To evaluate the feasibility of adding mechanical insufflation-exsufflation (MI-E) to a weaning protocol for tracheostomized patients undergoing prolonged mechanical ventilation (MV).
DesignSingle-center, open-label, randomized, controlled pilot and feasibility study.
SettingIntensive care unit in Brazil.
PatientsTracheostomized adults who were invasively ventilated for more than 14 days and who were weaned from MV.
InterventionsPatients were randomized into two groups: intervention group (IG) or a control group (CG). During weaning with a tracheostomy collar, the IG received two daily sessions of MI-E, each with 5 cycles (insufflation and exsufflation pressures of 45 cm H2O). The CG received conventional chest physiotherapy.
Main variables of interestThe primary outcomes were the feasibility of adding MI-E to the weaning protocol and the rate of successful weaning.
ResultsA total of 47 patients were randomized, with 25 in the IG and 22 in the CG. All patients randomized to the MI-E group received the proposed protocol, completing all the scheduled MI-E sessions. Adverse events occurred in 14% of the MI-E sessions, but they did not lead to treatment interruption. Weaning success was observed in 76% of IG patients and 73% of CG patients (relative risk, 1.05; 95% CI, 0.75–1.46). No significant differences were observed between the IG and CG regarding ICU mortality (16% and 18%, respectively, p = 0.99) or hospital mortality (40% and 41%, respectively, p = 0.99).
ConclusionsThe addition of MI-E to the standard weaning protocol is feasible, but not result in a greater rate of successful weaning in tracheostomized patients.
Clinical trial registration numberU1111-1302-9035.
Evaluar la factibilidad de añadir insuflación-exsuflación mecánica (MI-E) a un protocolo de destete para pacientes con traqueostomía sometidos a ventilación mecánica (VM) prolongada.
DiseñoEstudio piloto y de factibilidad, abierto, aleatorizado y controlado en un solo centro.
ÁmbitoUnidad de cuidados intensivos en Brasil.
PacientesAdultos con traqueostomía que fueron ventilados de forma invasiva durante más de 14 días y que se encontraban en proceso de destete de la VM.
IntervencionesLos pacientes fueron aleatorizados en dos grupos: grupo de intervención (IG) y grupo de control (CG). Durante el destete con un collar de traqueostomía, el IG recibió dos sesiones diarias de MI-E, cada una con 5 ciclos (presiones de insuflación y exsuflación de 45 cm H2O). El CG recibió fisioterapia torácica convencional.
Variables principales de interésLos resultados primarios fueran la factibilidad de añadir insuflación-exsuflación mecánica (MI-E) a un protocolo de destete y la tasa de destete exitoso.
ResultadosUn total de 47 pacientes fueron aleatorizados, con 25 en el IG y 22 en el CG. Todos los pacientes del grupo MI-E recibieron el tratamiento propuesto y completaron todas sesiones de MI-E programadas. Los eventos adversos ocurrieron en el 14% de las sesiones de MI-E, pero no llevaron a la interrupción del tratamiento. El éxito del destete se observó en el 76% de los pacientes del IG y en el 73% de los pacientes del CG (riesgo relativo, 1.05; IC del 95%, 0.75 a 1.46). No se observaron diferencias significativas entre el IG y el CG respecto a la mortalidad en la UCI (16% y 18%, respectivamente, p = 0.99) ni a la mortalidad hospitalaria (40% y 41%, respectivamente, p = 0.99).
ConclusiónLa adición de MI-E al protocolo estándar de destete és factible, pero no resultó en una mayor tasa de destete exitoso en pacientes con traqueostomía.
Weaning from mechanical ventilation (MV) is a pivotal step in ventilatory support, as the risk of complications and mortality increases with increasing duration of MV.1,2 Weaning success rates are lower among patients with prolonged MV (defined as more than 21 days), and among those with prolonged weaning (defined as a weaning that was still not terminated 7 days after the first separation attempt). In such cases, the weaning period may represent 40–50% of the total MV duration.3
Moreover, hypersecretion and impaired airway clearance are important limitations in weaning patients from MV, often leading to postextubation respiratory failure and the need for reintubation.4 Patients on prolonged MV commonly present swallowing dysfunction and aspiration of oropharyngeal contents, conditions that can result in tracheobronchitis or pneumonia and increase the amount of respiratory secretion.5 Additionally, these patients may experience ineffective cough due to several factors, such as a reduction in the cough reflex induced by neurologic conditions or the cumulative effects of sedation, critical illness-associated weakness, a lack of cooperation due to delirium, and the impossibility of glottis closure due to the presence of a tracheal tube.6,7
Tracheostomy is usually indicated in patients with prolonged ventilation.8 Tracheostomy facilitates the suction of secretions from the trachea and central bronchus, representing an important strategy to wean patients from prolonged MV.9 However, suctioning secretions via the tracheal tube is ineffective for clearing the peripheral airways and less effective in clearing the left endobronchial tree, as the left mainstem bronchus has a smaller diameter than the right bronchus and leaves the trachea at a more acute angle.10
Different techniques can increase cough effectiveness, such as lung volume recruitment, manually assisted cough, and mechanically assisted cough, using a mechanical insufflation-exsufflation (MI-E) device.11 MI-E first inflates the lung with a positive pressure during a predetermined period (insufflation phase) and then applies a negative pressure (exsufflation period) that creates a peak flow high enough to move secretions toward more proximal airways, from where they can be suctioned.12
MI-E was originally used for the treatment of neuromuscular diseases, both to prevent pulmonary complications associated with sputum retention in nonintubated patients and to facilitate liberation from MV.13,14 Recently, the use of MI-E has increased for critically ill patients receiving MV, but evidence for its efficacy is still scarce.15 For example, Gonçalves et al. demonstrated that adding a MI-E protocol beginning immediately before extubation and maintained for 48 h after extubation reduced reintubation rates.16
As hypersecretion and impaired airway clearance are particularly frequent in tracheostomized patients on prolonged MV, we hypothesized that adding an MI-E protocol might facilitate the liberation of these patients from MV. Therefore, the aim of this pilot study was to determine the feasibility of adding MI-E to a structured protocol for the liberation of tracheostomized patients from MV.
Patients and methodsTrial design and oversightThis single-center, open-label, randomized, controlled pilot and feasibility study was conducted in the Intensive Care Unit (ICU) of the University Hospital of the Federal University of Juiz de Fora (Minas Gerais, Brazil) between January 2023 and December 2023; this was a clinical-surgical ICU with nine beds from a 150-bed teaching hospital. The study was approved by the local Research Ethics Committee (CAAE: 67109223.4.0000.5133), and the protocol was registered in the Brazilian Clinical Trials Registry (UTN code: U1111-1302-9035). Written informed consent was obtained from each patient or the next of kin before enrollment in the trial.
Trial populationThe study sample included patients aged 18 years or older admitted to the participant ICU, who were invasively ventilated for more than 14 days, who were tracheostomized and who met all the following criteria to undergo a weaning from MV protocol: 1. compensation for the underlying cause of respiratory failure; 2. the ability to undergo spontaneous ventilation; 3. adequate oxygenation, defined as either a PaO2/FIO2 of at least 150 or an peripheral oxygen saturation (SpO2) of at least 90%, fraction of inspired oxygen (FIO2) lower or equal to 40% and positive end expiratory pressure (PEEP) lower or equal to 8 cm H2O; 4. hemodynamic stability, defined as no use of vasopressors or the use of minimal doses; 5. a pH greater than 7.25; and 6. a core temperature lower than 38 °C. The exclusion criteria were cardiopulmonary instability, uncontrolled arrhythmia, preexisting neuromuscular disease, intracranial hypertension, thoracic trauma, bronchopleural fistula, pneumothorax, pneumomediastinum, hemoptysis, and life-sustaining treatment.
RandomizationPatients were randomized into two groups: control and MI-E. The randomization was performed through an unsystematic number sequence in a 1:1 ratio (control group:MI-E group) using a block size of four. The number sequence was generated by an independent and blinded observer using the Research Randomizer (http://www.randomizer.org). Randomization was carried out immediately before the beginning of the weaning protocol.
InterventionControl groupPatients randomized to the control group were submitted to a 7-day weaning protocol. During the first and second days, patients were disconnected from the ventilator and allowed to breathe through the tracheostomy for a maximum of 12 consecutive h. After these periods, the patients were reconnected to the ventilator and ventilated on pressure support for the remainder of the 24 h. From the third to the seventh days, the patients were allowed to breathe through the tracheostomy for 24 h/day. Successful weaning was achieved when the patient sustained five consecutive days of unassisted breathing through tracheostomy. In this case, the day of weaning was considered the first day when the patient sustained unassisted breathing for 24 h.
During the seven days, patients were monitored for the occurrence of the following criteria of protocol failure: a respiratory rate greater than 35 breaths/minute; accessory muscle use; paradoxical rib cage-abdominal movements; intercostal retractions; diaphoresis; oxygen desaturation (SpO2 persistently lower than 90% with an oxygen flow rate of at least 6 l/min); hemodynamic instability (heart rate persistently higher than 140 beats/min or systolic blood pressure lower than 90 mm Hg or higher than 180 mm Hg); and agitation. In case of failure, assist-control ventilation was instituted and maintained for the remainder of the 24 h. The protocol was reinitiated from the beginning (Day 1) on the next day or on the first day when the patient met the criteria for weaning from MV. Weaning was considered a failure when the patient was not successfully liberated from MV by 30 days after randomization.
Patients received respiratory and motor physiotherapy twice per day. Respiratory therapy included patient positioning and airways suction. Expiratory ribcage compressions and manual hyperinflation could be performed before airways suction, if indicated by the physiotherapist. Airways suctions were carried out throughout the day, if necessary. Motor physiotherapy included passive, active assisted (with manual assistance) and active (independent) upper and lower limb exercises in the supine position (a minimum of five repetitions for each joint). If these exercises were tolerated, the motor therapy was advanced to functional exercises such as sitting on the edge of bed, transferring to chair, standing up, pre-gait exercises, and walking.
MI-E groupPatients randomized to the MI-E group were submitted to the same weaning protocol and the same motor therapy, twice per day, received by those randomized to the control group. In addition, they received the treatment with MI-E with a cough assist (Philips Respironics, Murrysville, PA, USA). MI-E treatment was performed through the tracheostomy twice a day. In each section, patients were submitted to 5 cycles of insufflation at a pressure of 40 cm H2O for 2 seconds and exsufflation at a pressure of −45 cm H2O for 3 seconds, separated by a pause of 2 seconds. The procedure was finalized when no secretion could be aspirated through the tracheostomy. Extra airways suctions (without MI-E treatment) could be carried out throughout the day, if necessary.
Study measures and data collectionThe following characteristics related to admission to the ICU were obtained from the patients’ medical records: age, sex, main reason for admission to the ICU, cause of respiratory failure, SAPS 3 (Simplified Acute Physiology Score 3), SOFA (Sequential Organ Failure Assessment), and Charlson comorbidity index. On the day of enrollment in the study, we collected the SOFA score, duration of MV, duration of tracheostomy, ventilatory parameters and arterial blood gas analysis. The number of hours of unassisted breathing through tracheostomy per day and the presence of complications (ventilator-associated pneumonia, atelectasis, barotrauma, tachycardia, hypertension or hypotension, hemoptysis, oxygen desaturation, and tachypnea) were recorded daily for 30 days from the day of enrollment or until ICU discharge or death.
Outcome measuresThe primary outcomes were the feasibility of adding MI-E to the weaning protocol and the rate of successful weaning from MV. Feasibility was assessed by examining the adherence to the weaning protocol in the MI-E group. The secondary outcomes were the time to successful liberation; ICU, hospital and 60-day mortality rates; and adverse events associated with MI-E.
Statistical analysisThe analysis was performed on an intention-to-treat basis. Categorical variables are reported as frequencies and percentages, and continuous variables are reported as medians and interquartile ranges (IQRs). Comparisons between categorical variables were performed using Fisher’s exact test, and comparisons of continuous variables were performed using the Mann‒Whitney U test.
Patient weaning from MV was analyzed with the Kaplan‒Meier method and compared between the MI-E and control groups with the log-rank test. The same analysis was performed to compare mortality between the two groups. A logistic regression model was used to determine whether treatment with IE-M was independently associated with weaning success and hospital mortality. The following covariables were included in the model: age, SAPS-3 score, and Charlson comorbidity index score.
P values were two-sided, and the significance level was set at 0.05. All analyses were conducted with Stata 15.1 (Stata CorpLP, College Statio, TX, USA).
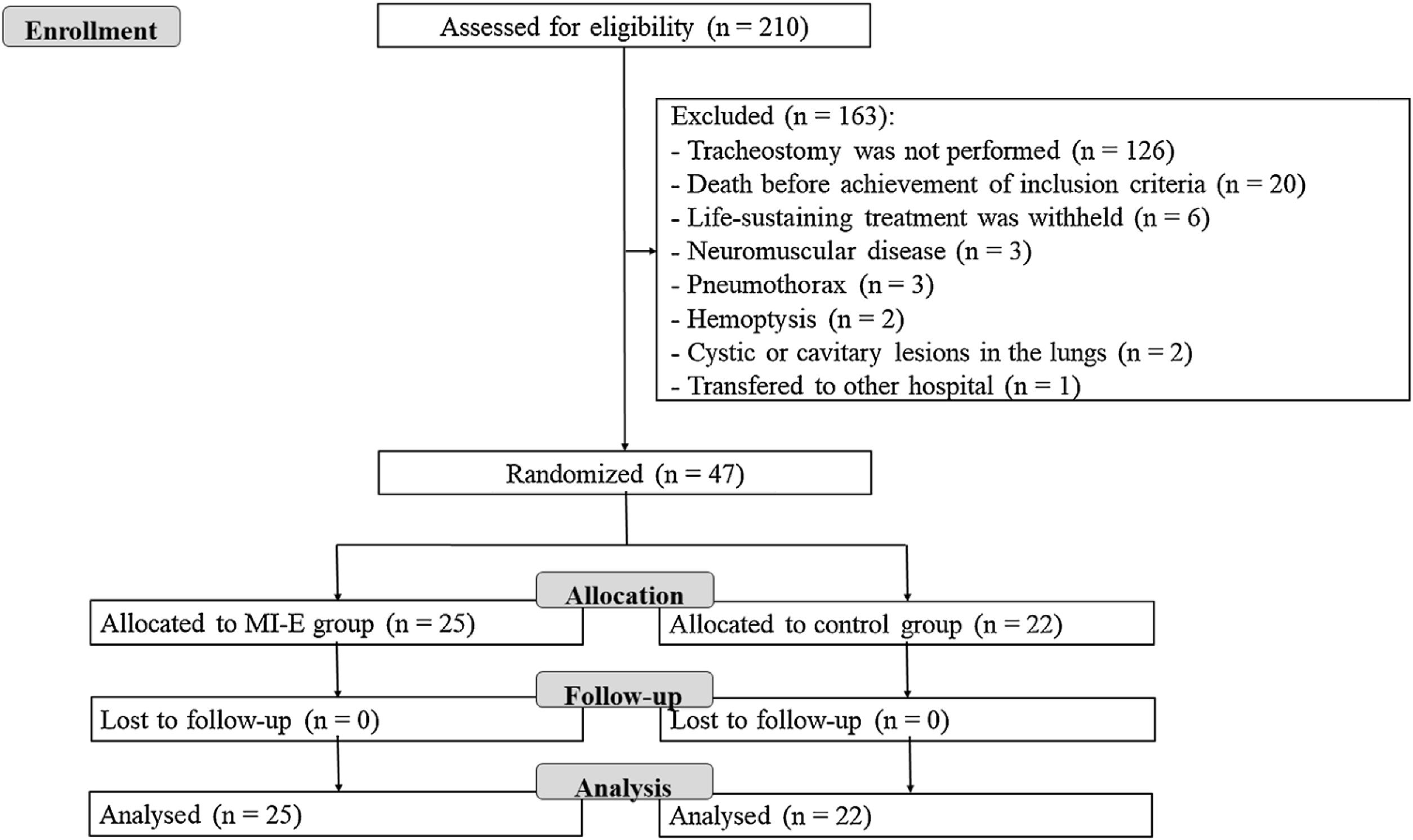
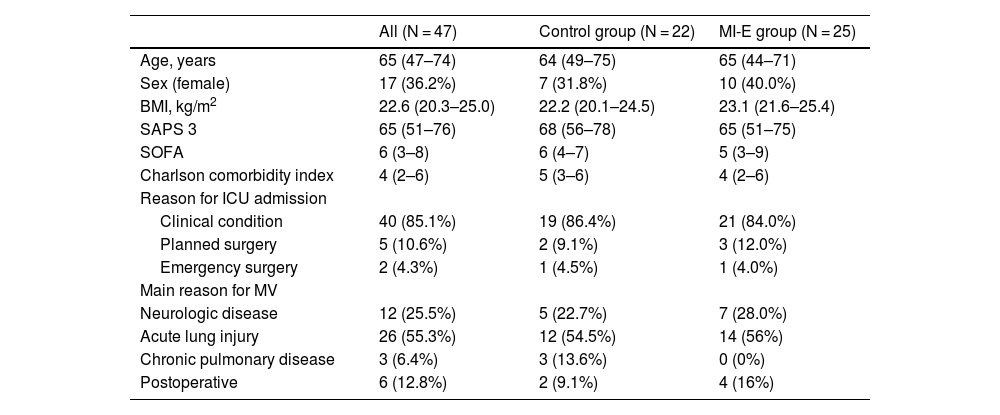
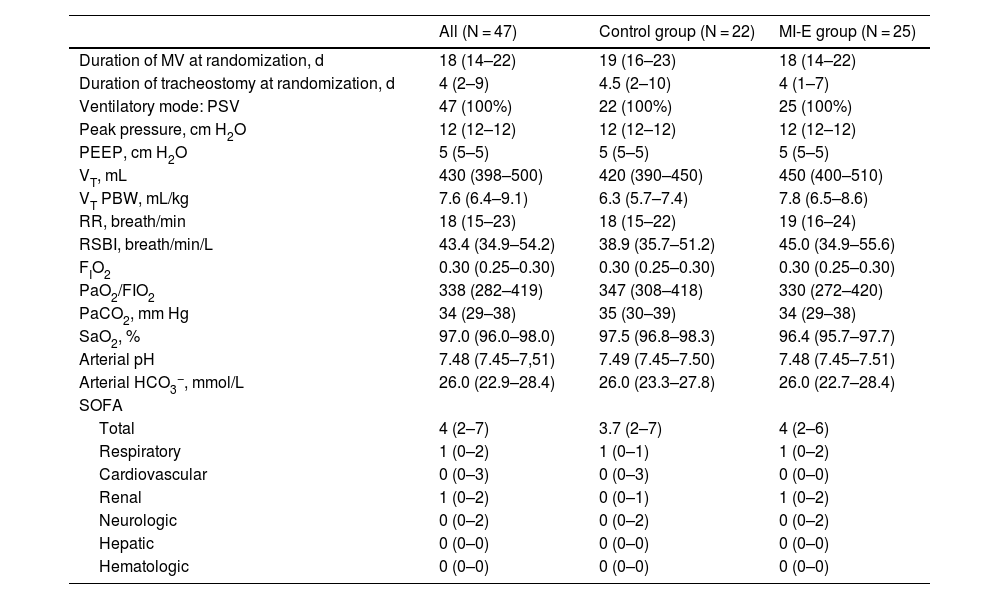
ResultsDuring the study period, 210 patients were intubated, and 84 were tracheostomized and screened for enrollment. Among these patients, 47 were randomized into two groups: the MI-E group (n = 25) and the control group (n = 22) (Fig. 1). The baseline characteristics of the patients at admission to the ICU were similar between the two groups. Most of the patients were admitted to the ICU due to clinical conditions, and acute lung injury and neurologic conditions were the main reasons for MV (Table 1). The clinical and ventilatory characteristics at randomization are shown in Table 2. At the beginning of weaning, patients had been on MV for a median of 18 days (14–22) and had been tracheostomized for a median of 4 days (2–9) (Table 2).
Baseline patient characteristics.
| All (N = 47) | Control group (N = 22) | MI-E group (N = 25) | |
|---|---|---|---|
| Age, years | 65 (47–74) | 64 (49–75) | 65 (44–71) |
| Sex (female) | 17 (36.2%) | 7 (31.8%) | 10 (40.0%) |
| BMI, kg/m2 | 22.6 (20.3–25.0) | 22.2 (20.1–24.5) | 23.1 (21.6–25.4) |
| SAPS 3 | 65 (51–76) | 68 (56–78) | 65 (51–75) |
| SOFA | 6 (3–8) | 6 (4–7) | 5 (3–9) |
| Charlson comorbidity index | 4 (2–6) | 5 (3–6) | 4 (2–6) |
| Reason for ICU admission | |||
| Clinical condition | 40 (85.1%) | 19 (86.4%) | 21 (84.0%) |
| Planned surgery | 5 (10.6%) | 2 (9.1%) | 3 (12.0%) |
| Emergency surgery | 2 (4.3%) | 1 (4.5%) | 1 (4.0%) |
| Main reason for MV | |||
| Neurologic disease | 12 (25.5%) | 5 (22.7%) | 7 (28.0%) |
| Acute lung injury | 26 (55.3%) | 12 (54.5%) | 14 (56%) |
| Chronic pulmonary disease | 3 (6.4%) | 3 (13.6%) | 0 (0%) |
| Postoperative | 6 (12.8%) | 2 (9.1%) | 4 (16%) |
The data are presented as the number of patients (%) or median (IQR).
BMI: body mass index; IQR: interquartile range; MI-E: mechanical insufflation-exsufflation; MV: mechanical ventilation; SAPS: simplified acute physiology score; SOFA: sequence organ failure assessment.
Clinical and ventilatory characteristics of patients at randomization.
| All (N = 47) | Control group (N = 22) | MI-E group (N = 25) | |
|---|---|---|---|
| Duration of MV at randomization, d | 18 (14–22) | 19 (16–23) | 18 (14–22) |
| Duration of tracheostomy at randomization, d | 4 (2–9) | 4.5 (2–10) | 4 (1–7) |
| Ventilatory mode: PSV | 47 (100%) | 22 (100%) | 25 (100%) |
| Peak pressure, cm H2O | 12 (12–12) | 12 (12–12) | 12 (12–12) |
| PEEP, cm H2O | 5 (5–5) | 5 (5–5) | 5 (5–5) |
| VT, mL | 430 (398–500) | 420 (390–450) | 450 (400–510) |
| VT PBW, mL/kg | 7.6 (6.4–9.1) | 6.3 (5.7–7.4) | 7.8 (6.5–8.6) |
| RR, breath/min | 18 (15–23) | 18 (15–22) | 19 (16–24) |
| RSBI, breath/min/L | 43.4 (34.9–54.2) | 38.9 (35.7–51.2) | 45.0 (34.9–55.6) |
| FIO2 | 0.30 (0.25–0.30) | 0.30 (0.25–0.30) | 0.30 (0.25–0.30) |
| PaO2/FIO2 | 338 (282–419) | 347 (308–418) | 330 (272–420) |
| PaCO2, mm Hg | 34 (29–38) | 35 (30–39) | 34 (29–38) |
| SaO2, % | 97.0 (96.0–98.0) | 97.5 (96.8–98.3) | 96.4 (95.7–97.7) |
| Arterial pH | 7.48 (7.45–7,51) | 7.49 (7.45–7.50) | 7.48 (7.45–7.51) |
| Arterial HCO3−, mmol/L | 26.0 (22.9–28.4) | 26.0 (23.3–27.8) | 26.0 (22.7–28.4) |
| SOFA | |||
| Total | 4 (2–7) | 3.7 (2–7) | 4 (2–6) |
| Respiratory | 1 (0–2) | 1 (0–1) | 1 (0–2) |
| Cardiovascular | 0 (0–3) | 0 (0–3) | 0 (0–0) |
| Renal | 1 (0–2) | 0 (0–1) | 1 (0–2) |
| Neurologic | 0 (0–2) | 0 (0–2) | 0 (0–2) |
| Hepatic | 0 (0–0) | 0 (0–0) | 0 (0–0) |
| Hematologic | 0 (0–0) | 0 (0–0) | 0 (0–0) |
The data are presented as the number of patients (%) or median (IQR).
FIO2: fraction of inspired oxygen; HCO3-: bicarbonate; PaCO2: partial pressure of arterial carbon dioxide; PaO2: partial pressure of arterial oxygen; IQR: interquartile range; MI-E: mechanical insufflation-exsufflation; MV: mechanical ventilation; PEEP: positive end expiratory pressure; PSV: pressure support ventilation; RSBI: rapid shallow breathing index (calculated as the ratio of RR to VT); RR: respiratory rate; SaO2: arterial oxygen saturation; SOFA: sequence organ failure assessment; VT: tidal volume.
All patients randomized to the MI-E group received the proposed protocol, completing all the scheduled MI-E sessions (100% of adherence to the protocol). A total of 270 MI-E sessions were analyzed during the study, and none was interrupted due to adverse events.
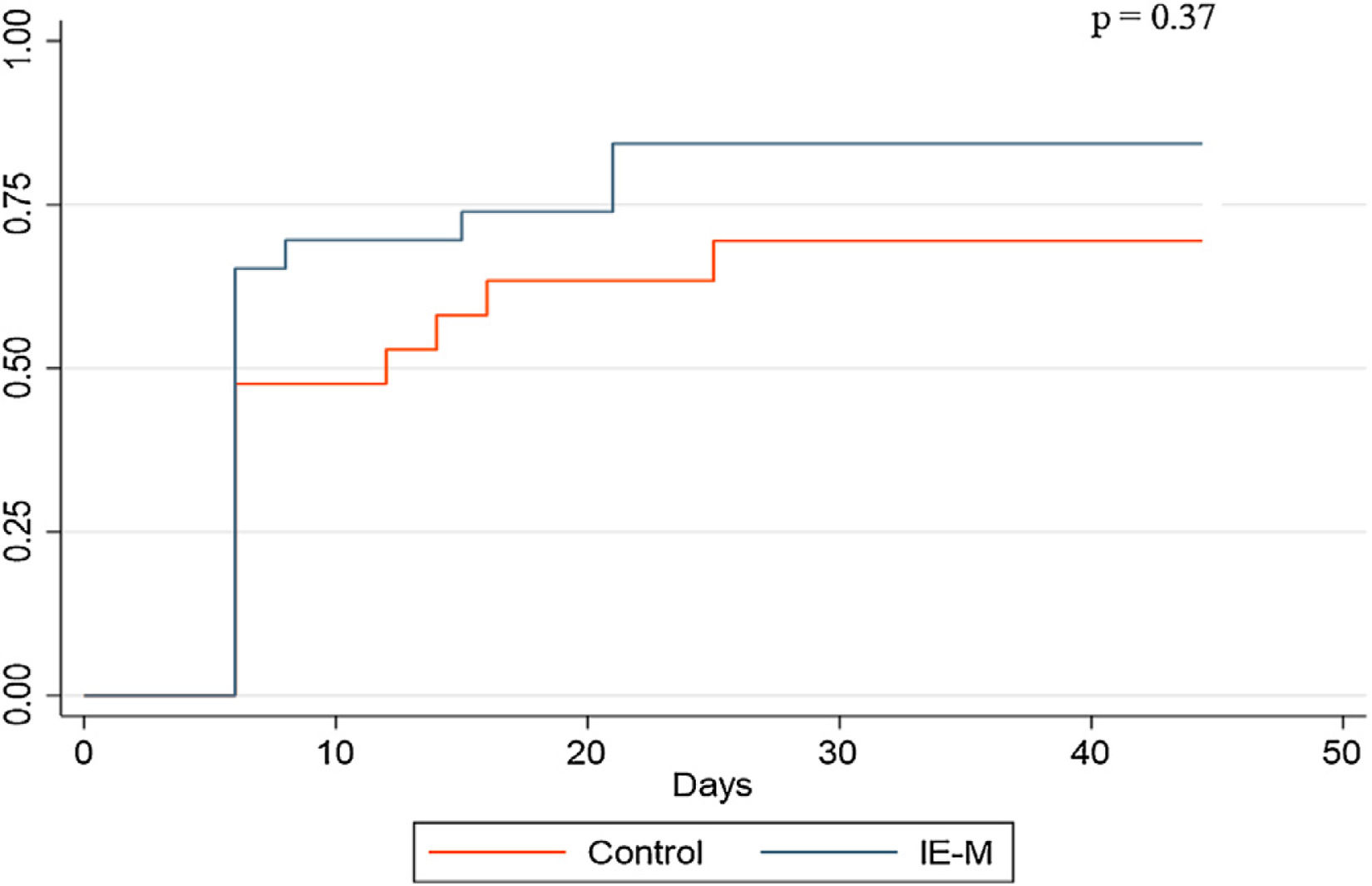
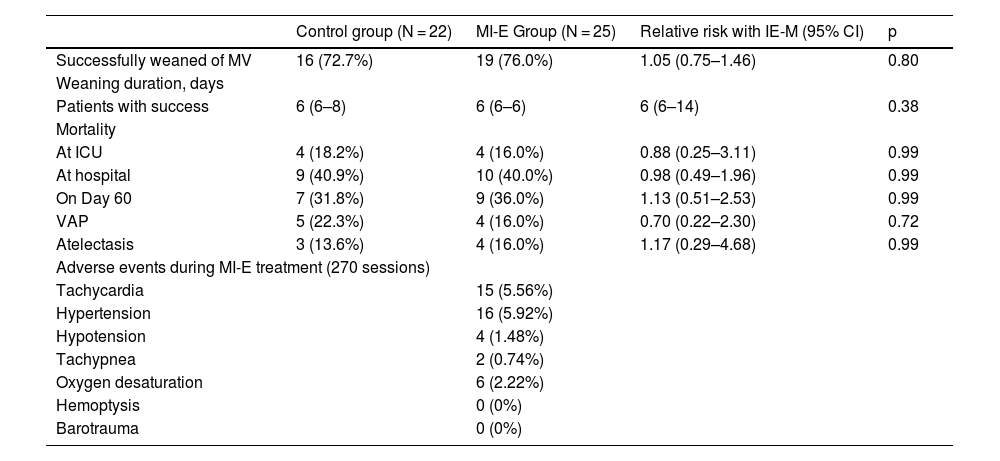
The rate of successful weaning was similar in the two groups: 76.0% in the IE-M group and 72.7% in the control group (relative risk = 1.05, 95% CI = 0.75–1.46, p = 0.75). Among patients who achieved successful weaning, the median weaning time was similar in the two groups: 6 days (IQR, 6–6) in the MI-E group and 6 days (IQR, 6–8) in the control group. There was no significant difference in ICU, hospital or 60-day mortality between the two groups (Table 3).
Primary and secondary outcomes according to study group.
| Control group (N = 22) | MI-E Group (N = 25) | Relative risk with IE-M (95% CI) | p | |
|---|---|---|---|---|
| Successfully weaned of MV | 16 (72.7%) | 19 (76.0%) | 1.05 (0.75–1.46) | 0.80 |
| Weaning duration, days | ||||
| Patients with success | 6 (6–8) | 6 (6–6) | 6 (6–14) | 0.38 |
| Mortality | ||||
| At ICU | 4 (18.2%) | 4 (16.0%) | 0.88 (0.25–3.11) | 0.99 |
| At hospital | 9 (40.9%) | 10 (40.0%) | 0.98 (0.49–1.96) | 0.99 |
| On Day 60 | 7 (31.8%) | 9 (36.0%) | 1.13 (0.51–2.53) | 0.99 |
| VAP | 5 (22.3%) | 4 (16.0%) | 0.70 (0.22–2.30) | 0.72 |
| Atelectasis | 3 (13.6%) | 4 (16.0%) | 1.17 (0.29–4.68) | 0.99 |
| Adverse events during MI-E treatment (270 sessions) | ||||
| Tachycardia | 15 (5.56%) | |||
| Hypertension | 16 (5.92%) | |||
| Hypotension | 4 (1.48%) | |||
| Tachypnea | 2 (0.74%) | |||
| Oxygen desaturation | 6 (2.22%) | |||
| Hemoptysis | 0 (0%) | |||
| Barotrauma | 0 (0%) | |||
The data are presented as the number of patients (%) or median (IQR).
MI-E: mechanical insufflation-exsufflation; MV: mechanical ventilation; VAP: ventilator-associated pneumonia.
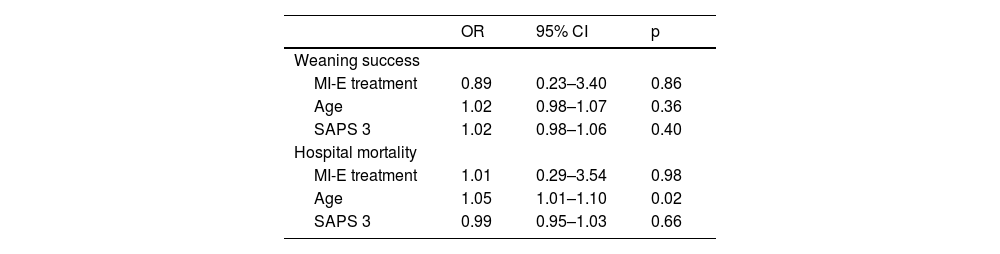
A Kaplan‒Meier plot of the proportions of patients who were successfully weaned in the two groups is shown in Fig. 2. After adjustment for age, SAPS 3 score and the Charlson comorbidity index, treatment with MI-E was neither independently associated with weaning success nor with hospital mortality (Table 4).
Multivariate logistic regression to evaluate the associations between mechanical insufflation-exsufflation and weaning success and hospital mortality.
| OR | 95% CI | p | |
|---|---|---|---|
| Weaning success | |||
| MI-E treatment | 0.89 | 0.23–3.40 | 0.86 |
| Age | 1.02 | 0.98–1.07 | 0.36 |
| SAPS 3 | 1.02 | 0.98–1.06 | 0.40 |
| Hospital mortality | |||
| MI-E treatment | 1.01 | 0.29–3.54 | 0.98 |
| Age | 1.05 | 1.01–1.10 | 0.02 |
| SAPS 3 | 0.99 | 0.95–1.03 | 0.66 |
MI-E: mechanical insufflation-exsufflation; OR: odds ratio; CI: confidence interval; SAPS: simplified acute physiology score.
Treatment with MI-E was safe. The most frequent adverse events were transitory tachycardia, hypertension, and oxygen desaturation (Table 3).
DiscussionThis pilot, single-center, randomized clinical trial is the first to evaluate the feasibility of MI-E in the weaning of tracheostomized patients receiving prolonged MV. MI-E treatment was feasible, and adverse events were infrequent in the MI-E group and did not result in interruption of treatment. We found that adding MI-E to a structured weaning protocol did not increase its success rate.
Patients on prolonged MV have difficulty weaning from MV. Ventilator dependence is multifactorial and includes the severity of the illness that causes respiratory failure, comorbid conditions, an imbalance between increased breathing and respiratory muscle capacity, ineffective airway clearance, complications of long-term ICU care (especially ventilator-acquired pneumonia and ICU-acquired weakness), and psychological factors.17,18 Tobin et al. conducted a randomized clinical trial to compare two strategies (pressure support and a tracheostomy collar) for weaning from prolonged MV in tracheotomized patients. In their study, 49% of the patients were successfully weaned, and the weaning time was shorter with the tracheostomy collar strategy.19 Using the same tracheostomy collar weaning protocol, 72% of the patients in our study were successfully weaned. This better weaning success rate might be explained by the different clinical profiles of our patients, who had been ventilated for a shorter period of time before the beginning of the weaning protocol.
In our study, the addition of MI-E neither increased the weaning success rate nor reduced its duration. Our results are in accordance with those obtained by Wibart et al., who conducted a randomized clinical trial to determine the impact of the use of MI-E after extubation in patients with intensive care unit-acquired weakness. They did not find any positive effect of MI-E on preventing postextubation respiratory failure, the reintubation rate or mortality.20 In contrast, Gonçalves et al. demonstrated that the inclusion of MI-E in a weaning protocol reduced reintubation rates.16 The differences among these studies might be related to patient characteristics, mainly their ability to depurate airway secretions. In our study, we did not restrict the inclusion criteria to patients with hypersecretion, allowing the inclusion of patients in whom the benefits of MI-E could be smaller or absent. Moreover, our patients were tracheostomized, and the importance of MI-E in improving secretion clearance might be greater in the postextubation period than in tracheostomy.
MI-E was well tolerated, and the adverse events that occurred during MI-E were transient and did not require treatment interruption. The most frequent adverse events that occurred during MI-E in the present study were tachypnea, oxygen desaturation and hemodynamic instability, which is in accordance with the literature.21–23 Barotrauma and hemoptysis, which are potentially more severe adverse events associated with MI-E, were not observed in our study.
This study has several limitations. First, this was a single-center study, and the results may not be generalizable to other critical care patients. Second, as a pilot study, the sample size was not large enough to achieve sufficient power to identify significant differences in clinical outcomes between the two groups. Third, all the patients who were randomized to the MI-E group received the same treatment, with two sessions per day of MI-E, regardless of their clinical condition. Adjusting the number of MI-E sessions to patient necessity might have improved its effectiveness.
In conclusion, adding MI-E to a structured weaning protocol is feasible. However, MI-E treatment did not increase the success rate, reduce the duration, or decrease the mortality of tracheostomized patients on prolonged MV. Since this was a pilot study with some limitations, further multicenter studies should be conducted. In these possible studies, more restrictive inclusion criteria to select patients with greater potential benefits from MI-E should be considered.
Credit authorship contribution statement- Cristiane B. Netto: conception, design of the study, acquisition of data, analysis, interpretation of data, literature search, drafting the article, final approval of the version to be published.
- Maycon M. Reboredo: conception and design of the study, analysis and interpretation of data, literature search, drafting the article, final approval of the version to be published.
- Rodrigo S. Vieira: conception and design of the study, acquisition of data, analysis and interpretation of data, literature search, drafting the article, final approval of the version to be published.
- Lídia M. C. Fonseca: conception and design of the study, analysis and interpretation of data, revising the article providing intellectual content of critical importance to the work described, final approval of the version to be published.
- Elivaldo Cyrillo Junior: acquisition of data, analysis and interpretation of data, revising the article providing intellectual content of critical importance to the work described, final approval of the version to be published.
- André L. D. Souza: acquisition of data, analysis and interpretation of data, revising the article providing intellectual content of critical importance to the work described, final approval of the version to be published.
- Erich V. Carvalho: conception and design of the study, analysis and interpretation of data, drafting the article, final approval of the version to be published.
- Bruno V. Pinheiro: conception and design of the study, analysis and interpretation of data, drafting the article, final approval of the version to be published.
FundingThis study was supported by a National Council for Scientific and Technological Development – CNPq.
None.