Edited by: Juan Antonio Llompart-Pou - Specialist in Intensive Care Medicine, Doctorate in Health Sciences, Universitat Illes Balears. Department of Intensive Care Medicine, Trauma and Neurocritical ICU, Hospital Universitari Son Espases. Palma, Spain
Last update: November 2025
More infoCritical care management of aneurysmal subarachnoid hemorrhage (aSAH) remains a major challenge. Despite the recent publication of guidelines from the American Heart Association/American Stroke Association and the Neurocritical Care Society, there are many controversial questions in the intensive care unit (ICU) management of this population. The authors provide an analysis of common issues in the ICU and provide guidance on the daily management of this specific population of neurocritical care patients.
El manejo en la unidad de cuidados intensivos (UCI) de los pacientes con hemorragia subaracnoidea aneurismática continua siendo un reto. A pesar de la publicación de las guías de la American Heart Association/American Stroke Association y la Neurocritical Care Society todavía existen muchos aspectos controvertidos en el manejo de esta población en la UCI. Los autores proporcionan un detenido análisis de los problemas habituales en la UCI y proporcionan recomendaciones en el manejo diario de esta población específica de pacientes neurocríticos.
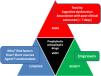
Critical care management of aneurysmal subarachnoid hemorrhage (aSAH) remains a major challenge worldwide.1,2 The lack of class I recommendations and the evolving knowledge of early brain injury3 and delayed cerebral ischemia (DCI)4 faces neurointensivists with uncertainity, despite recently published guidelines by the American Heart Association/American Stroke Association (AHA/ASA)5 and the Neurocritical Care Society (NCS).6 Whilst the AHA/ASA guidelines cover a comprehensive approach from the natural history and clinical presentation to prevention of recurrence,5 the NCS guideline focused on the critical care management.6 However, they used an approach using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) framework limited to 12 management questions that could probably be most impacted by new evidence and evolving management paradigms in the last decade.6 Thus, NCS guidelines were developed around specific clinical questions in a population, intervention, comparison, and outcomes (PICO) format,6,7 resulting mostly in non-conclusive recommendations in a daily basis for the neurointensivist. Thus, we provide a comprehensive review of controversial topics in the intensive care unit (ICU) management of aSAH (Fig. 1).
Which is the optimal blood pressure before coilling and clipping?The exact role of blood pressure (BP) management to prevent early rebleeding before aneurysm securing remains to be determined yet. The NCS guidelines state that there is insufficient evidence to recommend a BP reduction goal for the treatment of hypertension before aneurysm treatment in aSAH patients, since the quality of available evidence was too low to support the recommendation of a target for BP reduction vs no BP reduction.6 However, NCS guidelines specifically state that “Lack of evidence to recommend a specific blood pressure reduction goal does not necessarily imply that blood pressure reduction is not helpful before aneurysm treatment”.6 On the other hand, the AHA/ASA guidelines state that in patients with aSAH and unsecured aneurysm, frequent BP monitoring and BP control with shortacting medications is recommended to avoid severe hypotension, hypertension, and BP variability.5
The rationale behind these recommendations relies on the low quality of studies in this setting. Retrospective studies8 and meta-analysis9 addressed a higher risk of rebleeding in patients with systolic blood pressure (SBP) >160 mmHg, but an aggressive approach has not been associated with reduced reebleeding.10 Additionally, systemic hypotension is linked to compromised cerebral perfusion and can be associated with DCI11 and BP variability could be also associated with rebleeding.12 Taking these data into consideration, it can only be recommended that extreme values and marked variability in BP must be avoided. In the authors experience, in initially hypertensive aSAH patients, a target of SBP 140−160 mmHg avoiding fluctuations seems reasonable.
Anyway, a RCT is clearly required to provide the necessary evidence to define the balance of potential benefits and risks of BP lowering in the acute phase of SAH, since it could be a promising strategy to improve outcomes and could be initiated during the transport of patients by ambulance to an appropriate facility.13
What is the optimal timming for coiling or clipping?Due to the high risk of poor outcome and mortality after aneurism rebleeding, ruptured aneurysms should be secured as soon as possible.5 Timing of the ruptured aneurysm treatment was directly examined in one randomized trial on good-grade patients, which demonstrated that early surgery (up to 3 days after SAH) was related to lower death and dependence at three months.14 This data has been corroborated in other types of studies in which early securing of the aneurysm (<24 h) produced better outcomes,15–17 especially in the endovascular treatment group.17 However, these data are not unequivocal.18
Indeed, from a pathophysiological rationale and considering that rebleeding is especially relevant in poor grade aSAH within 0–12 hours from stroke 19, inmediate (<6 h) clipping/coiling strategies appear reasonable but its benefit has to be proven yet.
From one side it has been calculated that implementing such an scheme of ultra-early management would only provide treatment for a very limited number of possible rebleedings, making a modest reduction in the rebleeding rates (around 0.3% of reduction of incidence of rebleeding).20 On the other hand, this would impose significant stress to the teams, determining that many procedures would be performed by less experienced teams and with worse equipment. Therefore, since experience is relevant in final outcomes, most centers (and the authors) accept to perform the treatment as soon as possible by the team that usually treats aneurysms by clipping or by coiling, preferably in the first 24 h. Best results are obtained when both treatments can be delivered. However when there is a ruptured aneurysm producing a large cerebral hematoma, urgent surgery accompanied with surgical clipping of the aneurysm has demonstrated a large reduction in mortality and a higher rate of independent outcome.21 Although it is feasible to first coil and then evacuating the hematoma, it seems more logical not to delay hematoma evacuation and perform concomitant aneurysm clipping.
Which is the optimal blood pressure target after securing the aneurysm?There is a paucity of studies addressing this topic, making it an appealing area for planning multicenter studies and therefore, improve outcomes.22 In consequence, there is a huge variability in commonly used targets and indeed, there is no consensus even in using mean arterial pressure (MAP) or SBP targets.23
In the MANTRA survey, when the aneurysm was secured 34.9% of the responders considered MAP targets (median (IQR) 90 (75–100) mmHg, 16.8% used SBP targets (median (IQR) 155 (140–180) mmHg, 33.2 % used both MAP and SBP (median (IQR) 90 (80–100) and median (IQR) 160 (140–180) mmHg, respectively) and 15.1% allowed spontaneous hypertension.23
The body of the literature is not enough to consider an optimal target,5 so the dichotomy between hypoperfusion in low BP states and the potential side effects of hypertension must be outweighted. Based in their own experience, the authors recommend a wide target between SBP 120−180 mmHg and MBP 80−120 mmHg, with higher targets used in symptomatic patients. Different clinical factors must be taken into consideration too, including age, prior chronic hypertension, the autoregulation status and the occurrence of intracranial hypertension and/or DCI symptoms. These groups of patients will be usually managed in the upper range proposed. Future studies must address this question.13
Is there a role for intravenous nimodipine?Nimodipine is a dihydropyridine calcium channel antagonist that blocks the flux of extracellular calcium through L-type, voltage-gated calcium channels. It is approved for the prevention and treatment of neurological deficits in patients with aSAH, but since it affects myriad cell types throughout the body, it likely has more complex mechanisms of action than the simple inhibition of cerebral vasoconstriction.24 This is in line with current pathophysiological approaches, which consider that not only vasospasm but more complex and multifactorial mechanisms drive to DCI resulting in cerebral infarctions and poor neurological outcomes.25
In 1983, Allen et al. conducted a prospective, double-blind, randomized, placebo-controlled trial including 125 patients with aSAH within 96 h of stroke, to determine whether treatment with the calcium blocker nimodipine given orally would prevent or reduce the severity of ischemic neurologic deficits from arterial spasm. A deficit from cerebral arterial spasm that persisted and was severe or caused death by the end of the 21-day treatment period occurred in 8 of 60 patients in the placebo group and in 1 of 56 of the oral nimodipine group (P = 0.03, Fisher's exact test).26 Oral/enteral nimodipine as prophylaxis of cerebral vasospasm was then implemented in the management of aSAH worldwide. However, the potential beneficial results on outcomes were limited to the oral/enteral administration and the number of patients needed to treat was 19 for the oral/enteral formulation.27 Intravenous administration of calcium antagonists could not be recommended for routine practice on the basis of the evidence summarized in a systematic review in 2007.27
Indeed, the AHA/ASA guidelines currently recommend the use of oral/enteral nimodipine 60 mg q 4 h during 21 days (class I, level of evidence A) to improve neurological outcomes.5 Moreover, the NCS guidelines specifically state that other routes for their administration are not supported by sufficient evidence.6
On the other hand, some recent studies have shown that iv nimodipine is non-inferior to oral/enteral nimodipine in terms of improving outcomes. A recent meta-analysis including 10 studies and 1527 patients showed that oral/enteral and intravenous nimodipine were both effective in preventing poor outcome, DCI, and delayed ischemic neurological deficit. Neither treatment was effective in improving case fatality.28
At this point, and while awaiting a more solid evidence, the authors strongly advocate for the oral/enteral administration of nimodipine and avoidance of the intravenous formulation, since the intravenous use has been associated with more frequent episodes of hypotension,29–31 which is a major determinant of poor outcomes.
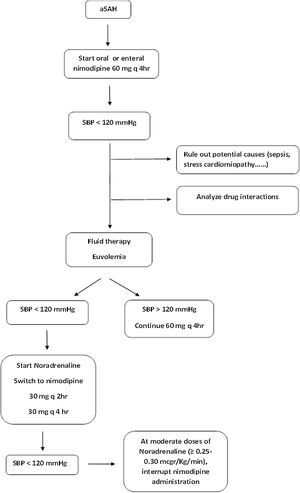
What to do in cases with oral/enteral nimodipine-induced hypotension?As stated, the AHA/ASA guidelines recommend the use of oral/enteral nimodipine 60 mg q 4 h during 21 days (class I, level of evidence A) to improve neurological outcomes.5 In our environment, the quality indicators of the Spanish Society of Intensive Care Medicine (SEMICYUC) consider a standard its use within 12 h of stroke.32
Given in the oral or enteral form, hypotension affects one out of ten patients compared with one out of 3 patients when using the iv formulation.31 Additionally, different observational studies have addressed that administering the full dose of oral/enteral nimodipine during 21 days is achieved in less than half of the cases, ranging from 33 to 57%,33–35 being arterial hypotension the most common underlying reason.33–35 Hypotension was 3 times more common when nimodipine was administered as an oral/enteral solution compared to given as tablets, despite no difference in plasma levels as measured by liquid chromatography-tandem mass spectrometry.34 The main factor associated with oral/enteral nimodipine administration interruption was poor neurological grade.33,34 In this context, dose reduction, shortening of the interval of administration or even discontinuation of the treatment are commonly used strategies.33–35
However, to the best of our knowledge, there are no studies specifically addressing this topic nor evidence to support a specific strategy, so the optimal management of oral/enteral nimodipine therapy (dose splitting, reduction, or withholding) in patients unable to tolerate the hemodynamic side effects or who are receiving vasopressors for BP augmentation for treatment of DCI remains unknown.6 In this point, we provide a guidance algorithm based in the authors experience (Fig. 2).
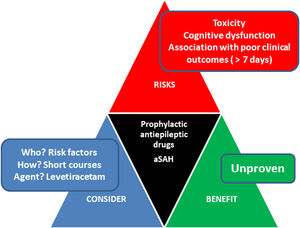
Is there a role for prophylactic antiepileptic drugs?Seizures are a well-known complication following aSAH.25 Underlying mechanisms are incompletely understood but might include neuroinflammation and cerebral hyperemia.25 The pathophysiological pathway to impact outcomes remains unclear.25
Incidence ranges from 0 to 31%, being more frequent in the early phase.2 Late seizures and convulsive status are less frequent.5 Risk factors to present early seizures include middle cerebral artery aneurysms, high clinical/radiological grade (Hunt Hess grade >3 or Fisher grade III/IV), cortical infarction, or hydrocephalus.5 In this context, before aneurysm treatment, short-term seizure prophylaxis with antiepileptic drugs (AEDs) might be considered given the fear of rebleeding,5 but administration beyond this very early phase is highly controversial. Long-term epilepsy is infrequent but associated with worse outcomes25 and the impact of prophylaxis in clinical outcomes remains to be determined yet.5
A recent metaanalysis including a total of 5 studies concluded that long-term exposure to prophylactic AEDs (more than 3 days) led to poor clinical outcomes (OR 1.55; 95% CI 1.01–2.39; p = 0.045) (959 patients, 3 studies) and that no association between the duration of prophylactic AEDs use and the occurrence of in-hospital seizures (OR 0.62; 95% CI 0.18–2.15; p = 0.447) was found (1024 patients, 4 studies).36 The definition of a short course ranges from 3 to 7 days from aneurysm securing. Whilst further prospective research comparing the effectiveness of different AEDs in patients with aSAH is needed, levetiracetam seems better tolerated that phenytoin.37 Phenytoin has been also linked to poorer clinical outcomes.5
Recently published AHA/ASA guidelines state that in patients with SAH and high-seizure risk features, the use of prophylactic antiseizure medications may be reasonable to prevent seizures.5 Recent meta-analyses suggest that compared with short-term use (<3 days), the long-term use (>3 days) of prophylactic antiseizure medications in patients with aSAH has a similar effect on in-hospital seizure prevention but is associated with poor clinical outcomes.
Traslating these data into the clinical setting, the neurointensivist must balance the risk and benefits of using short courses of AEDs (Fig. 3). Thus, in patients presenting without seizures and with risk factors, we only recommend considering AEDs for a maximum of 3 days in those cases with fluctuating or depressed consciousness (usually levetiracetam 1000 mg q 12 h). In patients presenting with seizures treatment with levetiracetam for a maximum of 7 days (usually levetiracetam 1000 mg q 12 h following a loading dose 40−60 mg/Kg) is indicated, although these recommendations lack of supporting randomized controlled trials.
Is there a role for routine use of antifibrinolytics?Rebleeding before aneurysm obliteration is an important cause of death and disability in patients with aSAH, probably related to the dissolution of the blood clot at the site of the aneurysm rupture by natural fibrinolytic activity. This historically led to investigations on the use of antifibrinolytics (aminocaproic and tranexamic acid) in patients with aSAH. Preliminary studies showed a reduced risk of rebleeding, minor effects on complications (specially when using short courses) and no clear effects on outcomes.38,39 More recently, the Ultra-early tranexamic acid after subarachnoid haemorrhage (ULTRA) trial showed that using 1 g bolus followed by 1 g q 8 h terminated immediately before aneurysm treatment, or 24 h after start of the medication, was associated with a non-significant reduction of rebleeding (even in the setting of high rebleeding rates) with no effects on outcomes (as evaluated by using the modified Rankin Scale).40 More recently, a systematic review and meta-analysis performed by the Cochrane Database of Systematic Reviews including all randomized controlled trials comparing oral or intravenous antifibrinolytic drugs (tranexamic acid, epsilon amino-caproic acid, or an equivalent) found that the rate of rebleeding was decreased but with no impact on DCI, hydrocephalus or outcomes.41 In the authors opinion, this settles the question whether a routine use of antifibrinolytics is indicated in patients with aSAH, especially when strategies include early aneurysm treatment (<24 h). Antifibrinolytics must not be routinely used. In this setting, Rabinstein suggests that only in patients presenting to the emergency department very early, a single dose of tranexamic acid might still be considered, particularly if transfer to another centre is necessary for aneurysm treatment.42 However, a potential benefit of this strategy has to be proven yet.
Is transcranial sonography useful for monitoring vasospasm-induced DCI?Cerebral vasospasm is one of the components that may lead to DCI. Monitoring of cerebral vasospasm is troublesome, especially in patients under sedative agents. In our environment, the use of transcranial sonography to detect arterial narrowing reflected by an increase in the velocity of cerebral blood flow is common,43 especially in the proximal segment of the middle cerebral artery (MCA). A recent meta-analysis showed a sensitivity, specificity, positive predictive value and negative value of 66.7%, 89.5%, 93.7% and 53.4% for transcranial doppler and 81.5%, 96.6%, 98.2% y 69.1% for transcranial color-coded duplex sonography in the MCA.44 Taken together, these data suggest that transcranial sonography can be useful to detect severe arterial narrowing, but it is not useful to exclude it. In addition, it has to be considered that commonly used criteria are variable, including the measurements of static values (alone or augmented by the use of the Lindegaard index in the MCA and the Sviri ratio in the basilar artery), the use of the arterio-venous index or the increase of the velocities in the first days ater aSAH, which could be the best indicator of arterial narrowing.45 It has to be noted that the evidence suporting the role of transcranial sonography in the anterior and posterior cerebral arteries remains very limited.45 Despite these considerations, transcranial sonography remains the most commonly used method for the diagnosis and monitoring of DCI in unconscious patients.23 In the authors experience, the increase of mean velocities in daily evaluations during the initial 7 days after hemorrhage constitutes the best indicator of arterial narrowing.
Another controversial point is to determine which is the role of transcranial sonography in the neurological outcomes of patients with aSAH. A recent population-based study using data from the United Kingdom and Ireland Subarachnoid Haemorrhage (UKISAH) Registry including 2029 patients treated ≤3 days of hemorrhage showed that centers who screened for vasospasm using transcranial sonography had poorer in-hospital outcomes and similar rates of DCI diagnosis compared to centers that did not.46 In addition, the National Institute for Health and Care Excellence (NICE) guidelines especifically urge to “Do not use transcranial doppler monitoring to guide clinical management of an aneurysmal subarachnoid hemorrhage except in the context of clinical research”.47 These data suggest a cautious and judicious use of transcranial sonography to rule out cerebral vasospasm is mandatory, always by experienced hands in the technique and in the aSAH pathophysiology.
Is CT angiography useful for monitoring vasospasm-induced DCI?Initial studies showed that computed tomography angiography (CT angiography) was a reliable, minimally invasive imaging modality for the detection and assessment of the severity of vasospasm following aSAH.48–50 Its widely acknowledged limitation was the inability to measure the diameter of some arteries due to metallic artifacts.50
This led to an increased use of the technique. However, as occurs in the case of transcranial sonography, CT angiography has elevated specifity but poor sensitivity to identify arterial narrowing, making its use unfeasible as a screening test.51 Additional concerns include a relatively low repeatability among inter- and intra-observers52,53 and differences in the definitions used.52,53
In any case, its role in the monitoring of vasospasm is widely dependent of institutional protocols. Recently, Van del Harst et al.54 compared transcranial sonography and CT angiography in 59 patients with aSAH. The agreement between transcranial Doppler and CT angiography was only 0.47. The diagnostic accuracy of both CT angiography and transcranial Doppler for detection of cerebral vasospasm as well as for predicting DCI and functional outcome was limited, being the highest accuracy for predicting unfavorable outcome on the 5th day after bleeding 0.61 for transcranial Doppler vs 0.27 for CT angiography. However, according to a recent survey, CT angiography is still the imaging modality most used to confirm the presence of cerebral vasospasm as the cause of DCI.23
CT perfusion (CTP) is another imaging modality used in the diagnosis of DCI and vasospasm following aSAH. It may be particularly helpful when clinical examination is limited and/or sonographic windows are poor. There is no clear consensus of diagnostic criteria for vasospasm via CTP, but using an increased mean transient time (>6.0 s) and decreased cerebral blood flow (<30%) has shown correlation with vasospasm.55 However, thresholds for abnormal limits of cerebral blood volume, cerebral blood flow, mean transit time (MTT), and Tmax have not been firmly validated with large patient samples.56
Blood pressure augmentation or inotropic support in DCI management?The lack of high-quality studies remains a major limitation when evaluating hemodynamic support in patients at high-risk or developing DCI. Indeed, existing guidelines made different recommendations with variable degrees of evidence on this topic.22 There is a clear consensus on maintaning euvolemia,22 but the use of vasoactive agents or inotropes remains controversial.
On one hand, the NCS guidelines state that there are insufficient quality data to recommend for or against BP or cardiac output augmentation for the prevention and treatment of DCI and recommend that, due to the associated risks, the use of these interventions should be judicious and tailored to the patient’s individual hemodynamic profile.6 On the other hand, the AHA/ASA guidelines state that in patients with aSAH and symptomatic vasospasm, elevating SBP values may be reasonable to reduce the progression and severity of DCI,5 despite the only randomized controlled trial evaluating induced hypertension in aSAH patients was inconclusive and discontinued prematurely.57 This trial, aiming at recruiting 240 aSAH patients was prematurely ended, based on lack of effect on cerebral perfusion and slow recruitment when 21 patients had been randomized to induced hypertension and 20 patients to no hypertension. With induced hypertension, the adjusted risk ratio for poor outcome was 1.0 (95% confidence interval, 0.6–1.8) and the risk ratio for serious adverse events 2.1 (95% confidence interval, 0.9–5.0).57 Therefore, such recommendation is mainly based on observational data coming from large, multicenter studies indicating an improvement after induced hypertension in 80% of symptomatic patients.58 Indeed, according to the MANTRA survey, induced arterial hypertension was the first measure taken by 89.2% of the responders using noradrenaline in 97.7% of the cases.23
Data on the use of inotropes in this setting are limited too. In a pilot study finally evaluating 35 patients, Rondeau et al. compared norepinephrine-induced hypertension with dobutamine-induced augmentation in cardiac index and found no difference in angiographic vasospasm between the groups.59 Lakhal et al., analyzed the use of milrinone in a controlled observational study conducted in an academic hospital with prospectively and retrospectively collected data. Consecutive patients with cerebral vasospasm following aSAH and treated with both IV milrinone and induced hypertension were compared with a historical control group receiving hypertension alone. Despite its premature discontinuation in 29% of patients as a result of its poor tolerance, IV milrinone was associated with a lower rate of endovascular angioplasty and a positive impact on long-term neurological and radiological outcomes,60 opening the window to well-designed studies in the field. In this direction, Mutoh et al. compared ealy-goal directed therapy (EGDT) guided by preload volume and cardiac output using transpulmonary thermodilution with patients receiving standard treatment guided by fluid balance or central venous pressure, assisted by minimally invasive cardiac output monitoring.61 The study was performed in two Japanese centers. In the EGDT group, DCI was treated with dobutamine or milrinone to achieve a global end-diastolic volumen index (GEDI) 800 tp 900 ml/m2 and the cardiac index to 4 L/min/m2 or clinical improvement. No diferences in DCI rates were found in the whole group, but when applied to patients with poor-grade aSAH the authors found a lower incidence of DCI in the EGDT group (5% vs 14%; P = 0.036) and a higher frequency of favorable functional 3-month outcomes (52% vs 36%; P = 0.026). In addition, in the small subset of patients with coexisting cardiopulonary complications those who received EGDT had a better functional 3-month outcome (63% vs 38%; P = 0.045) without differences in DCI rates.61 More recently, Anestberger et al. published the results of an unblinded, single-center study including 108 aSAH patients.62 In the goal directed group, and using a protocol that included the following hemodynamic targets (GEDI > 640 ml/m2; extravascular lung water index <10 ml/Kg, MAP > 100 mmHg in cases with vasospasm and cardiac index 2.5 L/min/m2) found that the rate of DCI was lower in the goal-directed therapy group (13% vs 32%; p = 0.021).62 More scarce is the experience using levosimendam, limited to case reports or retrospective case control studies63 and with an anecdotal use in our environment.23
Taken together, close hemodynamic monitoring and directed therapy could be beneficial in the prevention of DCI and functional outcomes, specially in poor-grade aSAH9,10,61 and in aSAH patients with associated cardiac dysfunction.61 Due to the lack of multicenter studies and the different criteria used, the optimal hemodynamic targets remain to be determined.
Can we start antiplatelets in patients with external ventricular drainage?The increasing use of stents in aSAH patients has improved the feasibility of the endovascular treatment of wide neck and blister-like aneurysms.64 However, the use of stents or stent assisted coiling determines the need to use antiplatelet treatment to avoid platelet aggregation in the stent that could cause in-stent thrombosis and embolic stroke. The use of dual antiplatelet treatment in the acute phase of the management of these patients is controversial due to the frequent need of interventions related to the treatment of acute hydrocephalus by means of external ventricular drainage (EVD).65 Different reports shown that EVD-related hemorrhage risk increases in patients treated with stents and can be as high as nearly 30% of the cases,66–69 but in most cases, secondary hemorrhages are not symptomatic nor life threatening. Therefore, the percentage of major hemorrhages seems less than 5% while being on antiplatelets.66–69 To reduce this potential complication, most authors support that when stenting is planned for the treatment of the ruptured aneurysm, EVD should be put in place before the endovascular procedure.68 This reduces significantly the risk of hemorrhage. However, hydrocephalus not always occurs in the ultra early stage and it can appear or be evident after the treatment of the aneurysm. On the other hand, once the EVD is set in place, the following issue is removing it while being on antiplatelets. Most authors support the need of stopping temporarily treatment with antiplatelets and remove the EVD after platelet transfusion.68 Antiplatelet treatment can be then resumed after the removal of the EVD.
The continuous development of stents and stent assisted coiling techniques difficults a universal recommendation on antiplatelets management in patients with aSAH treated with EVD. An adjusted and guided management may impact bleeding events,70,71 but its effect of clinical outcomes has to be proven.
How and when can the ventricular drainage be safely removed?EVD for the treatment of acute hydrocephalus after SAH is a common procedure as nearly as 30% present this acute complication due to blockage of CSF circulation. However, up to nearly 40% of patients with an EVD will develop chronic hydrocephalus, requiring the insertion of a definitive ventriculoperitoneal (VP) shunt.72 There is still discussion on the timing of removal of the EVD, and also on the way of managing this removal, prompt closure/clamping or gradual weaning.73–75 Regarding the timing, most physicians will defer the discontinuation 4–7 days after the ictus if the patient is in a stable condition.74 Discontinuation will be delayed if there is suspicion of vasospasm.74 The volume of CSF drained/24 h has been also related to the need of a definitive VP shunt76,77 and most physicians would not close the drainage if the volume of CSF drained is more than 150 ml/day.74 Some studies have shown that acute clamping trials are related to reduced ICU and in-hospital length of stay and maybe less EVD-related infections.75,78 Both the AHA/ASA5 and the NCS6 guidelines state that there can be no clear recommendations favoring one method over another attending to the lack of well-designed randomized controlled trials, despite secondary outcomes, such as ICU or in-hospital legth of stay, are improved with direct clamping.
In clinical practice and using one or other method, EVD closure trials are usually stopped and the drain reopened if there is clinical or radiological (increase in the size of the ventricles in CT scan) deterioration, ICP rise or peri-EVD CSF leakage. Repeated failure in closure or weaning trials are related to definitive VP shunt placement and, therefore, after a patient has failed to progress after two or three trials it would be safer to set a definitive VP shunt.74
LimitationsThis manuscript presents some limitations inherent to its nature. This manuscript did not intend to be a systematic nor a scoping review, but a narrative review aiming to discuss common controversials questions in the ICU management of aSAH, based in a critical analysis from the authors. After a careful in-depth analysis, recommendations are summarized in Fig. 4.
ConclusionsIn this practical review, we have discussed common controversies in the ICU management of patients with aSAH. From their point of view, the authors provided guidance on the daily management of this specific population of neurocritical care patients.
Conflict of interestsThe authors declare no conflict of interest related to this manuscript.
FundingNone.