The judicious use of existing antibiotics is essential for preserving their activity against infections. In the era of multi-drug resistance, this is of particular importance in clinical areas characterized by high antibiotic use, such as the ICU. Antibiotic dose optimization in critically ill patients requires sound knowledge not only of the altered physiology in serious infections – including severe sepsis, septic shock and ventilator-associated pneumonia – but also of the pathogen–drug exposure relationship (i.e. pharmacokinetic/pharmacodynamic index). An important consideration is the fact that extreme shifts in organ function, such as those seen in hyperdynamic patients or those with multiple organ dysfunction syndrome, can have an impact upon drug exposure, and constant vigilance is required when reviewing antibiotic dosing regimens in the critically ill. The use of continuous renal replacement therapy and extracorporeal membrane oxygenation remain important interventions in these patients; however, both of these treatments can have a profound effect on antibiotic exposure. We suggest placing emphasis on the use of therapeutic drug monitoring and dose individualization when optimizing therapy in these settings.
El uso sensato de los antibióticos existentes resulta fundamental para mantener su actividad contra las infecciones. En la era de la resistencia a múltiples fármacos, esto resulta especialmente importante en áreas clínicas caracterizadas por un uso elevado de antibióticos, como por ejemplo las UCI. La optimización de la dosis de antibióticos en pacientes críticamente enfermos requiere sólidos conocimientos no solo sobre las alteraciones fisiológicas asociadas a las infecciones graves (incluida la sepsis grave, el choque séptico y la neumonía asociada a la ventilación) sino también sobre la relación entre patógenos y la exposición a fármacos (esto es, el índice farmacocinético/farmacodinámico). Es importante considerar el hecho de que los cambios extremos en la función orgánica, como los observados en pacientes hiperdinámicos o en aquellos con síndrome de disfunción multiorgánica, pueden tener un efecto sobre la exposición a los fármacos, por lo que se requiere una vigilancia constante al revisar los regímenes posológicos de los antibióticos en los pacientes críticamente enfermos. La terapia de reemplazo renal continuo y la oxigenación por membrana extracorporal siguen constituyendo intervenciones importantes en este tipo de pacientes; no obstante, ambos tratamientos pueden tener un profundo impacto sobre la exposición a los antibióticos. Sugerimos poner un especial énfasis sobre el uso de la monitorización farmacoterapéutica y sobre la individualización de la dosis al optimizar el tratamiento en estos entornos terapéuticos.
Given an era of accelerating bacterial resistance and dwindling antimicrobial resources, optimization of existing antibiotic therapy is an increasingly important consideration for clinicians worldwide. There is perhaps added significance among intensive care unit (ICU) patients, as these patients are at high risk of infection-related mortality.1 They are also more prone to acute and often marked changes in pathophysiology and are subject to invasive intervention, both of which have the potential to significantly alter drug exposure.2
Antibiotics are one of the most commonly prescribed drug classes in the ICU and they have the ability to dramatically improve patient outcomes.3,4 Therefore, it stands to reason that maximizing the likelihood of eradicating infection whilst lowering the risk of unwanted effects should be the end goal for all prescribed antibiotic therapy. This is perhaps why determining the relationship between exposure of antibiotics in vivo (i.e. pharmacokinetics, PK) and what clinical response it elicits (pharmacodynamics, PD) has gained much prominence in critical care literature. The purpose of this review is to discuss variations in PK that commonly occur among the critically ill, through both existing pathophysiology as well as associated interventions, and to offer insights on how antibiotic therapy can be tailored accordingly so as to optimize desired PD effects.
Severe sepsis and septic shockSepsis is a common occurrence within the ICU and is defined as a systemic inflammatory response to infection, which can be complicated by organ dysfunction (‘severe sepsis’) or persistent hypotension refractory to fluid resuscitation (‘septic shock’).5 Although the incidence of sepsis worldwide is currently unknown, extrapolations from United States data conservatively estimate an occurrence of 19 million cases annually.6,7 Added to this high burden are reports indicating a severe sepsis mortality rate of nearly 30% in developed countries.8,9
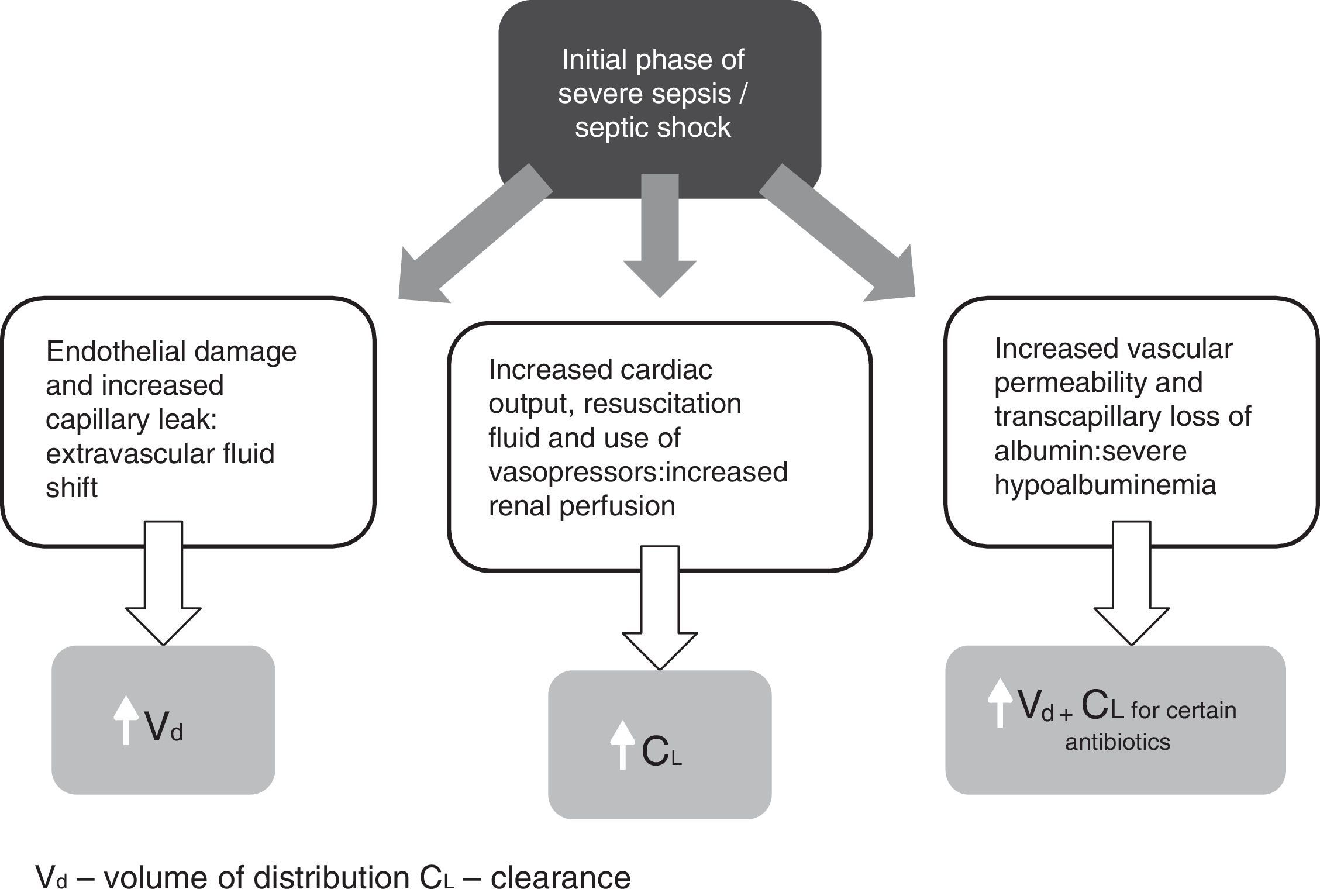
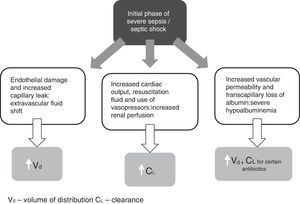
Initial pathophysiology and its impact on antibiotic pharmacokinetics (Fig. 1)One of the first clinical observations during severe sepsis and septic shock is significant fluid shift from the intravascular compartment (i.e. blood vessels) into interstitial space due to endothelial damage and increased capillary leak.10 The resultant hypotension is often fulminant, requiring urgent and aggressive delivery of intravenous resuscitation fluid, which in turn can further increase interstitial volume. Depending on the physiochemical properties of the antibiotic, these large amounts of extravascular fluid may have a significant impact on drug distribution and hence dosing strategies. For example, initial doses of hydrophilic antibiotics (i.e. that concentrate more in blood and interstitial fluid of tissues) will need to take into account these increased volumes. In this case, due to the greater volume of distribution (Vd) noted for these drugs, use of loading doses should be considered to ensure early achievement of therapeutic concentrations.11,12 Lipophilic antibiotics (i.e. that concentrate intra-cellularly and in adipose tissue), on the other hand, are not greatly influenced by changes in fluid volume and may not require alterations in initial dosing.13
A main element of the systemic inflammatory response during sepsis is that initial vasodilation is associated with a hyperdynamic cardiovascular state.14 Increased cardiac output is further driven by use of resuscitation fluid and vasopressors, resulting in increased blood flow to major organs including the kidneys. Although the exact mechanism has not been fully elucidated, the end result may be that of increased renal blood blow and glomerular filtration leading to enhanced renal drug elimination, termed ‘augmented renal clearance’ (ARC). ARC has previously been defined as a creatinine clearance ≥ 130mL/min/1.73m2.15 There are now numerous reports of the prevalence of ARC in septic as well as other groups of critically ill patients.16–18
Mathematical formulae to estimate glomerular filtration rate have all been derived from outside of the ICU and so have limited applicability in determining this increased renal clearance.19 Additionally, serum creatinine concentrations are not sensitive enough to identify patients with ARC and so a measured urine creatinine clearance collection over a specified period remains the preferred method for clinical use.17 Recent data suggests that a 2-hour creatinine clearance may be a suitable substitute for longer collection times, such as the standard 24-hour creatinine clearance,20 though the importance of accuracy of sample collection is required with shorter collection times. Although generally not applicable for determining initial antibiotic dosing regimens, it may be an important consideration in some patients, particularly younger males,21 with severe sepsis and septic shock, as important data is accumulating that ARC is associated with subtherapeutic exposure to renally cleared antibiotics such as the β-lactams.22
Another common phenomenon soon after the initial phase of severe sepsis and septic shock is the pronounced reduction in serum albumin concentrations (hypoalbuminemia).23 Albumin constitutes nearly two-thirds of all human serum proteins and is the primary protein responsible for drug-protein binding.24 Previously defined as a serum albumin concentration less than 25g/L,25 hypoalbuminemia has a direct impact on the PK of antibiotics, particularly on those antibiotics that are highly protein bound.26 Firstly, it increases the unbound or ‘free’ fraction of drug. This is important given that it is only the unbound drug that can exert a pharmacodynamic effect. As hypoalbuminemic states can increase the amount of drug that distributes into tissues, this is likely to have a direct effect on the Vd of an antibiotic. Finally, as only the unbound drug in the intravascular compartment is available for metabolism and elimination, low serum albumin concentrations may mean that there is higher proportion of unbound drug available for clearance leading to low concentrations toward the end of the dosing interval.
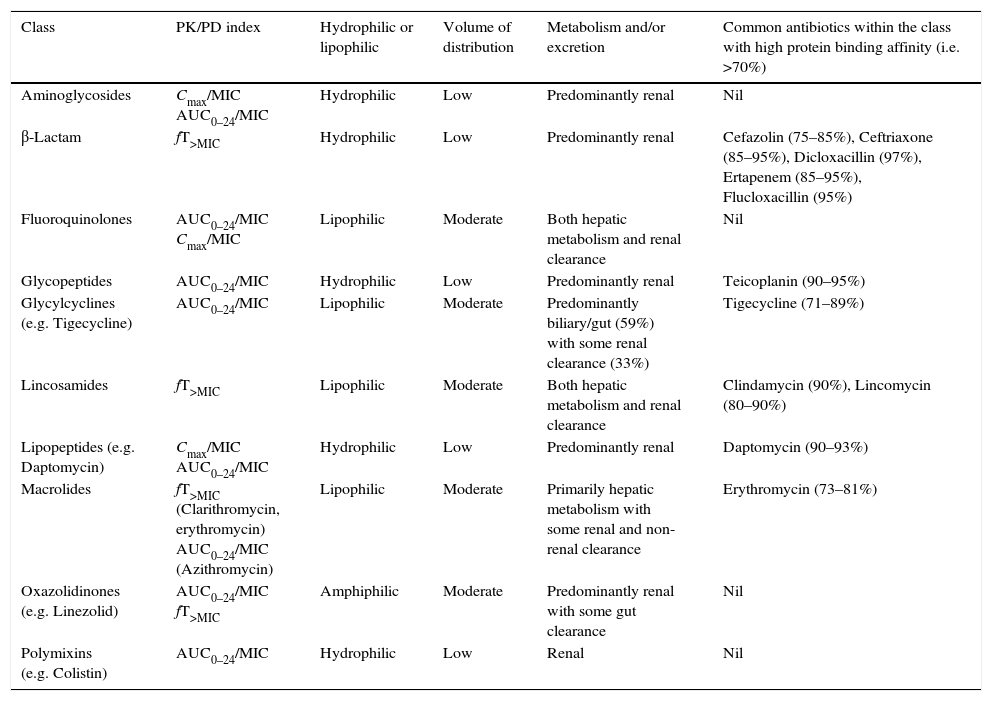
Now that the main considerations for altered drug pharmacokinetics during the initial phase of severe sepsis and septic shock have been described, the next step is to apply this knowledge when formulating dosing strategies for antibiotics based on their pathogen-exposure relationship, also known as the pharmacokinetic/pharmacodynamic (PK/PD) index (see Table 1 for summary of PK/PD index of antibiotic classes).
Summary of PK/PD index and properties of antibiotic classes. (Data for volume of distribution, metabolism and/or excretion, and protein binding26,92).
| Class | PK/PD index | Hydrophilic or lipophilic | Volume of distribution | Metabolism and/or excretion | Common antibiotics within the class with high protein binding affinity (i.e. >70%) |
|---|---|---|---|---|---|
| Aminoglycosides | Cmax/MIC AUC0–24/MIC | Hydrophilic | Low | Predominantly renal | Nil |
| β-Lactam | fT>MIC | Hydrophilic | Low | Predominantly renal | Cefazolin (75–85%), Ceftriaxone (85–95%), Dicloxacillin (97%), Ertapenem (85–95%), Flucloxacillin (95%) |
| Fluoroquinolones | AUC0–24/MIC Cmax/MIC | Lipophilic | Moderate | Both hepatic metabolism and renal clearance | Nil |
| Glycopeptides | AUC0–24/MIC | Hydrophilic | Low | Predominantly renal | Teicoplanin (90–95%) |
| Glycylcyclines (e.g. Tigecycline) | AUC0–24/MIC | Lipophilic | Moderate | Predominantly biliary/gut (59%) with some renal clearance (33%) | Tigecycline (71–89%) |
| Lincosamides | fT>MIC | Lipophilic | Moderate | Both hepatic metabolism and renal clearance | Clindamycin (90%), Lincomycin (80–90%) |
| Lipopeptides (e.g. Daptomycin) | Cmax/MIC AUC0–24/MIC | Hydrophilic | Low | Predominantly renal | Daptomycin (90–93%) |
| Macrolides | fT>MIC (Clarithromycin, erythromycin) AUC0–24/MIC (Azithromycin) | Lipophilic | Moderate | Primarily hepatic metabolism with some renal and non-renal clearance | Erythromycin (73–81%) |
| Oxazolidinones (e.g. Linezolid) | AUC0–24/MIC fT>MIC | Amphiphilic | Moderate | Predominantly renal with some gut clearance | Nil |
| Polymixins (e.g. Colistin) | AUC0–24/MIC | Hydrophilic | Low | Renal | Nil |
fT>MIC – time that the unbound or ‘free’ concentration of the drug exceeds the minimum inhibitory concentration (MIC).
Cmax/MIC – maximum antibiotic concentration (Cmax) to MIC ratio.
AUC0–24/MIC – area under the concentration-time curve during a 24-hour time period (AUC0–24) to the MIC ratio.
Antibiotics that belong to this category exhibit maximum bactericidal activity when unbound concentrations of the drug exceed the minimum inhibitory concentration (MIC) of the bacterial pathogen (fT>MIC). The main classes of antibiotics in this group include β-lactams (penicillins, cephalosporins, carbapenems, monobactams) and lincosamides (clindamycin and lincomycin).
From these, the broad-spectrum β-lactams are perhaps the most important group in the critical care setting. Particularly for Gram-negative infections, the maximal effect of bacterial killing and suppression of resistance emergence for β-lactams has previously been shown to be when the unbound trough drug concentration to MIC ratio is four to six.27–29 Increasing MICs among bacterial isolates from hospital ICUs has meant that aggressive dosing of β-lactams is a fundamental consideration in the setting of severe sepsis and septic shock. Added to this, the hydrophilicity of these agents means that they are not only prone to the effects of an increased Vd, but also increased clearance in those patients exhibiting ARC.
It is for these reasons that use of continuous or extended infusions with front-loading doses, as opposed to traditional intermittent bolus doses, have gained prominence when administering β-lactams in the ICU.30 Although some studies have shown favorable results in terms of continuous infusions attaining higher plasma β-lactam concentrations and increased rates of clinical cure when compared to intermittent bolus administration,31,32 a large randomized controlled trial is still required to confirm differences, if any, in patient outcomes.
Another important consideration in the optimization of β-lactam therapy in the ICU is the use of therapeutic drug monitoring (TDM). Measuring trough concentrations of the free fraction of β-lactams in plasma is a useful tool in ensuring that PK/PD targets for efficacy are being met. Use of TDM may be particularly important when faced with the conundrum of treating multi-resistant pathogens with high MICs to β-lactams in the setting of patients with potentially increased renal clearance.33 A trough fT>MIC ratio of one is generally recommended when administering intermittent bolus or extended infusions, whilst for continuous infusions, a steady-state fT>MIC ratio of between four to six is recommended.34 There is, however, scope for more robust studies that better define optimal β-lactam PK/PD targets.35
Lincosamides represent the other main antibiotic class demonstrating primarily a time-dependent PK/PD index. Being more lipophilic than the β-lactams, the Vd of lincosamides such as lincomycin and clindamycin are less likely to be affected by fluid shifts seen in ICU patients. However, as both antibiotics have significant protein binding (i.e. 80–90%) and are subject to hepatic as well as renal clearance, altered PK may arise in the setting of hypoalbuminemia as well as ARC. Notably, these effects are yet to be extensively investigated and so dosing regimen adjustments of lincosamides in critical care patients are largely theoretical and currently based on expert opinion.
Concentration-dependent antibioticsEfficacy best correlates with peak antibiotic concentration (Cmax) to MIC ratio (Cmax/MIC) for antibiotics with a concentration dependent PK/PD index. Aminoglycosides and metronidazole belong in this category and there are important implications when dosing these agents in the setting of increased Vd such as is seen in severe sepsis, burns and trauma.
For example, conventional dosing of aminoglycosides used in patients with septic shock have been shown consistently to have difficulty in attaining specified targets.36–38 As they are hydrophilic in nature, the pharmacokinetics of aminoglycosides such as amikacin, gentamicin and tobramycin are heavily influenced by factors such as a positive fluid balance and ARC.39,40
All of the above point to a need for a better understanding of how TDM can optimize aminoglycoside therapy in the ICU. Practically, sampling half an hour post administration of the aminoglycoside will help approximate the Cmax, with the corresponding MIC determined either via a known or suspected pathogen isolate or clinical breakpoint based on epidemiological data. Subsequent sampling anywhere between 6 and 14hours post-dose to a trough concentration will be a useful indicator for aminoglycoside clearance when the data is then analyzed using Bayesian dosing software. One upside with the aminoglycosides from a dosing point of view is that these molecules do not have a high albumin affinity, and so TDM strategies do not necessarily need to take into account potential effects of hypoalbuminemia on unbound concentrations.
Concentration-dependent with time-dependence antibioticsThe area under the concentration-time curve during a 24-hour time period (AUC0–24) to the MIC ratio (AUC0–24/MIC) is a valuable PK/PD index to describe the efficacy of antimicrobials, including glycopeptides, oxazolidinones, fluoroquinolones, polymixins, daptomycin, azithromycin and tigecycline.
For vancomycin, a PD target of AUC0–24/MIC of 400 has been advocated to achieve clinical effectiveness with vancomycin in the treatment of Staphylococcus aureus pneumonia and bacteremia,41,42 In the absence of clinical data, this ratio is a suitable PD target for other Gram-positive pathogens such as Enterococcus spp. and coagulase-negative Staphylococcus spp. However, with MICs creep for organisms such as methicillin resistant Staphylococcus aureus (MRSA),43 targets of AUC0–24/MIC of 400 are increasingly difficult to attain in the setting of severe sepsis.
To illustrate, for an isolate with an MIC of 2mg/L, the clinical breakpoint based on the European Committee on Antimicrobial Susceptibility Testing (EUCAST) data,44 an AUC0–24 of 800mg/L/h (a trough concentration of more than 20–25mg/L) would be required, increasing risks of toxicity.12 As simulated models have suggested a daily dose of 3–4g of vancomycin are needed to achieve 90% probability of attaining the above PD for an isolate with an MIC 1mg/L in ICU patients (not including patients with augmented renal clearance that would require even higher doses),45 this would again mean proportionally higher doses and more risk of toxicity when treating isolates with higher vancomycin MICs. Use of continuous vancomycin infusions have been proposed as a means to achieve target concentrations faster,46 as well as ensuring consistency in serum concentrations.47 However, there is only retrospective outcome data on the advantages of continuous versus intermittent vancomycin dosing.48
Fluoroquinolones, such as ciprofloxacin, display predominantly concentration-dependent killing with time-dependent microbiologic eradication. As ciprofloxacin is lipophilic and concentrates in tissues, changes in fluid redistribution associated with sepsis do not have a marked impact when compared to more hydrophilic antibiotics.49 However, an initial ciprofloxacin dose of 400mg intravenously (IV) has been shown to be inadequate in attaining desired AUC0–24/MIC ratios of ≥125 or Cmax/MIC ratios of >10 in the critical care setting.50 Additionally, a dosing regimen of 400mg 12-hourly IV reached optimal killing concentrations for pathogens with MICs < 0.25mg/L, a relatively low threshold for isolates in the ICU.51 Subsequently, the use of 600mg 12-hourly IV has been recommended as the optimal dosing strategy for ciprofloxacin in ICU patients not in renal failure, however, there is yet to be any trial data to validate this altered dosing regimen.
Similar to the fluoroquinolones, daptomycin exhibits a largely concentration-dependent effect together with some time-dependent killing. Daptomycin, however, is a much more hydrophilic molecule with higher protein binding (up to 93%), and as such, in vivo drug distribution may be greatly influenced by changes in Vd and hypoalbuminemia. An approved dose of 6mg/kg 24-hourly for the treatment of methicillin-resistant Staphylococcus aureus bacteraemia has shown to provide lower daptomycin exposure in the critically ill, with doses of between 8 and 10mg/kg 24-hourly having a higher probability of attaining target AUC0–24/MIC ratios.52 Of significance, regular monitoring of creatinine phosphokinase, as a marker for rhabdomyolysis, needs to be considered when utilizing higher daptomycin doses, as a rise in trough drug concentration (Cmin) have been associated with this form of toxicity.53
As the first oxazolidinone antibiotic, linezolid is effective in the treatment of Gram-positive infections, and has excellent distribution into tissues such as the lungs.54 Both AUC0–24/MIC and fT>MIC have been used to define the activity of linezolid, with optimal PK/PD indices in plasma reported as 80–120 (AUC0–24/MIC) and 2–10mg/L (Cmin) based on pathogen MICs of ≤2mg/L.55 However, although previous data has shown that standard doses of 600mg 12-hourly achieve target plasma concentrations in the general population,56 there have been several reports of subtherapeutic concentrations and treatment failure with high inter- and intra-patient variability among the critically ill.57–60
The use of a 300mg loading dose in combination with a 900mg continuous infusion on day 1 of treatment, followed by a continuous infusion of 1200mg 24-hourly thereafter has been shown to be more likely attain AUC0–24/MIC ratio of >80 compared with standard intermittent doses in a small group of critically ill patients.57 Although this may be a reasonable alternative in critically ill patients with normal renal function, larger trial data with infection-related outcomes and accounting for concomitant antibiotic therapy are required to validate these preliminary findings.
Importantly, doses higher than 1200mg per day of linezolid may be required in some instances. A well-defined linear relationship between Cmin and AUC0–24 means that there is potential to utilize Cmin as a useful PK/PD index for TDM in the future,60,61 especially in the context of higher doses. A recent study from Lopez-García et al. evaluated the preliminary safety of doses 600mg 8-hourly as part of a TDM program and found that where the Cmin remained below 6mg/L, no hematological toxicity was observed.62 In some of the patients in this analysis, the increased dose resulted in a higher Cmin and hematological toxicity was observed which highlights the importance of accurate dose adaptation.
The emergence of multi-drug resistant Gram-negative pathogens and a lack of novel antibiotics has meant that older drug classes, such as the polymyxins, have re-emerged as important treatment options. Colistin, or polymixin E, is becoming a more widely prescribed antibiotic in the ICU. Dose optimization of colistin remains complex, in part due to substantial inter-patient variability in the in vivo conversion of colistin methanesulfonate ([CMS], the prodrug) to colistin.63 The bacterial killing profile of colistin best correlates with AUC0–24/MIC, however, optimal PK/PD targets in critically ill patients are yet to be fully confirmed.64,65
One problem that has specific relevance in the critical care setting is the delay in exposure to effective therapy. Conversion of intravenously administered CMS to colistin may take several hours to achieve plasma concentrations deemed high enough to be effective. Added to this, the extent of conversion has been found to be low in the setting of normal renal function, due primarily to increased tubular secretion of CMS prior to conversion. As such, caution has been advised when using colistin as monotherapy, and the use of combination therapy, such as with carbapenems, is advocated.66 When used in conjunction with other antibiotics, use of loading doses factoring in target MICs and body weight, and maintenance doses calculated based on renal clearance is generally recommended for critically ill patients.63,67
Antibiotic dosing in multi-organ dysfunction syndrome (MODS)Termed MODS when two or more organ systems are involved, worsening organ function caused by severe infection is associated with grave outcomes and treatment failure.68 After the initial phase of severe sepsis and septic shock, ongoing sepsis-induced hypoperfusion to vital organs and tissues as a result of cardiovascular dysfunction can have a profound effect on the PK profile of both hydrophilic and lipophilic antibiotics.69 Additionally, as these patients are often sicker and in urgent need of anti-infective therapy, there is a balance between appropriate and excessive exposure to these drugs. It is important to remember that this balance is not necessarily easy to attain in the setting of fluctuating hemodynamics and organ function, and that there will always be a degree of uncertainty when adjusting dosing regimens in MODS.
Hepatic impairmentAntibiotics that are lipophilic and/or highly albumin-bound may undergo extensive liver metabolism, resulting in metabolites that are more easily eliminated. Severe hepatic dysfunction or hepatic failure may compromise this metabolism in a number of ways including a compromise in hepatic blood flow, reduced hepatocyte function and decreased biliary excretion.70 There is a risk in the presence of very severe dysfunction that accumulation of these antibiotics has the potential to result in toxicity.
One example of an antibiotic that requires dosage adjustment in hepatic dysfunction is tigecycline, which is both lipophilic and highly protein bound (71–89%). Predominantly eliminated in feces via bile acid conjugates,71 tigecycline elimination has been shown to be reduced in hepatic failure, with a 43% increase in elimination half life in patients with severe hepatic impairment.69 Thus, the manufacturer recommends halving the usual maintenance dose in severe liver disease, whilst maintaining the usual loading dose.72
Renal impairmentRenal dysfunction has dosing implications for parent compounds of hydrophilic antibiotics as well as renally cleared metabolites of some moderately lipophilic antibiotics. Renal function needs to be continually and judiciously monitored so that prompt dosage adjustments for these drugs can be made so as to minimize toxicity risk. It is important to note that the PK/PD index of the antibiotic contributes significantly to how adjustments in doses and dosing regimens are made.
For time dependent antibiotics cleared via the kidneys, such as the β-lactams, a dosage reduction rather than decreasing dosing frequency is likely to be the optimal choice in reducing drug accumulation but ensuring the fT>MIC is maintained. Conversely, concentration dependence antibiotics will require extending the dosing frequency rather than reducing the dose; therefore maximizing bacterial killing by preserving the Cmax/MIC. For concentration-dependent with time-dependence antibiotics, dosage adjustments in renal failure are usually derived based on which component of the PK/PD index predominates. For example, as the fluoroquinolones have predominant concentration-dependence with time dependence, adjusting in renal failure may entail prolonging the dosing interval rather than altering the dose.
Antibiotic dosing in continuous renal replacement therapy (CRRT)Persistent renal failure and the accumulation of metabolic waste products such as urea often warrant the need for intervention among critically ill patients. Therefore, the use of renal replacement therapy is common practice in the ICU, with CRRT, intermittent, and hybrid forms of therapy utilized. Use of CRRT involves modalities such as continuous venovenous hemofiltration (CVVH), continuous venovenous hemodialysis (CVVHD) and continuous venovenous hemodiafiltration (CVVHDF), with no best method currently established in the literature. Consequently, there is widespread heterogeneity in how CRRT is employed in the critical care setting, with variability in doses of CRRT employed, blood and dialysate flow rate, types of filters and dialysis membranes used and surface area.73,74 Added to this are the practical issues associated with reduced delivery of CRRT such as interruptions due to filter clotting and patient time away for diagnostic and procedural purposes.
Although each of these types of CRRT are efficient in removing hydrophilic antibiotics, the variation in practice means that unfortunately there is no ‘one size fits all’ in dosing recommendations for these drugs in CRRT. This may be even more appreciable when considering that the residual endogenous clearance of patients has been reported as a more useful covariate in predicting systemic antibiotic clearance than treatment with CRRT.75 Nevertheless, a brief review of antibiotic dosing studies in CRRT may help decipher some useful recommendations for dose optimization in this challenging cohort.
For example, although standardizing glycopeptide dosing across different types of CRRT is complex and problematic,76 some data report up to 50% of total glycopeptide clearance is achieved via CVVH.77,78 Based on these findings in CVVH, some investigators recommend adopting a vancomycin maintenance dose of 500–750mg 12-hourly to achieve target steady-state Cmin of 15–20mg/L.78 Others have proposed use of a 35mg/kg loading dose, most likely higher than necessary, followed by use of a 14mg/kg continuous infusion to expedite attainment of adequate Cmin values in the setting of both CVVH and CVVHDF.79 Teicoplanin clearance in CRRT may be even more variable due to hypoalbuminemic states likely leading to increased drug elimination. Use of higher doses or dose supplementation may be advisable in this setting,77 with TDM being the mainstay in guiding therapy as inter-individual variability cannot be completely controlled.80,81
Aminoglycoside exposure in the setting of CRRT has also been described.82,83 So as to attain optimal PK/PD targets in the critically ill, investigators have shown that doses of these antibiotics should remain consistent with those used in patients not on CRRT. However, data show that drug concentrations at 24hours (i.e. Cmin) remain above the threshold for renal toxicity risk,83 and so extending the dosing frequency beyond this period, to perhaps 36-hourly, should be considered together with use of TDM to determine adequate clearance.
β-Lactam dosing in CRRT remains difficult, with considerable variability in dosing required to achieve PK/PD targets and a high dependency on the type of CRRT employed.74,81 Having said this, there is now more data to support the strategy of using dosing regimens which increase the exposure of these antibiotics, when compared to other renal replacement therapies such as intermittent haemodialysis.84 Indeed, some investigators recommend using similar doses of β-lactams to those used in the absence of renal failure during the first 48hours of CRRT.85
A recent randomized controlled trial of intermittent bolus versus continuous infusion (CI) meropenem in CVVH illustrated some interesting points.86 Firstly and most importantly, the dosing regimen of meropenem used in both groups correlated with the total daily dosage recommended to be used in patients with ‘normal’ renal function. Secondly, PK/PD targets were more rapidly attained and sustained in the CI group. Finally, results indicated that if more resistant pathogens were being targeted then CI had a higher likelihood to result in more consistent achievement of PK/PD targets, indicating a selected application for meropenem CI in CRRT.
Antibiotic dosing in extracorporeal membrane oxygenation (ECMO)ECMO is an important intervention in patients with severe cardiorespiratory failure and can take on two forms: (1) venovenous (VV) ECMO which oxygenates blood from the large central veins and returns it to the venous system for distribution via the heart, thereby supporting lung function, (2) venoarterial (VA) ECMO which oxygenates blood drawn from the right atrium and returns it to the arterial system via the distal descending aorta, hence providing both respiratory and cardiac support. Both these forms require the movement of blood through an extracorporeal circuit consisting of polyvinyl chloride (PVC) tubing, an oxygenator and a heat exchanger.87 From a PK point of view, however, there is potential for drug degradation and/or adsorption of drugs to the extracorporeal circuit.88,89
Recent ex vivo experimental studies have shown significantly lower drug recovery at 24hours in the ECMO circuit compared to controls for meropenem and highly protein bound antibiotics such as ceftriaxone.89,90 As ECMO circuits represent an additional PK compartment through the use of priming solutions and the ongoing need for blood products and maintenance fluid, the Vd, particularly for hydrophilic antibiotics, may be increased.87
Interestingly, it is not only the PK profile of hydrophilic drugs that have the potential to be altered. Lipophilic drugs, such as voriconazole, have also been shown to sequester in the ECMO circuit, necessitating higher doses to maintain therapeutic concentrations.91 This is mainly thought to be due to adsorption to ECMO membranes and tubing,87 however, data from ex vivo models suggest that this cannot be extrapolated for lipophilic drugs in general. In the same study that illustrated significant losses of ceftriaxone to the ECMO circuit, Shekar and colleagues did not find the same degree of losses for ciprofloxacin and linezolid, antibiotics considered to be more lipophilic than the β-lactams.90
It is clear from these preliminary findings that much work remains to be done in quantifying the degree of altered antibiotic exposure during ECMO so that therapy can be optimized in this setting. Considering patients on ECMO may also require other treatments that impact on antibiotic exposure, such as RRT, the use of TDM and individualizing dosing regimens appears to be a useful intervention until more robust dosing data become available.
ConclusionIn summary, optimization of antibiotic therapy in the critically ill is a complex endeavor that needs to take into account various factors including different treatment modalities that can influence the success of therapy. Central to any dosing strategy, however, is knowledge of the PK/PD of the antibiotic in question. As early and appropriate antibiotic therapy is vital to ensuring beneficial outcomes in severe sepsis and septic shock, understanding initial pathophysiology and its impact on antibiotic PK is crucial to ensuring optimal use of these live saving drugs. Further, there should be an appreciation of the dynamic nature of pathophysiological effects on PK and how manifestations such as MODS can occur, requiring prompt review and appropriate alteration of therapy. Finally, interventions such as CRRT and ECMO have the ability to effect antibiotic concentrations. Given this complexity of dosing in critically ill patients, we believe that where possible, TDM should be utilized to ensure patients achieve therapeutic antibiotic concentrations.
Conflict of interestThe authors declare no conflict of interest.