The first wave of the COVID-19 pandemic challenged health systems worldwide. Its high capacity to generate critical patients required rapid expansion of beds beyond the classical intensive care unit (ICU) to other spaces termed “expanded ICU (eICU)”, as well as repurposing and redistribution of resources, personnel from other areas and specialised personnel. This new situation made it more difficult to comply with the recommendations for prevention of health care-associated infections (HAIs) what added to the higher exposition of patients to immunosuppressive treatments or requiring long ICU stays, made them more susceptible to HAIs. Several studies have shown higher rates of HAIs in COVID-19, but none of them as a principal factor in critically ill patients.1–8
Our aims were to determine the rates, aetiology, risk factors and impact of HAIs diagnosed in critically ill patients with COVID-19 pneumonia admitted to the different COVID-19 ICUs. It is a retrospective, observational, single-centre study. We evaluated all nosocomial bacteraemias (bloodstream infections or BSI), urinary tract (UTI), respiratory (VLRTI), pressure ulcer (IPU) and tracheostomy site infections (TSI) in patients with COVID-19 pneumonia admitted to one COVID-19 ICU (principal ICU or one of our 3 eICUs). The HAI incidence rates (IR) were analysed according to the patients’ location. Risk factors for HAI were identified from multivariate analysis. ICU length of stay (LOS) and mortality were analysed according to the number of HAIs (0, 1, 2, or ≥3). More details are shown in the online supplemental material.
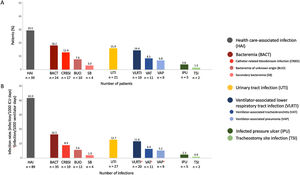
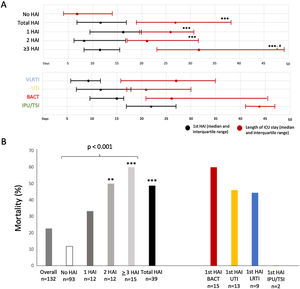
A total of 158 patients were admitted to the ICUs and 132 (83.5%) were included (Figure 1e). Transfer between the different ICUs are shown in Figure 2e. Eighty-nine HAIs (IR 42.0episodes/1000d) were identified in 39 patients (29.5%). The most common HAIs were BSI (IR 16.5), UTI (IR 12.7) and VLRTI (IR 11.6) (Fig. 1). The original ICU had the lowest rates of HAI (Figure 3e). The distribution of different HAIs over time and in the different ICUs is shown in Figure 4Ae and 4Be. Characteristics of patients according to HAIs are shown in Table 1e. Only renal replacement therapy (RRT, OR 8.74 [1.40–54.49]) and length of ICU stay (OR 1.11 [1.03–1.21]) were independently associated with HAI. One-hundred and fourteen microorganisms were identified, the most common being P. aeruginosa (18.4%), enterococci (17.5%) and Candida spp (25.0%) (Figure 5e and Table 2e). Of all the causative organisms isolated, 18.4% were MDR (Table 3e). The distribution of the microorganisms over time for each of the HAIs in each unit is shown in Figure 4Ce. Among patients with HAI while in the ICU, 69.2% had more than one (Figure 6e) and the ICU LOS and mortality showed statistically significant differences according to the number of HAIs present (Fig. 2).
Prevalence (A) and rates (B) of the different types of HAI in patients with pneumonia secondary to COVID-19 admitted to the COVID-19 critical care area.
HAI: health-care associated infection; BACT: bacteraemia; CRBSI: catheter-related bloodstream infection; BUO: bacteraemia of unknown origin; SB: secondary bacteraemia; UTI: urinary tract infection; VLRTI: ventilator-associated lower respiratory tract infection; VAT: ventilator-associated tracheobronchitis; VAP: ventilator-associated pneumonia; IPU: infected pressure ulcer; TSI: tracheostomy site infection.
There are multiple reasons that could explain the increase in HAIs in the population with COVID-19 admitted to ICUs. Structural reasons (opening of new ICU beds in other spaces in the hospital or eICU), organisational reasons (incorporation of new teams of doctors and nurses not previously trained in critical care) and functional reasons (changes in the standards of patient care, use of PPI during long shifts) may all play a role and make it difficult to adhere to the recommendations proposed by different scientific societies for HAI prevention.9 These aspects of care and organisation varied in the different units of the COVID ICUs and would explain the differences between them in the rates and time until onset of HAI. In addition, the need for an eICU itself meant increased transfer of patients between ICUs, which could have predisposed the development of HAI. Finally, individual patient susceptibility to infection could also have played a role in those patients with more than one HAI.
Characteristics of patients with COVID-19 who developed HAI, were similar to those published in other series of patients requiring ICU admission.10–12 Their profile was that of older age, with increased illness severity, a history of stroke and/or diabetes, need for invasive devices or proning and RRT. On tests from the first 24h in ICU, no differences were observed in markers of immune response, inflammation or altered clotting, which indicates that in the onset of HAI, exposure to external risk factors are more relevant. RRT was identified as an individual risk factor for HAI. This could be related to the need for more than one venous catheter and the manipulations involved in placing and managing them. Among 16 patients who required RRT, 10 patients (62.5%) had at least one episode of catheter related BSI (CRBSI) or bacteriemia of unknown origin (12 episodes in total). BSI post vascular catheter placement in patients that require RRT may indicate an area of quality improvement at the time of catheter insertion and subsequent care.
Among the microorganisms identified as causing HAIs, many were part of the primary and secondary endogenous microflora (species of Candida, Enterococcus and enterobacterales, in particular K. pneumoniae) while those associated with cross transmission or reservoirs were less common. Endogenous flora was particularly common in UTIs and primary bacteraemias, suggesting spread from the gut flora. In contrast, P. aeruginosa, the most prevalent pathogen in our hospital for colonisation or nosocomial infection was associated with cross transmission particularly in patients who developed VLRTIs. Surprisingly, the presence of MDR pathogens was low and heterogeneous, and there were no identified outbreaks.
Total ICU LOS conditioned the development of HAIs. Patients who developed one or more HAIs had longer total ICU LOS, although in most cases ICU LOS prior to HAI and total ICU LOS in patients without HAI were similar. Presence of HAI and total number of HAIs per patient were associated with higher ICU mortality. Unfortunately, 20% of patients who died in the ICU had a diagnosis of HAI within 72h of death.
In conclusion, the high rates of HAI, the differences observed between the main ICU and the three expanded units, the movement of patients between ICUs and the significant clinical impact and use of resources indicate that we must plan specific interventions to minimise such patterns in future pandemics and epidemics.