The goals of this project were to compare both the anatomic and physiologic severity scores in trauma patients admitted to intensive care unit (ICU), and to elaborate mixed statistical models to improve the precision of the scores.
MethodsA prospective study of cohorts. The combined medical/surgical ICU in a secondary university hospital. Seven hundred and eighty trauma patients admitted to ICU older than 16 years of age. Anatomic models (ISS and NISS) were compared and combined with physiological models (T-RTS, APACHE II [APII], and MPM II). The probability of death was calculated following the TRISS method. The discrimination was assessed using ROC curves (ABC [CI 95%]), and the calibration using the Hosmer–Lemeshow's H test. The mixed models were elaborated with the tree classification method type Chi Square Automatic Interaction Detection.
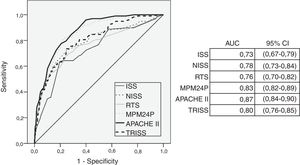
ResultsA 14% global mortality was recorded. The physiological models presented the best discrimination values (APII of 0.87 [0.84–0.90]). All models were affected by bad calibration (p<0.01). The best mixed model resulted from the combination of APII and ISS (0.88 [0.83–0.90]). This model was able to differentiate between a 7.5% mortality for elderly patients with pathological antecedents and a 25% mortality in patients presenting traumatic brain injury, from a pool of patients with APII values ranging from 10 to 17 and an ISS threshold of 22.
ConclusionsThe physiological models perform better than the anatomical models in traumatic patients admitted to the ICU. Patients with low scores in the physiological models require an anatomic analysis of the injuries to determine their severity.
Comparar los scores de gravedad anatómicos y fisiológicos en los pacientes traumáticos ingresados en unidad de cuidados intensivos (UCI) y crear modelos combinados que mejoren su precisión.
MétodoEstudio prospectivo de cohorte observacional. UCI polivalente de un hospital universitario de 2.° nivel. Se incluyeron los pacientes traumáticos mayores de 16 años ingresados en UCI (n=780). Se comparan los modelos anatómicos (ISS, NISS) con los modelos fisiológicos (T-RTS, APACHE II [APII]), MPM II). La probabilidad de muerte se calculó según metodología TRISS. La discriminación se valoró mediante curvas ROC (ABC [IC 95%]) y la calibración con el estadístico H de Hosmer–Lemeshow. Los modelos combinados se realizaron con metodología de árboles de clasificación tipo Chi Square Automatic Interaction Detection.
ResultadosMortalidad global del 14%. Los modelos fisiológicos son los que presentan mejores valores de discriminación (APII con 0,87 [0,84–0,90]). Todos los modelos tienen una mala calibración (p<0,01). El mejor modelo combinado es el que utiliza APII junto a ISS (0,88 [0,83–0,90]) y encuentra un grupo de pacientes (valores de APII entre 10–17) que necesita la puntuación ISS (punto de corte de 22) para diferenciar entre mortalidad del 7,5% asociada a pacientes mayores con antecedentes patológicos y del 25,0% en pacientes con mayor presencia de TCE.
ConclusionesLos modelos fisiológicos presentan ventajas sobre los anatómicos en los pacientes traumáticos ingresados en UCI. Los pacientes con puntuaciones bajas en los modelos fisiológicos requieren del análisis anatómico de las lesiones para determinar su gravedad.
Severity scores have been developed to describe the severity of lesions or correlate the prognosis by establishing mortality risk through numerical probability values.1,2
The severity of a given trauma depends on the patient and his physiological reserve (age, comorbidities), the anatomical lesions sustained, and the physiological repercussions (hemodynamic, respiratory, and neurological). Also, we should remember here that the final outcome will depend on the quality of care provided to our patients during the entire healthcare process.3,4
In the prehospital stage, the goal of any given score is to triage the patients with the most serious injuries easy and quickly, so they can be transferred to a hospital depending on their actual healthcare needs. The Triage-revised Trauma Score (RTS)5 evaluates the physiological repercussions at the respiratory level (respiratory rate); hemodynamic level (systolic blood pressure); and neurological level according to the Glasgow Coma Scale (GCS). Recently, the GAP score that includes the GCS, the systolic blood pressure and age has been published.6 Emergency rooms still use the Trauma and Injury Severity Score (TRISS) as a model that provides a score on mortality risk.7 The TRISS is a mixed model that combines the physiological repercussion (RTS) based on the anatomical injuries sustained (Injury Severity Score [ISS]) and the mechanism of lesion formation (blunt trauma or penetrating).7
During the last few years different severity scores have come out such as the Probability of Survival [PS] score,8 the Trauma Risk Adjustment Model [TRAM]4 or the Revised Injury Severity Classification [RISC I-II9]). They have improved the discrimination and calibration capacity when including other variables such as comorbidities; mechanical ventilation; hemoglobin; base excess; etc. available at the ER setting.
In the intensive care unit (ICU) setting severity scores such as the Acute Physiology and Chronic Health Evaluation (APACHE II), the Simplified Acute Physiologic Score (SAPS II) or Mortality Probability Models (MPM II) are used. These scores assess the former state of the patent (age), any associated comorbidities and the physiological repercussions. Nonetheless, they have not been designed specifically for trauma populations since they do not make anatomical assessments of the lesions sustained or the mechanisms of lesion formation.
To date there is no consensus on what severity score (anatomical, physiological, or mixed) should be used in trauma patients admitted to the ICU.3
The goal of this study is to compare anatomical (ISS,11 New Injury Severity Score [NISS]12), physiological (RTS,5 MPM II-24,13 APACHE II14) and mixed scoring systems (TRISS7) in severe trauma patients the first 24h after ICU admission and find models capable of combining these scores to better their accuracy.
Material and methodProspective cohort study conducted from January 2005 through December 2012 at a 16 bed-polyvalent ICU of the Hospital Universitario Arnau de Vilanova de Lleida, Spain (with 450 beds) with an area of reference of 450,000 inhabitants. Our center is one II 2b-level center because we have one neurosurgery unit and another intensive care medicine unit available 24h a day.
Inclusion criterion: all patients over 16, due to trauma reasons and hospitalized at the ICU for more than 24h.
Exclusion criterion: incomplete data – transfer to another center or death 30 days after admission.
The hospital ethics committee was informed that the study was going to be conducted. It was deemed unnecessary to obtain the patients’ informed consent since the data collected was indispensable for their diagnosis and clinical follow-up. The patients’ anonymity was guaranteed at all time.
Both the epidemiological variables of age, sex, date of admission to the ICU, etiology (crash, work or other type of accident) and the mechanism of lesion formation were included.
The concept of prior organ failure was used following the definition of the APACHE II model.14 Comorbidity was categorized based on the classification established by the American Society of Anesthesiologists Physical Status (ASA-PS): normal (ASA-PS-1), moderate (ASA-PS-2) and severe (ASA-PS-3, -4, -5).15
Severity scoresThe variables of respiratory rate, blood pressure and neurological level (GCS) in the prehospital stage were gathered to estimate the RTS.5
Trauma lesions were studied according to the Abbreviated Injury Scale (AIS-2005) updated 200816 to estimate the anatomical injury severity index (ISS, NISS).11,12 Survival probability was estimated prospectively according to the TRISS methodology.7
The MPM II13 and APACHE II14 severity scores were estimated with data from the first 24h of ICU admission.
The ICU stay and mortality rate at 30 days were monitored both in the ICU and during the patients’ stay at the hospital room.
Statistical analysisDiscreet variables were expressed as percentages, and continuous variables as mean±standard deviation. For inter-group comparison purposes (survivors and non-survivors) the Mann–Whitney statistical test was used for the continuous variables (without assuming the parametric characteristics of the variables) and the chi-square test for the categorical ones. To correlate the different scores analyzed, Spearman's rank correlation coefficient was estimated.
The accuracy of the probabilistic models of mortality at 30 days used and created was assessed by measuring their properties of discrimination and calibration; discriminating through ROC curves and estimating the area under the curve with their 95% confidence interval. Calibration assessed the match between the observed mortality and the expected mortality based on these probabilistic models and also calculating the Hosmer–Lemeshow H statistic (assuming good calibration with p>0.05).17,18
Combined models with the different scores used were created using the classification tree method (AnswerTree module of the SPSS statistics package [version 20.0]) with the possibility of using the CHAID (Chi Square Automatic Interaction Detection) classification tree. Model parameters. Partition rules: Use the chi-square test (with Bonferroni correction) to determine the statistical significance of the recursive partitions it generates. Internal validation system (cross validation) with 10 partitions. Tree growth stopping criterion with reduced total error and minimal number of cases in the terminal nodes (25 cases). Splitting nodes and merging categories of significant level with p<0.05.19
The statistical analysis was conducted using the SPSS statistics package (v20.0). p<0.05 were considered statistically significant.
ResultsDuring the study period, 786 patients met the inclusion criteria – 6 were excluded since they had been transferred to a different center so 780 patients remained in the study group.
Table 1 shows the demographic characteristics based on survival. In general, the patients are predominantly young males with few prior conditions admitted to the hospital emergency room. Patients who die have more prior conditions (arterial hypertension, heart disease, organ failure), or comorbidities (ASA), and their ICU and hospital stays are shorter.
Mortality-based demographic characteristics (n=780).
| All (n=780) | Alive (n=682) | Dead (n=98) | pc | |
|---|---|---|---|---|
| Age (years)a | 46.4±19 | 45.5±18 | 52.2±23 | 0.013 |
| Sex (male) (%) | 77.7 | 77.1 | 81.6 | 0.316 |
| Clinical history (%) | ||||
| Diabetes | 6.9 | 6.9 | 6.8 | 0.927 |
| AHT | 14.7 | 13.5 | 23.5 | 0.012 |
| COPD | 4.1 | 4 | 4.1 | 0.991 |
| Heart disease | 2.9 | 2.2 | 8.2 | 0.001 |
| OF | 1.7 | 1.2 | 4.1 | 0.046 |
| ASA-PS (%) | <0.001 | |||
| Normal | 75.6 | 77.9 | 60.2 | |
| Moderate | 21.5 | 19.9 | 32.7 | |
| Severe | 2.8 | 2.2 | 7.1 | |
| Etiology (%) | 0.149 | |||
| Crash | 60 | 60.6 | 56.1 | |
| Work accident | 14.9 | 15.4 | 11.2 | |
| Other | 25.1 | 24 | 32.7 | |
| Origin (%) | 0.013 | |||
| ER | 75.6 | 77.3 | 64.3 | |
| OR | 17.2 | 15.7 | 26.6 | |
| HR | 3.1 | 2.8 | 5.1 | |
| District hospital | 4.1 | 4.3 | 3.1 | |
| Stay (days)b | ||||
| ICU | 8 (4–18) | 8 (5–19) | 5 (2–11) | <0.001 |
| Hospital | 19 (11–34) | 21 (12–36) | 6 (3–13) | <0.001 |
ASA-PS: American Society of Anesthesiologists Physical Status; COPD: chronic obstructive pulmonary disease; AHT: arterial hypertension; OF: organ failure; ICU: intensive care unit; ER: emergency room; OR: operating room; HR: hospital room.
Table 2 shows the scores obtained in the different score systems studied. All values were significantly higher in patients who died except for the RTS (that showed lower values). Due to the way it has been designed, we have to make an inverse interpretation (the RTS shows lower values in patients who are most critically ill).
Mortality-based scores obtained in risk adjustment models (n=780).
| All (n=780) | Alive (n=682) | Dead (n=98) | pa | |
|---|---|---|---|---|
| Anatomical | ||||
| ISS | 18±9 17 (12–25) | 17±8 16 (10–22) | 25±11 25 (17–29) | <0.001 |
| NISS | 25±11 24 (17–29) | 23±10 22 (17–29) | 36±13 34 (27–43) | <0.001 |
| Physiological | ||||
| RTS | 7±1 7 (6–8) | 7±1 7 (6–8) | 5±2 5 (4–7) | <0.001 |
| MPM II-24 | 14±20 7 (4–18) | 11±10 6 (3–13) | 36±20 32 (17–53) | <0.001 |
| APACHE-II | 12±7 11 (6–16) | 10±6 10 (6–14) | 21±6 20 (16–25) | <0.001 |
| Mixed system | ||||
| TRISS | 12±19 5 (1–13) | 9±15 4 (1–9) | 34±30 22 (7–53) | <0.001 |
APACHE II: Acute Physiology and Chronic Health Evaluation; ISS: Injury Severity Score; MPM II: Mortality Probability Models; NISS: New Injury Severity Score; RTS: Revised Trauma Score; TRISS: Trauma and Injury Severity Score.
Values expressed as mean±standard deviation and mean (interquartile range).
There is a good correlation among the different statistical models used (see Table 3). The negative value of some correlation values is due to the aforementioned characteristics of the RTS.
Correlation among the different values obtained from mortality risk scoring systems (n=780).
| ISS | NISS | RTS | MPM II-24 | APACHE II | |
|---|---|---|---|---|---|
| NISS | 0.783 | – | |||
| RTS | −0.429 | −0.430 | – | ||
| MPM II-24 | 0.353 | 0.357 | −0.584 | – | |
| APACHE-II | 0.374 | 0.386 | −0.662 | 0.787 | – |
| TRISS | 0.636 | 0.571 | −0.726 | 0.698 | 0.668 |
APACHE II: Acute Physiology and Chronic Health Evaluation; ISS: Injury Severity Score; MPM II: Mortality Probability Models; NISS: New Injury Severity Score; RTS: Revised Trauma Score; TRISS: Trauma and Injury Severity Score.
Values expressed as Spearman's rank correlation coefficient. All with p<0.001.
Fig. 1 shows the discriminative capacity of the score systems based on their area under the ROC curve. The APACHE II model obtains the highest of them all (0.88 [0.83–0.90]).
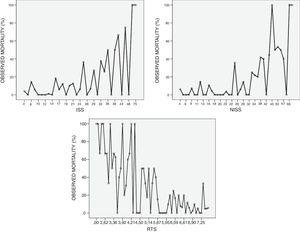
Fig. 2 shows the behavioral pattern of ISS and NISS anatomical scoring systems as well as the physiological Triage-revised Trauma Score (RTS).5Fig. 2 shows that mortality is not an increasing function on the ISS at ICU admission. It also shows mortality peaks in the squared values and a poor correlation in low values. The same thing happens with the NISS scoring system. The RTS has a linear behavioral pattern.
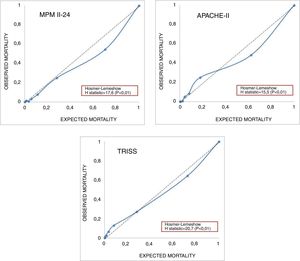
Fig. 3 shows the calibration curves of the physiological models APACHE II and MPM II-24 and TRISS. The Hosmer–Lemeshow H statistic values are included here and they show poor calibration. The APACHE II model shows that with lower mortality rates, higher observed mortality rates, and vice versa when the probabilistic models of mortality are high.
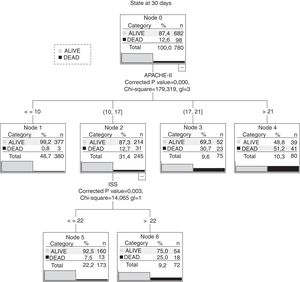
The classification tree method with the best values of area under the ROC curve (0.88 [0.84–0.90]) is the one that combines APACHE II and ISS scoring systems (Fig. 4). It picks as the first variable the APACHE II score and then establishes 4 increasing consistent subdivisions of mortality rate. It is in the group of values scoring between 10 and 17 in the APACHE II score (with 245 patients) where the ISS scoring system is capable of making a distinction between 2 subgroups with a cut-off point of 22. Fig. 4 shows the terminal nodes 5 and 6.
The analysis of these 2 subgroups (see Table 4) confirmed that even though there are no differences in the average APACHE II score, the mortality rate is very different (7.5% vs. 25%; p<0.001). These 2 subgroups are different in some variables. The subgroup that scores ≤22 on the ISS shows older patients more prone to having a prior clinical history of organ failure and comorbidities. Patients scoring >22 on the ISS show lower GCS values. We saw that the presence of cranioencephalic trauma was more significant in this group of patients (77.8 vs. 48; p=0.046).
Differential characteristics of the group of patients who scored 11–17 in the APACHE II scoring system based on an ISS score above or below 22 points (n=245).
| ISS≤22 (n=173) | ISS>22 (n=72) | pb | |
|---|---|---|---|
| Age (years)a | 52.1±19 | 42.7±18 | <0.001 |
| Sex (male) (%) | 74 | 81.9 | 0.182 |
| GCSa | 12±3 | 9±4 | <0.001 |
| APACHE IIa | 14±2 | 14±2 | 0.460 |
| OF (%) | 35.3 | 13.9 | <0.001 |
| ASA-PS (%) | 0.002 | ||
| Normal | 64.7 | 86.1 | |
| Moderate | 30.6 | 13.9 | |
| Severe | 4.6 | 0 | |
| Mortality (%) | 7.5 | 25 | <0.001 |
APACHE II: Acute Physiology and Chronic Health Evaluation; ASA-PS: American Society of Anesthesiologists Physical Status; GCS: Glasgow Coma Score; OF: organ failure; ISS: Injury Severity Score.
The trauma population admitted to our ICU has similar characteristics to other series both national and internationally. We are dealing with young predominantly male patients with few comorbidities according to the ASA-PS classification and a 14% mortality rate. In our series, car crashes amount to more than 50% of admissions though the incidence rate of falls is starting to grow.9,20
The severity of trauma can be defined based on the degree of severity of anatomical lesions (AIS, ISS, NISS). The ISS anatomical model does not follow an increasing function of mortality.21,22 It has been confirmed that the ISS scoring system underestimates mortality at low values and overestimates it at the highest possible values.23 This same pattern can be seen in our series.
Back in 1997 Osler et al. proposed a modification of the ISS scoring system including the most serious anatomical lesions regardless of the body area damaged (NISS).12 Most studies agree that the NISS has a greater capacity for discrimination and calibration compared to the ISS as it is our case. The NISS is equivalent to the ISS in mildly injured patients but improves significantly in severely injured patients and those who have sustained cranioencephalic traumas.24
An exclusively anatomical assessment is not enough to assess severity since severity also depends on the physiological repercussions.25 Traditionally, the most widely used model is the TRISS method that combines anatomical and physiological aspects.7 It scores the anatomical lesion (ISS) plus the physiological repercussions (RTS) and the physiological reserve (age) depending on the type of trauma sustained (blunt or penetrating). The TRISS method is an excellent prognostic scoring system in young patients with serious anatomical lesions (ISS>15) but it underestimates mortality in patients above 60 years old.26 Today there is a progressive increase in the age of patients who sustain severe traumas.9,20
Chico-Fernández et al. used the TRISS methodology in a study of severe trauma patients admitted to Spanish ICUs with good levels of discrimination and poor calibration especially with closed traumatic injuries. In the same study, the TRISS methodology underestimated mortality in patients with low predicted mortality rates and overestimated it in patients with high predicted rates.27
We should remember here that the physiological assessment provided by the TRISS methodology is based on the RTS score that was designed for triage purposes at the site of the accident and to know when to transfer this or that patient to a trauma center.28
It is important to assess the severity of the study population. In a study conducted by Kahloul et al. on less severe trauma patients (ISS=9), the anatomical scores provide better calibrations than the physiological ones – mixed models barely improve calibration.29 The more severe the physiological repercussions are due to the severity of the lesion, such as in the subgroup of patients admitted to the ICU, the more important it will be to use physiological scores.
The mortality prediction models used at the ICU (APACHE II, SAPS II and MPM II-24) were not specifically designed for the trauma population.10 These scores provide information on the physiological reserve and the physiological repercussion of the trauma but lack anatomical assessments.
At the beginning of the 1990s the first studies that analyzed the APACHE scale in trauma populations were published and, at the time, the APACHE II scale looked like a good predictor of mortality compared to the RTS and ISS.30 Then some authors confirmed that there was a poor correlation with mortality and only in subgroups of patients (those with high comorbidities and those who had sustained cranioencephalic trauma) it improved its discrimination capacity.31,32 In the study conducted by Dossett et al. the APACHE II scale looked like a good predictor of mortality due to the importance of physiological variables such as temperature, serum creatinine and GCS.33
In a study conducted with 6905 severe trauma patients admitted to the ICU, the SAPS II physiological model overestimated mortality especially in the subgroup of patients who underwent urgent surgeries and in patients under 60. The fusion of both scores (SAPS-TRISS) improves risk adjustment.26
During the last few years there has been a significant increase in the age of trauma patients and associated comorbidities and this has had greater physiological repercussions.27 This may explain why physiological or mixed scoring systems have a better calibration and discrimination capacities.1,29
Our findings confirm the good correlation among the different scores studied, which in turn confirms the correlation between the severity of anatomical lesions and physiological repercussions.
Although the APACHE II physiological model has a greater discrimination capacity, it should be complemented with the anatomical model (ISS) in certain groups of patients. The classification tree method is capable of identifying these groups with differential characteristics. The group of patients with relatively low scores in the APACHE II (in our findings between 10 and 17 points) should be distinguished based on their average damage sustained measured using the ISS. This score obtained in the APACHE II may be due to older patients with associated comorbidities, or young patients with cranioencephalic trauma and neurological damage (measured using the Glasgow score). It is in these young patients and without a prior clinical history of chronic disease where neurological damage conditions higher mortality rates, which does not seem to discriminate a score of the APACHE II model.
The main limitation of our study is that it was conducted in one center only and with a limited number of patients. To be able to generalize our findings it would need to be tested in other groups of patients.
Another limitation of our study has to do with the poor calibration obtained by the different models applied to our series of trauma patients admitted to ICUs. Other studies also show this finding too.4,34 This makes us look for new models or modify the ones we already have to improve accuracy when it comes to determining severity in this group of patients.
In trauma patients admitted to the ICU, the physiological models have some advantages compared to the anatomical ones. There are subgroups of these patients (with low scores obtained in the physiological scores) that require the participation of anatomical repercussion if we want to establish severity.
AuthorshipLS and JT designed the study and analyzed the findings. All authors participated in the process of data mining, all provided their own interpretation of the results, and then drew the corresponding conclusions. Also, all authors contributed to the manuscript and accepted its final version.
Conflicts of interestNo conflicts of interest were reported whatsoever.
Please cite this article as: Serviá L, Badia M, Montserrat N, Trujillano J. Gravedad en pacientes traumáticos ingresados en UCI. Modelos fisiológicos y anatómicos. Med Intensiva. 2019;43:26–34.