Different studies have determined blood linezolid concentrations. However, the largest studies reporting data on factors associated with subtherapeutic linezolid concentrations in critically ill patients account for less than 60 patients. Thus, the objective of our study was to determine what factors were associated with subtherapeutic linezolid concentrations in critically ill patients in a larger series of patients.
DesignHistorical cohort study.
SettingOne Spanish Intensive Care Unit.
PatientsCritically ill adult patients who received linezolid due to suspected or confirmed infection by multidrug-drug-resistant Gram-positive bacteria during 2022 and 2023.
InterventionsBlood samples were collected to determine linezolid concentrations (Cmin) immediately before dosing after at least 48 h from starting linezolid therapy.
Main variable of interestSubtherapeutic linezolid concentrations.
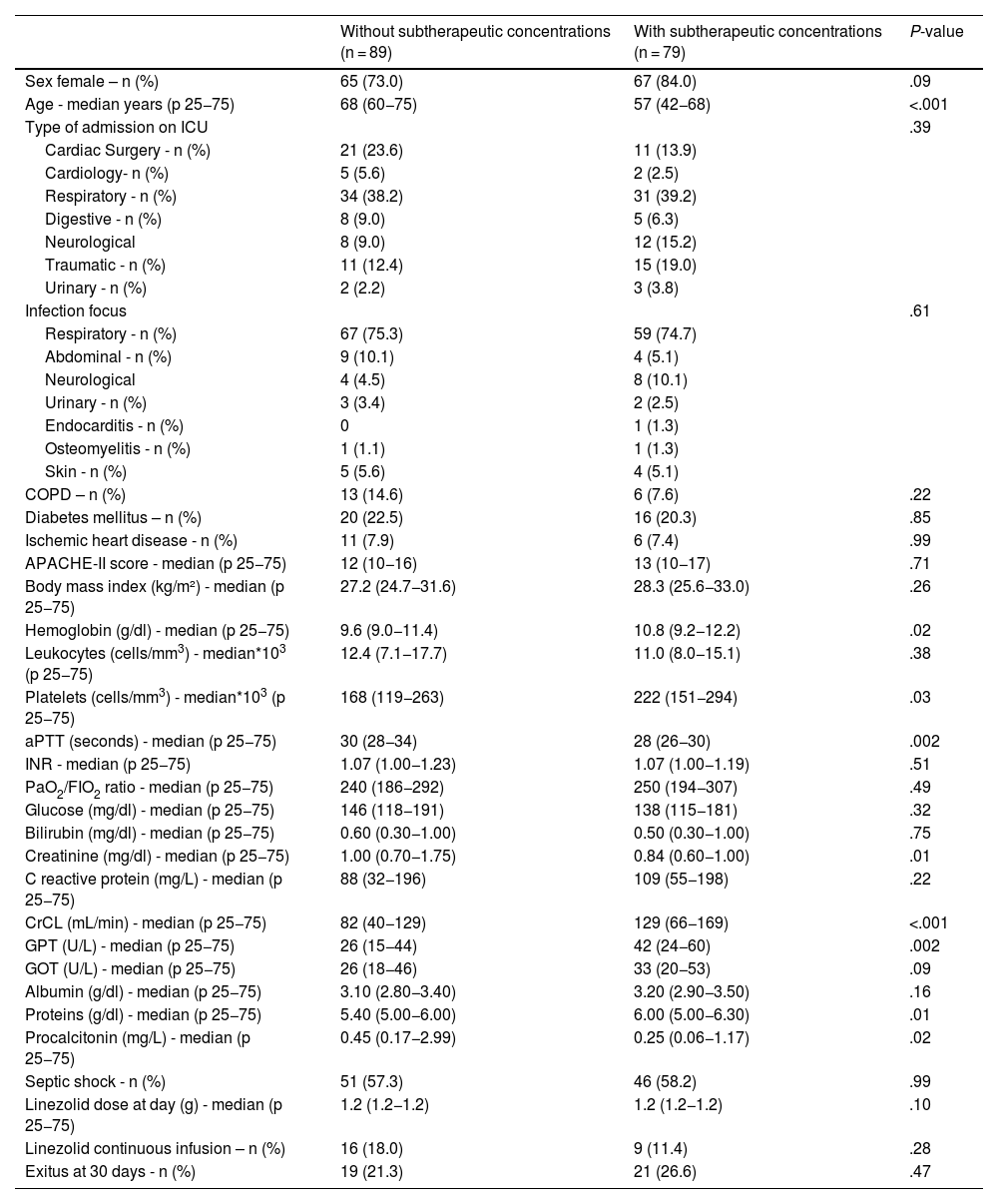
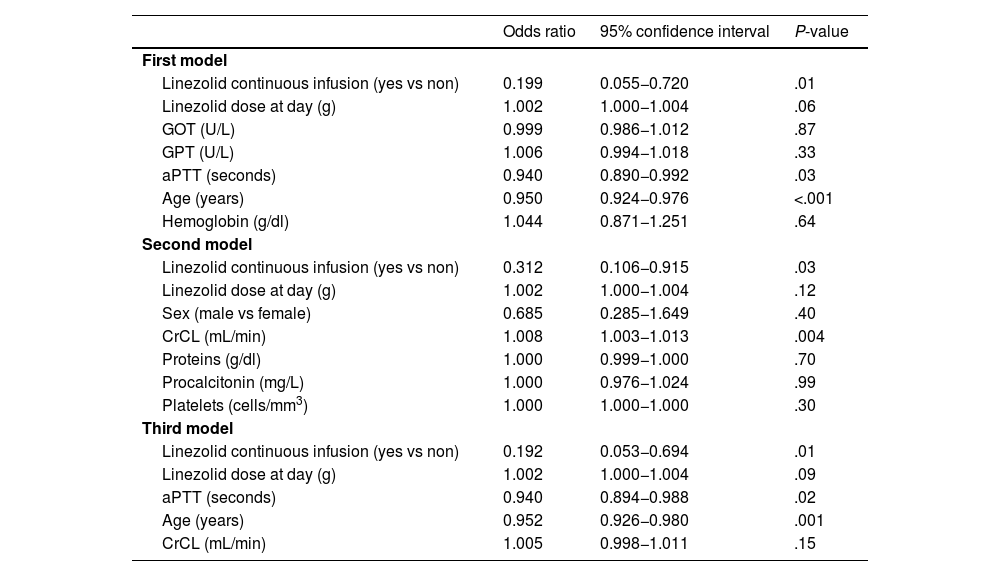
ResultsWe included a total of 168 patients. We found 79 (47.0%) patients with and 89 (53.0%) patients without subtherapeutic linezolid concentrations. Multiple logistic regression showed that linezolid continuous infusion (OR = 0.192; 95% CI = 0.053–0.694; P = .01) and older age (OR = 0.952; 95% CI = 0.926–0.980; P = .001) were associated with lower risk of subtherapeutic linezolid concentrations.
ConclusionsAs far as we know, this is the largest study reporting data on factors associated with subtherapeutic linezolid concentrations in critically ill patients. To our knowledge, our study is the first to report that linezolid continuous infusion was independently associated with lower risk of subtherapeutic linezolid concentrations in critically ill patients.
En varios estudios se han determinado los niveles sanguíneos de linezolid. Sin embargo, los estudios de mayor tamaño muestral que reportaron datos sobre los factores asociados con concentraciones subterapéuticas de linezolid en pacientes críticos incluyeron menos de 60 pacientes. Por lo tanto, el objetivo de este estudio consiste en determinar que factores se asocian con concentraciones subterapéuticas en pacientes críticos en una serie de mayor tamaño muestral.
DiseñoEstudio de cohortes histórico.
ÁmbitoUna Unidad de Cuidados Intensivos española.
PacientesPacientes críticos adultos que recibieron tratamiento con linezolid por la sospecha o confirmación de infección por bacterias grampositivas multirresistentes durante 2022 y 2023.
IntervencionesSe tomaron muestras sanguíneas para determinar los niveles valle de linezolid después de 48 horas de haber iniciado el tratamiento con linezolid e inmediatamente antes de la siguiente dosis.
Variable de interés principalConcentración subterapéutica de linezolid.
ResultadosSe incluyeron un total de 168 pacientes, 79 (47.0%) con y 89 (53.0%) sin concentración subterapéutica de linezolid. El análisis de regresión logistica mostró que la administración de linezolid en infusión continua (OR = 0.192; 95% CI = 0.053–0.694; P = .01) y mayor edad (OR = 0.952; 95% CI = 0.926–0.980; P = .001) se asociaron con un menor riesgo de concentración subterapéutica.
ConclusionesQue nosotros sepamos, este es el estudio de mayor tamaño muestral reportando datos sobre los factores que se asocian con concentraciones subterapéuticas de linezolid en pacientes críticos. Que nosotros sepamos, este es el primer estudio que reporta que la administración de linezolid en infusión continua reduce el riesgo de concentración subterapéutica en pacientes críticos.
Article
Go to the members area of the website of the SEMICYUC (www.semicyuc.org )and click the link to the magazine.