Since the publication of the “2012 National Consensus Document on Donation in Asystole”,1 and ever since the “Royal Decree 1723/2012”2 became effective back in January, 2013 the programs of controlled asystole donations (CAD) in Spain have grown exponentially.
CAD have become the most obvious way to expand the number of transplants with a total of 370 donors in 2016, which is a 75 per cent increase compared to the previous year. This type of donation amounts to 18 per cent of the total of deceased donors, that is, one out of every five donors is a donor in controlled asystole. At present, a total of 68 Spanish hospitals have active programs on this type of donation.3
As it occurs with encephalic death donations (EDD), the role that intensivists play in the management of CAD is essential. Most CAD occur in patients in situations of catastrophic neurological damage with need for mechanical ventilatory support who remain hospitalized in our units, and whose families have accepted their decision of life support treatment limitation (LSTL). In some cases, the withdrawal of all measures is conducted after treatment has been rejected following the explicit wishes of the patient or his/her representatives.
The normothermal abdominal perfusion (NAP) with extracorporeal membrane oxygenation (ECMO) has been the most widely used preservation method for the last few years (22 per cent in 2015) for various reasons: there is a wide experience using it in uncontrolled asystole donations (UCAD); the surgical technique is similar to the one used with EDD; it is less conditioned by the donor's anatomy; there is a possibility of conducting intraoperative assessments of the donor's organs with expanded criteria; and there is the possibility of conducing intraoperative biopsies for organ assessment. The 2015 results suggest a lower delayed graft function and a higher survival rate of the hepatic graft whenever ECMO systems are used as a preservation method.4
The Spanish experience with the use of NAP with ECMO for the management of UCAD has encouraged other countries like Great Britain5 and the United States6 to use it as a preservation method for CAD. In the experience of the University of Michigan,6 the results achieved in renal, hepatic, and pancreatic transplants are the same as in EDD, and far more superior than the super-rapid recovery technique used in CAD–shorter delays in the early graft function.
One of the aspects that make it difficult to spread the use of NAP with ECMO is that it is a complex procedure that is not available in every hospital, especially those of rural areas, where there can be patients who have made LSTL decisions who can still be donors in CAD.
The Complejo Hospitalario Universitario de Granada has been running the UCAD program since 2010,7 and the CAD program since 2013. From 2010 there have been 85 asystole donations, and 71 abdominal preservations with ECMO systems in normothermia have been conducted. Among them, 23 have been in CAD, and in 20 of them, the NAP with ECMO system was used, which allowed 37 renal transplants, and 9 hepatic transplants. The ECMO system is run by transplant coordinators–all of us intensivists from the Intensive Care Unit here at the Complejo Hospitalario Universitario de Granada.
We share the experience of having had four ECMOs trips outside our hospital since 2015: 2 to the Hospital Clínico San Cecilio–within the same city (1km away), and 2 to a different province–Jaén, Spain. The trip to this province was to the Complejo Hospitalario de Jaén (92km away), and to the Hospital San Agustín de Linares (136km away). The mobile team deployed included one doctor from the transplant coordination team (intensivist); one nurse from the transplant coordination team; one vascular surgeon (for ECMO cannulation); and one urologist from the team of renal extraction. A second urologist was provided by the center where the extraction is conducted. This is the mobile team that will be deployed together with the material needed for the cannulation and the machine required for the ECMO system. The first two cases were conducted using the ECMO Maquet Rotaflow® system, and ever since 2016 we have been using the ECMO Maquet Cardiohelp® system (better monitoring and mobility). No general surgeon traveled with the team because the four cases reported were just renal donors (due to contraindication for hepatic donation).
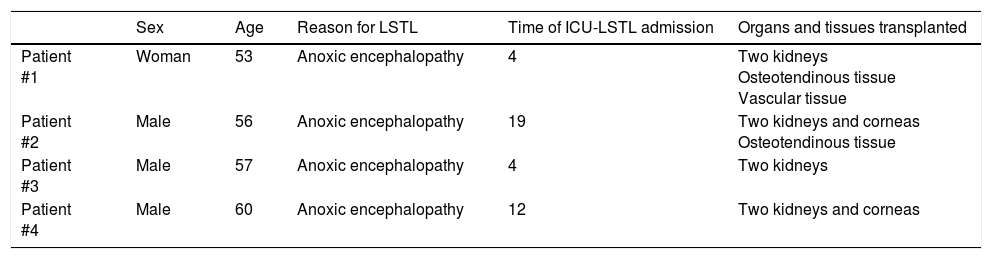
The donors were hospitalized in the intensive care units (ICU) of the aforementioned hospitals. Three males, and one woman with an average age of 56.5 years old (53–60). The reason for LSTL was anoxic encephalopathy in all four cases. The average time elapsed from ICU admission until LSTL was 9.75 days (4–19). Eight renal transplants; 4 corneal transplants; 2 extractions of osteotendinous tissue; and 1 extraction of vascular tissue were conducted (Table 1). Both the renal grafts and the receivers have had good progression. After digging into the available medical literature, we have not found any series published so far about donors in asystole being preserved using this method of deployed ECMO mobile teams for NAP in CAD.
Characteristics of donors.
| Sex | Age | Reason for LSTL | Time of ICU-LSTL admission | Organs and tissues transplanted | |
|---|---|---|---|---|---|
| Patient #1 | Woman | 53 | Anoxic encephalopathy | 4 | Two kidneys Osteotendinous tissue Vascular tissue |
| Patient #2 | Male | 56 | Anoxic encephalopathy | 19 | Two kidneys and corneas Osteotendinous tissue |
| Patient #3 | Male | 57 | Anoxic encephalopathy | 4 | Two kidneys |
| Patient #4 | Male | 60 | Anoxic encephalopathy | 12 | Two kidneys and corneas |
LSTL: life support treatment limitation; ICU-LSTL: intensive care unit-life support treatment limitation.
The role played by intensivists in the field of organ donation is essential ever since the very first programs of donation and transplant were implemented in Spain, until the latest strategies in detection and maintenance of donors.8 In just a few years, CAD have become the most important way to increase the number of organ donations in our country. Recent high-interest recommendations from SEMICYUC task forces on bioethics and transplants on how to avoid therapeutic obstination; make LSTL decisions; and assess the possibility of CAD in patients who have made LSTL decisions see us as the most relevant professionals in the management of this type of donation.9 Our capacitation in the management of life support systems allows us to guarantee the adequate preservation of the donor's organs in CAD through the use of ECMO systems. In our opinion, deploying mobile teams of intensivist transplant coordinators with ECMO systems makes CAD possible in hospitals where this resource is not available,10 which, in turn, increases the chances of donation in our ICUs.
Please cite this article as: Pérez-Villares JM, Lara-Rosales R. Desplazamientos interhospitalarios de un equipo móvil para preservación de órganos con oxigenación por membrana extracorpórea en donantes en asistolia controlada. Med Intensiva. 2018;42:131–133.