monitoring of the cardiac rhythm is thus required, with the prevention or treatment of arrhythmias and the implantation of a pacemaker if necessary.
In conclusion, Takotsubo syndrome is an infrequent cause of cardiogenic shock in the postoperative period of heart surgery, though it must be considered in patients with apparently uncomplicated surgery, normal coronary vessels and compatible echocardiographic findings, since the prognosis is generally good when adequate supportive treatment is provided.
Financial supportThis study has received no financial support.
Please cite this article as: García-Delgado M, García-Huertas D, Navarrete-Sánchez I, Olivencia-Peña L, Garrido JM. Soporte con oxigenación de membrana extracorpórea en un síndrome de Takotsubo y QT largo tras cirugía cardiaca. Med Intensiva. 2017;41:441–443.