Arterial hypotension (usually defined as systolic blood pressure of <90mmHg, or mean arterial pressure of <65mmHg, or a decrease of >40mmHg),1 often represents the first clinical sign of an acute decompensated cardiovascular system, and it is the most frequent indication for fluid administration in critically-ill patients.2 However, the arterial blood pressure (ABP) response to intravenous volume expansion (VE) is somehow unpredictable: some patients exhibit an increase and others not.3 Therefore, fluid administration, if aimed to restore and maintain ABP, could lead to an unnecessary fluid overload, delayed vasoactive therapy or even an increased mortality.4
In the following, we describe the physiological basis of the flow-pressure relationship to understand why, in some hypotensive patients, VE seems to be effective in terms of arterial pressure increase, whereas in others patients ABP remains unchanged, even if cardiac output (CO) increases.
What is arterial hypotension?Arterial pressure results from the interaction between the blood flow generated by the ventricle and the arterial system.5 ABP therefore is a regulated variable modulated within narrow limits: as long as the homeostatic autoregulation is preserved, there could be different combinations of arterial system and blood flow conditions for the same level of arterial pressure.6 Consequently, arterial hypotension should be considered as the consequence of loss of balance between ventricular output and the modulation of the arterial system.7 Moreover, systemic hypotension is a frequent preamble to organ hypoperfusion, since below a critical arterial pressure level, organ perfusion becomes compromised and regional blood flow will depend directly on systemic pressure.6 For this reason, a sustained low blood pressure, or even a sole episode of hypotension, has been associated with a poor outcome.8–11 Similarly, restoring and maintain a minimum perfusion pressure is one the main goals of current hemodynamic resuscitation protocols. However, it is important to remark that, as ABP is a regulated variable, a normal ABP value does not necessarily discard the absence of hypoperfusion.
Arterial pressure and cardiac output relationship during fluid administrationSince arterial pressure depends on blood flow and the arterial system, changes in ABP may therefore be related to variations in one or both factors. Consequently, even if blood flow varies, as may occur after VE, the ABP will be ultimately determined by the arterial system regulation.
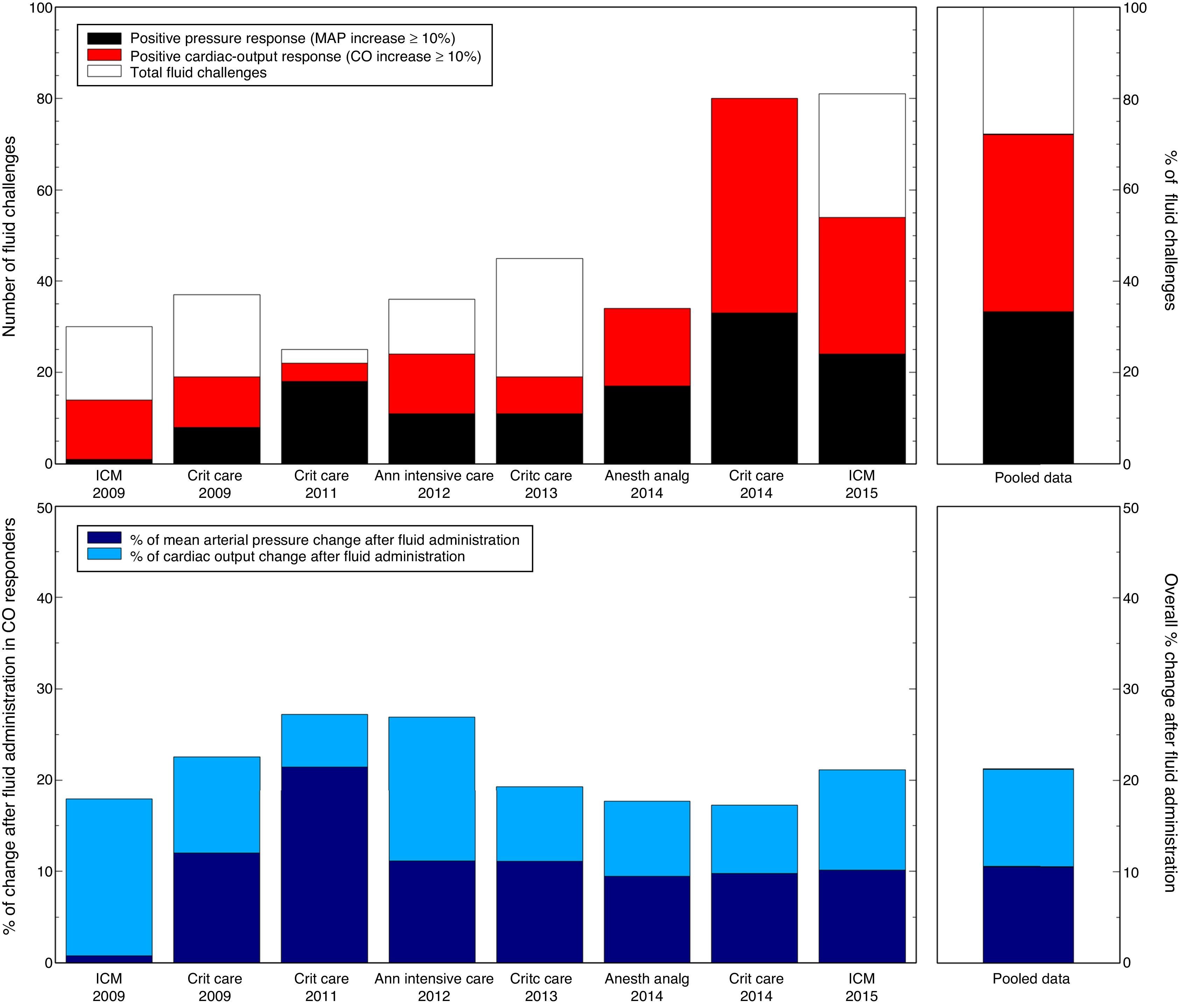
From this physiological phenomenon, some clinical deductions can be drawn. First, a change in the arterial system determines the lack of ABP change after VE, even if CO increases.3 This also explains why CO and ABP changes induced by VE are usually poorly related.3 Secondly, using ABP as a surrogate for CO changes could lead to wrong assumptions, as the arterial system modulation could potentially modify the pressure-flow relationship and alter the ABP response. Lastly, whereas a positive CO response to VE is typically found in half of the critically-ill patients, ABP increase with fluids may be observed even in a lower proportion of cases, because of the effects of individual arterial system regulation (Fig. 1). Moreover, this regulation could be also responsible of the short-lived benefits on ABP after VE.12
Rate of positive arterial pressure response after fluid administration.
Proportion of pressure-responders (mean arterial pressure, MAP increase ≥10%) and preload-responders (cardiac output, CO increase ≥10%). A ventriculo-arterial coupling ratio 1:1 was assumed. So, for a CO increase of 10%, a MAP increase of 10% should be expected. Please, note that preload-responder and pressure-responder definitions could differ from that defined in the original publication.
References used for this illustration:
Monge Garcia MI, Gil Cano A, Diaz Monrove JC. Arterial pressure changes during the Valsalva maneuver to predict fluid responsiveness in spontaneously breathing patients. Intensive Care Med. 2009;35(1):77–84.
Monge Garcia MI, Gil Cano A, Diaz Monrove JC. Brachial artery peak velocity variation to predict fluid responsiveness in mechanically ventilated patients. Crit Care. 2009;13(5):R142.
Monge Garcia MI, Gil Cano A, Gracia Romero M. Dynamic arterial elastance to predict arterial pressure response to volume loading in preload-dependent patients. Crit Care. 2011;15(1):R15.
Monge Garcia MI, Gil Cano A, Gracia Romero M, Monterroso Pintado R, Perez Madueno V, Diaz Monrove JC. Non-invasive assessment of fluid responsiveness by changes in partial end-tidal CO2 pressure during a passive leg-raising maneuver. Annals of Intensive Care. 2012;2:9.
Monge Garcia MI, Romero MG, Cano AG, Rhodes A, Grounds RM, Cecconi M. Impact of arterial load on the agreement between pulse pressure analysis and esophageal Doppler. Crit Care. 2013;17(3):R113.
Cecconi M, Monge Garcia MI, Gracia Romero M, Mellinghoff J, Caliandro F, Grounds RM, et al. The Use of Pulse Pressure Variation and Stroke Volume Variation in Spontaneously Breathing Patients to Assess Dynamic Arterial Elastance and to Predict Arterial Pressure Response to Fluid Administration. Anesth Analg. 2014;120(1):76–84.
Monge Garcia M, Gracia Romero M, Gil Cano A, Aya HD, Rhodes A, Grounds R, et al. Dynamic arterial elastance as a predictor of arterial pressure response to fluid administration: a validation study. Crit Care. 2014;18(6):626.
Monge Garcia MI, Guijo Gonzalez P, Gracia Romero M, Gil Cano A, Oscier C, Rhodes A, et al. Effects of fluid administration on arterial load in septic shock patients. Intensive Care Med. 2015;41(7):1247–55.
Although ABP is frequently used as surrogate for changes in cardiac output for assessing the response to fluids,2 the physician should be aware that the lack of arterial pressure response after VE is not always related to a preload-independency condition. A good exercise to understand the physiological mechanisms by which fluid administration fails to improve arterial pressure is to consider the effects of VE on venous return, cardiac output, and arterial system.
To effectively increase ABP, intravascular expansion first needs to increase venous return, which is a function of the pressure gradient to the heart (mean filling pressure minus central venous pressure) and the venous resistance. If venous return and cardiac preload are positively increased, then the heart must operate on the ascending limb of the ventricular function curve (preload-dependency zone) to increase CO. Only if both ventricle are preload-dependent, a change in cardiac preload will modify CO. And finally, if CO increases, arterial system will eventually define the arterial pressure response.
Although the exact mechanisms are still not well elucidated, fluid expansion may deactivate the baroreflex-mediated increase of sympathetic activity induced by hypovolemia. Therefore, changes in arterial system may reflect a physiological adaptive mechanism of reduced sympathetic tone in response to a CO increase. In addition, increases in CO could lead to a modulation of the arterial system through a flow-mediated vascular relaxation induced by a shear stress stimulus and an augmented nitric oxide production by the endothelial glycocalyx. Finally, recruitment of previously closed arterial vessels could increase the effective diameter of the arterial system and hence reduce total arterial resistance.3 However, the precise mechanisms by which arterial system ultimately defines a specific ABP response after VE are still unknown.
Therefore, even if an increase in ABP after VE is likely an indicator of a positive CO increase, the absence of an ABP response to fluids could be related different mechanisms: a lack of a venous return increase, a non-preload dependence condition of one or both ventricles, or the effects of modulation of the arterial system.
Treating arterial hypotension: fluids, vasopressors, or both?Since hypotension may be the final pathway of diverse pathological conditions, the adequate treatment for a hypotensive patient should necessary be aimed to correct the underlying etiology. However, regardless the mechanism, eventually the management of hypotension is usually reduced to decide whether to increase CO or to restore ABP with vasopressors. However, how much fluids or when to start vasopressors is usually performed arbitrarily or without any solid physiological rationale.
In the setting of arterial hypotension, one essentially needs to answer the following questions: will CO improve with fluids? If so, will increases in CO also increase ABP? If the answer to both question is yes, fluids could be used as the first choice. Otherwise, vasopressors should be initiated, although fluids may be also added if preload-responsiveness is preserved. Although dynamic indices of preload-responsiveness, such as stroke volume variation (SVV) or pulse pressure variation (PPV), are widely used for predicting the CO response to fluids, they cannot answer the question about the ABP. Therefore, knowing if a patient is preload-dependent or not, only resolve half of problem about hypotension.
Under the same functional principle, the dynamic arterial elastance (Eadyn), or the ratio of PPV to SVV, has been suggested as a functional assessment of arterial load.13 Taking advantage of well-known heart-lung interactions, Eadyn dynamically depicts the relationship of blood flow and the arterial system during a respiratory cycle, defining how the arterial pressure changes with cyclic variations in the stroke volume induced by intermittent positive-pressure ventilation. Consequently, Eadyn has been used for the prediction of ABP response to fluid challenge in preload-dependent patients. So, the higher the Eadyn, the higher the probability that an increase in CO will also increase ABP. On the contrary, if Eadyn is low, ABP will not increase, even if CO does. Therefore, Eadyn may be helpful in defining if hypotensive patients will benefit from fluids, or conversely, they will need vasopressors to increase arterial pressure.
ConclusionsArterial hypotension represents the imbalance between the flow generated by the heart and the arterial system regulation. Although, arterial hypotension may result from diverse pathophysiological mechanisms (hypovolemia, cardiac failure, vasoplegia, etc.), eventually all of them may converge on organ hypoperfusion.
When interpreting the arterial pressure response to VE, it should be considered that the effects on ABP involve the influence of venous system, cardiac function, and the arterial system. As ABP depends not only on flow but also on arterial system, therefore, even if CO increases in response to VE, the regulation of arterial system will ultimately define the final pressure response.
The proportion of patients that increase arterial pressure after VE is usually small, even if they are preload-responders, and in some cases, the increase in arterial pressure may be of little clinical relevance. We need to find new tools for predicting ABP response to fluids. In this regard, a functional approach, as provided by dynamic indexes of preload-responsiveness and Eadyn, may help to determine the most adequate therapy for hypotensive patients.
Source of fundingThis manuscript has no financial support.
Conflict of interestThe authors declare no conflict of interest regarding this paper.