An outbreak produced by a new coronavirus in the Chinese province of Wuhan was declared in December 2019.1 The disease caused by this virus (SARS-CoV-2) is known as COVID-19, and the resulting pandemic has placed a heavy burden upon Intensive Care Units (ICUs), with many critically ill patients.2 The diagnosis is established from the detection of viral RNA in nasopharyngeal smears or bronchoalveolar lavage (BAL) samples.3 The host immune response in turn is established from blood IgM and IgG determinations using rapid qualitative tests.
In the clinically less severe cases, viral RNA detection is maximum during the first two weeks from symptoms onset,4 and after 7–10 days an IgM and subsequent IgG immune response is observed.5 However, in the more seriously ill patients, or in individuals with a prolonged course of the disease, increased persistence of the virus has been reported, to after 21 days from symptoms onset.4,6 In these cases characterized by a prolonged course, increased persistence of the IgM titers has moreover also been observed.5 The implications of all this in relation to patient isolation withdrawal are very important.7 The United States Centers for Disease Control (CDC) proposes as a safe strategy the determination of two consecutively negative rRT-PCR (real time reverse-transcription polymerase chain reaction) tests for assessing the need for isolating individuals with COVID-19.8
We present a retrospective descriptive analysis of the records of 10 intubated critical patients with SARS-CoV-2 infection admitted to the ICU of a district hospital and subjected to mechanical ventilation for over a week. Permission was obtained from the patients or their families for data collection. Critically ill patients were regarded as those presenting critical hypoxemia (paO2/FiO2< 100 for over 12 h) in the first 5 days, renal failure requiring extrarenal replacement therapy, or the need for norepinephrine at doses > 1 μg/kg/min.
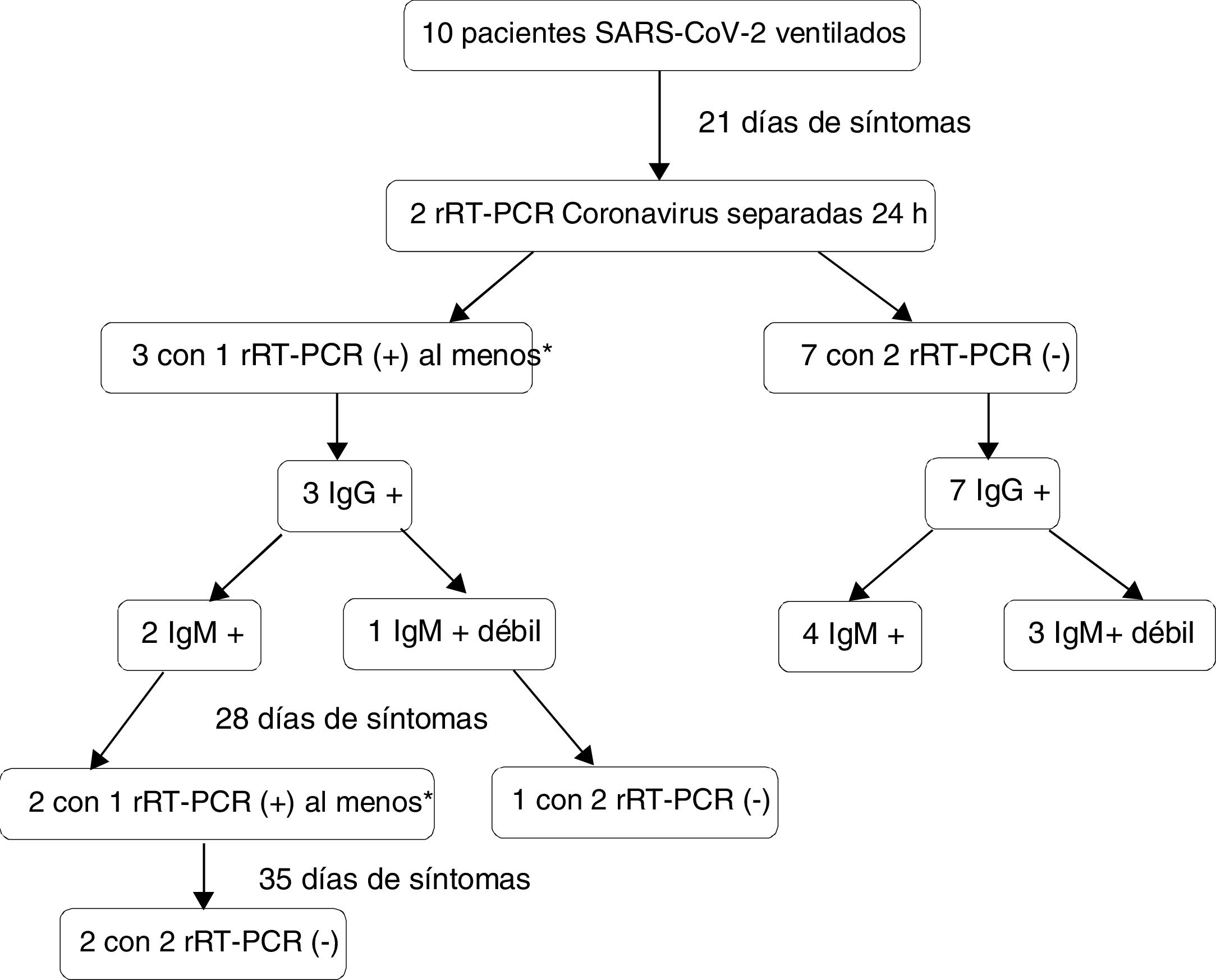
These patients were subjected to two coronavirus rRT-PCR tests spaced 24 h apart from 21 days after symptoms in order onset to assess persistent viral elimination. In the positive cases, the two tests were repeated on a weekly basis. At the same time we evaluated patient serological status based on the serum coronavirus IgM and IgG titers. Isolation was withdrawn in the negative cases, with monitoring of the professionals and relatives that had been in contact with the patient.
In our setting, in the presence of high circulating virus levels, we apply a screening algorithm with the detection of coronavirus envelope gene (E) as the parameter defining SARS-CoV-2 infection.9 The rRT-PCR amplification test (TIB Molbiol, Roche®) was performed in nasopharyngeal exudates or BAL samples, using the under 40 cycle as positivity criterion.10,11 The qualitative detection of IgG/IgM antibodies against SARS-CoV-2 was carried out using a solid phase immunochromatographic assay in whole blood, serum or plasma (Zhejiang Orient Gene Biotech Co., Ltd). In comparison with rRT-PCR, this assay has a sensitivity of 87.9% and 97.2% for IgM and IgG, respectively, and a combined specificity of 100%.
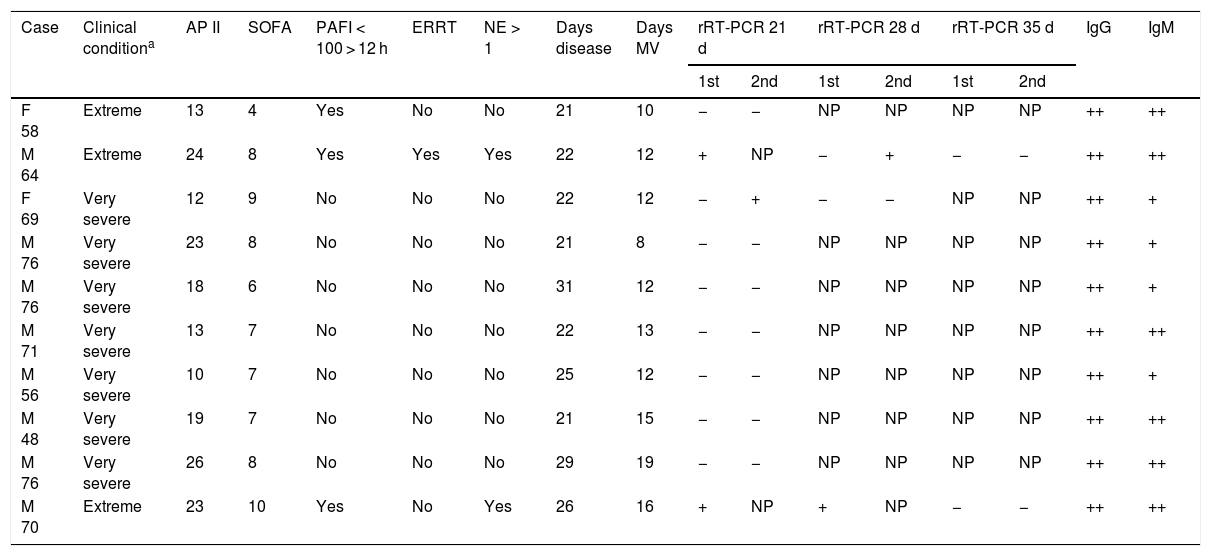
Table 1 describes the analyzed patients, who presented a mean (± standard deviation [SD]) APACHE II score of 18.1 ± 5.8 after 24 h, and a mean SOFA score of 7.4 ± 1.6 after 72 h. Males predominated in a proportion of 4:1. The mean duration of the clinical condition at the time of rRT-PCR was 24 ± 3.6 days, the mean ICU stay at that time was 16.3 ± 3.5 days, and the mean duration of mechanical ventilation was 12.9 ± 3.1 days.
Characteristics and diagnostic tests of the critical patients with SARS-CoV-2 infection subjected to mechanical ventilation.
| Case | Clinical conditiona | AP II | SOFA | PAFI < 100 > 12 h | ERRT | NE > 1 | Days disease | Days MV | rRT-PCR 21 d | rRT-PCR 28 d | rRT-PCR 35 d | IgG | IgM | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1st | 2nd | 1st | 2nd | 1st | 2nd | |||||||||||
| F 58 | Extreme | 13 | 4 | Yes | No | No | 21 | 10 | − | − | NP | NP | NP | NP | ++ | ++ |
| M 64 | Extreme | 24 | 8 | Yes | Yes | Yes | 22 | 12 | + | NP | − | + | − | − | ++ | ++ |
| F 69 | Very severe | 12 | 9 | No | No | No | 22 | 12 | − | + | − | − | NP | NP | ++ | + |
| M 76 | Very severe | 23 | 8 | No | No | No | 21 | 8 | − | − | NP | NP | NP | NP | ++ | + |
| M 76 | Very severe | 18 | 6 | No | No | No | 31 | 12 | − | − | NP | NP | NP | NP | ++ | + |
| M 71 | Very severe | 13 | 7 | No | No | No | 22 | 13 | − | − | NP | NP | NP | NP | ++ | ++ |
| M 56 | Very severe | 10 | 7 | No | No | No | 25 | 12 | − | − | NP | NP | NP | NP | ++ | + |
| M 48 | Very severe | 19 | 7 | No | No | No | 21 | 15 | − | − | NP | NP | NP | NP | ++ | ++ |
| M 76 | Very severe | 26 | 8 | No | No | No | 29 | 19 | − | − | NP | NP | NP | NP | ++ | ++ |
| M 70 | Extreme | 23 | 10 | Yes | No | Yes | 26 | 16 | + | NP | + | NP | − | − | ++ | ++ |
AP II: APACHE II (Acute Physiology, Age, Chronic Health Evaluation) score after 24 h; d: days; M: male; IgG: immunoglobulin G; IgM: immunoglobulin F; NE > 1: norepinephrine at dose > 1 μg/kg/min; F: female; NP: not performed; PAFI: paO2/FiO2; rRT-PCR: real time reverse-transcription polymerase chain reaction for coronavirus RNA; SOFA: Sequential Organ Failure Assessment score after 72 h; ERRT: extrarenal replacement therapy; MV: mechanical ventilation.
In three cases (30%; 95% confidence interval [95%CI]: 7–65%), viral RNA was detected in one of the samples after the 21 day period. One patient presented a first negative test followed by a positive test. In the rest of the cases both tests proved negative (Fig. 1). With regard to the severity of the condition, two of the three extremely severe cases showed persistent viral RNA detection, versus one of the 7 very severe cases (p = 0.18). Of the three patients with positive rRT-PCR, two of them continued to present detectable viral RNA levels in at least one sample after one week. These two cases experienced negative conversion in the following week. All the patients developed an immune response as evaluated by the antibody titers, and none showed negative conversion of IgM on day 21 – though the IgM response was qualitatively lower in four of the 7 very severe cases, versus in none of the extremely severe cases (p = 0.20). Isolation withdrawal was decided in the negative cases, and after a mean follow-up of 13.5 ± 2.6 days, no symptoms were detected in either the families or the supervising professionals, and no positive PCR tests were recorded in any professional of the hospital.
The detection of viral RNA using rRT-PCR appears to be adequate for evaluating the need for isolating patients with SARS-CoV-2 infection.8,12 This has implications in relation to work load, the use of protective equipment and care, and moreover influences the patient mood state and facilitates reassignment of the patients in other areas.
Doubts remain as to whether the detection of viral RNA in late phases corresponds to truly infective viruses or to viral RNA fragments unable to generate new disease.12 Nevertheless, it seems reasonable to require two negative samples to confirm non-infectivity, taking into account the possibility of false-negative readings, as evidenced in two of our cases. The correlation between patient serological response and effective infection control is not clear. The persistence of IgM titers, perhaps more so in the more seriously ill cases, could be related to the intensity of presentation.
The limitations of this study are its limited sample size; the fact that rRT-PCR determination of an envelope gene common to other coronaviruses was used (despite being validated in high circulation situations); and the use of qualitative antibody testing.
In sum, latent infectivity in critical patients with SARS-CoV-2 after 21 days of disease has not been sufficiently established. Persistent viral RNA levels are evidenced beyond four weeks in the more seriously ill cases. The confirmation of two consecutively negative rRT-PCR tests and the detection of IgG antibody titers could be regarded as a valid strategy in deciding patient isolation withdrawal.
Please cite this article as: García Garmendia JL, Ramírez Arcos M, Barrero Almodóvar AE, Chávez Caballero M, Jorge Amigo V, Serrano Martino MC. Detección viral y respuesta serológica en pacientes críticos intubados con SARS-CoV-2. Implicaciones para retirada de aislamiento. Med Intensiva. 2020;155:586–588.