To evaluate the clinical characteristics, prevalence and factors associated with delirium in critical patients from 5 to 14 years of age.
DesignAn analytical, cross-sectional observational study was made. Delirium was assessed with the Pediatric-Confusion Assessment Method for the Intensive Care Unit (pCAM-ICU) and motor classification was established with the Delirium Rating Scale Revised-98.
SettingA pediatric Intensive Care Unit.
PatientsAll those admitted over a one-year period were assessed during the first 24–72h, or when possible in deeply sedated patients. Exclusion criteria: Patients in stupor or coma, with severe communication difficulty, subjected to deep sedation throughout admission, and those with denied consent.
ResultsTwenty-nine of the 156 assessed patients suffered delirium (18.6%) and 55.2% were hypoactive. The neurocognitive alterations evaluated by the pCAM-ICU were similar in the three motor groups. Intellectual disability (OR=17.54; 95%CI: 3.23–95.19), mechanical ventilation (OR=18.80; 95%CI: 4.29–82.28), liver failure (OR=54.88; 95%CI: 4.27–705.33), neurological disease (OR=4.41; 95%CI: 1.23–15.83), anticholinergic drug use (OR=3.23; 95%CI: 1.02–10.26), different psychotropic agents (OR=4.88; 95%CI: 1.42–16.73) and tachycardia (OR=4.74; 95%CI: 1.21–18.51) were associated to delirium according to the logistic regression analysis.
ConclusionThe frequency of delirium and hypoactivity was high. It is therefore necessary to routinely evaluate patients with standardized instruments. All patients presented with important neurocognitive alterations. Several factors related with the physiopathology of delirium were associated to the diagnosis; some of them are modifiable through the rationalization of medical care.
Evaluar las características clínicas, la prevalencia y los factores asociados al delirium en pacientes de entre 5 y 14 años de edad y en estado crítico.
DiseñoEstudio observacional analítico transversal. El delirium se evalúo con el Pediatric-Confusion Assessment Method for the Intensive Care Unit (pCAM-ICU) y la clasificación motora con la Delirium Rating Scale Revised-98.
ÁmbitoUnidad de cuidados intensivos pediátricos.
PacientesTodos los que ingresaron durante un año fueron evaluados durante las primeras 24–72h o cuando fue posible si estaban en sedación profunda. Excluidos: pacientes en estupor o coma, con dificultad grave para la comunicación, en sedación profunda durante todo el ingreso y los que no contaban con consentimiento.
ResultadosVeintinueve (18,6%) de los 156 pacientes evaluados tenían delirium y el 55,2% eran hipoactivos. Las alteraciones neurocognitivas evaluadas por el pCAM-ICU fueron similares en los 3 grupos motores. La discapacidad intelectual (OR=17,54; IC95%: 3,23-95,19), la ventilación mecánica (OR=18,80; IC95%: 4,29-82,28), el fallo hepático (OR=54,88; IC95%: 4,27-705,33), las enfermedades neurológicas (OR=4,41; IC95%: 1,23-15,83), el uso de anticolinérgicos (OR=3,23; IC95%: 1,02-10,26), diversos tipos de psicotrópicos (OR=4,88; IC95%: 1,42-16,73) y la taquicardia (OR=4,74; IC95%: 1,21-18,51) se asociaron al delirium según el análisis logístico.
ConclusiónLa frecuencia de delirium y del tipo hipoactivo es alta, por lo que es necesario evaluar rutinariamente los pacientes con instrumentos estandarizados. Todos los pacientes presentan alteración neurocognitiva importante. Varios factores relacionados con la fisiopatología del delirium se asocian al diagnóstico, algunos de los cuales son modificables mediante la racionalización de la atención médica.
Delirium is a neuropsychiatric syndrome of acute or subacute onset and with fluctuating symptoms.1 Based on investigation of the delirium phenotypes in adults, the syndrome is characterized by impaired mental functions manifesting when alertness is altered and three nuclear domains are affected: cognitive (attention, orientation, memory, visuospatial capacity); higher order thought (language, thought process, executive function); and circadian (sleep–waking, motor activity). Psychotic and affective symptoms are accessory.2,3 The disorder may have one or more medical or iatrogenic causes, and from the motor perspective it is defined as hyperactive, hypoactive, mixed or neither.4
Both the clinical characteristics of the syndrome and its risk factors have been little studied to date in the critically ill pediatric population, where it is often overlooked despite the increased morbidity–mortality it implies.5,6 In the Pediatric Intensive Care Unit (PICU), delirium prolongs admission and increases the treatment costs.7,8 Furthermore, it increases parent suffering and stress on the part of the treating professionals.9,10
The present study was designed to evaluate the prevalence and factors associated to delirium in the initial assessment of patients between 5 and 14 years of age admitted to a PICU over a period of one year. A standardized evaluation of delirium was made based on the Pediatric Confusion Assessment Method for the Intensive Care Unit (pCAM-ICU), as well as of its motor types, and a broad range of possible associated factors were explored. Likewise, we described the individual characteristics assessed by the items of the pCAM-ICU according to the motor type of the disorder.
Patients and methodsType of study and participantsIn order to evaluate the clinical characteristics, prevalence and factors associated to delirium in critically ill children, we designed a cross-sectional analytical observational study that was approved by the Ethics Committees of the Universidad Pontificia Bolivariana and Hospital Pablo Tobón Uribe (Medellin, Colombia). All the persons in charge of the patients, and the patients capable of doing so, gave written informed consent to participation in the study.
The patient evaluation period lasted one year. We included those patients between 5 and 14 years of age consecutively admitted to the PICU of Hospital Pablo Tobón Uribe – a high complexity university hospital. The PICU has 20 beds for children between 0 and 14 years of age. The Unit is general and open (encouraging the presence of the parents or care givers during admission of the patient).
We excluded patients with stupor or in coma, with communication problems precluding evaluation, receiving deep sedation throughout admission, and those cases in which informed consent could not be obtained.
InstrumentsMortality risk was assessed based on the Pediatric Risk of Mortality Score (PRISM).11 The properties of the neuropsychiatric scales employed are described below.
Pediatric Confusion Assessment Method for the Intensive Care Unit (pCAM-ICU)This instrument is an algorithm validated in children aged 5 years or older. It has excellent reliability (kappa=0.96) and validity in diagnosing delirium according to the DSM-IV-TR (sensitivity 83.0% and specificity 99.0%). The pCAM-ICU assesses the following: (a) the clinical course (possible acute changes versus baseline and/or fluctuation of the symptoms); (b) verbal or non-verbal attention according to the condition of the patient, based on the recognition of sequences of 10 items, where a score ≤7 indicates attention impairment; (c) alertness based on the Richmond Agitation Sedation Scale (RASS), which allows diagnosis of the disorder in the absence of deep sedation (score ≥4); and (d) the thought process, with 5 reasoning questions scored from 0 to 5, where a cut-off point ≤3 indicates alteration.12
The pCAM-ICU is positive for delirium if there are alterations in the first and second characteristic, and in either of the other two. In this study we used the version in Spanish,13 and for each motor type we recorded the frequency of acute onset, symptoms fluctuation and alteration of the RASS, as well as the median of the two clinical characteristics (attention and thought process).
Items of the Delirium Rating Scale – Revised-98 (DRS-R98) for the evaluation of motor functionThe Delirium Rating Scale – Revised-98 (DRS-R98) explores different clinical aspects of delirium, with response options ranging from 0 (normal) to 3 (maximum severity). Items #7 and #8 respectively evaluate increased and decreased motor activity, and can be combined to define mixed cases.14,15 In this study we only used the motor items. A score ≥1 in either of the two items defines hyperactive and hypoactive cases, respectively; cases with a score ≥1 in both items are mixed cases; and those scoring 0 in both items have no motor alterations.
ProceduresA pilot survey was carried out in which the study instruments were applied to 10 patients in order to standardize the procedures. The investigators held a meeting before the pilot survey, another meeting after the survey, and another two during the patient evaluation period, in order to maintain standardization.
The group conducting the field work consisted of four nurses, 5 psychiatrists and a pediatric intensivist. The patients of the study were evaluated during the first 24 and 72h of admission to the PICU. A psychiatrist and a nurse jointly compiled the sociodemographic and clinical data of the patients, applied the pCAM-ICU and classified the motor subtype of delirium with the DRS-R98.
Those patients administered deep sedation upon admission to the Unit (RASS≤3) were monitored daily until their condition allowed us to determine whether they suffered delirium (RASS≥3).
The main diagnoses leading to admission to the PICU were grouped according to the Delirium Etiology Checklist (DEC). The DEC allows the reporting of mutually excluding diagnostic categories that avoid data dispersion due to the low prevalences of all the individual medical conditions.16 This and the rest of the independent variables studied as possible risk factors related to delirium are defined in the section below.
Independent variables and statistical analysisWe defined a large group of factors for analysis, and controlled variables related to the study design. The independent variables were: age in years, gender, educational level, administration of deep sedation upon admission (yes/no, depending on whether the RASS score was ≤3), PRISM score, history of intellectual disability, epilepsy, vision problems or hearing problems, and each of the 6 most frequent main diagnoses upon admission according to the DEC. Furthermore, we determined whether any of the following characteristics had been recorded in the case history during the last 24–72h: postoperative condition, mechanical ventilation, respiratory failure, ineffective clearing of the airway, altered heart rate or blood pressure (tachycardia, bradycardia, hypertension or hypotension according to the criteria of the American Heart Association), hyperthermia (temperature≥38.0°C), hypothermia (temperature≤36.0°C), hypoxemia (PaO2<60mmHg), pain scored from 0 to 10 based on a visual analog scale (VAS), acidosis, electrolytic alterations, anemia, nutritional imbalances, neurological diseases of any kind, seizures, intoxication, heart, kidney or liver failure, metabolic disorders (e.g., blood glucose or thyroid alterations), infection, loss of skin integrity, constipation and restricted mobility. Lastly, we determined whether any of the following drug groups had been administered in the last 72h: analgesics, corticosteroids, opioids, antibiotics, anticholinergic agents, benzodiazepines, antipsychotics and – as a single variable – other psychotropic agents.
Discrete variables were reported as frequencies and percentages (%) and were compared using the chi-squared test (χ2). Continuous variables in turn were reported as the median and interquartile range (IQR) and were compared using the Mann–Whitney U-test (2 variables) or analysis of variance (ANOVA) with the Kruskal–Wallis test (more than 2 variables).
The analysis of the relationship between the independent variables and delirium was based on the evaluation of each individual variable (univariate analysis) and on the multivariate logistic analysis reported according to the recommendations of the literature.17 We calculated the odds ratio (OR) and 95% confidence interval (95%CI) of those variables found to be associated to delirium in the univariate analysis. The variables found to be significant were entered in the logistic regression model using the conditional backward approach (we chose the strict probability criterion of ≤0.05 as entry condition and >0.05 as exclusion criterion). Fitting of the model was evaluated with the Hosmer and Lemeshow test. We reported the Wald statistic with its p-value for each beta-coefficient, as well as the OR and 95%CI of each variable.
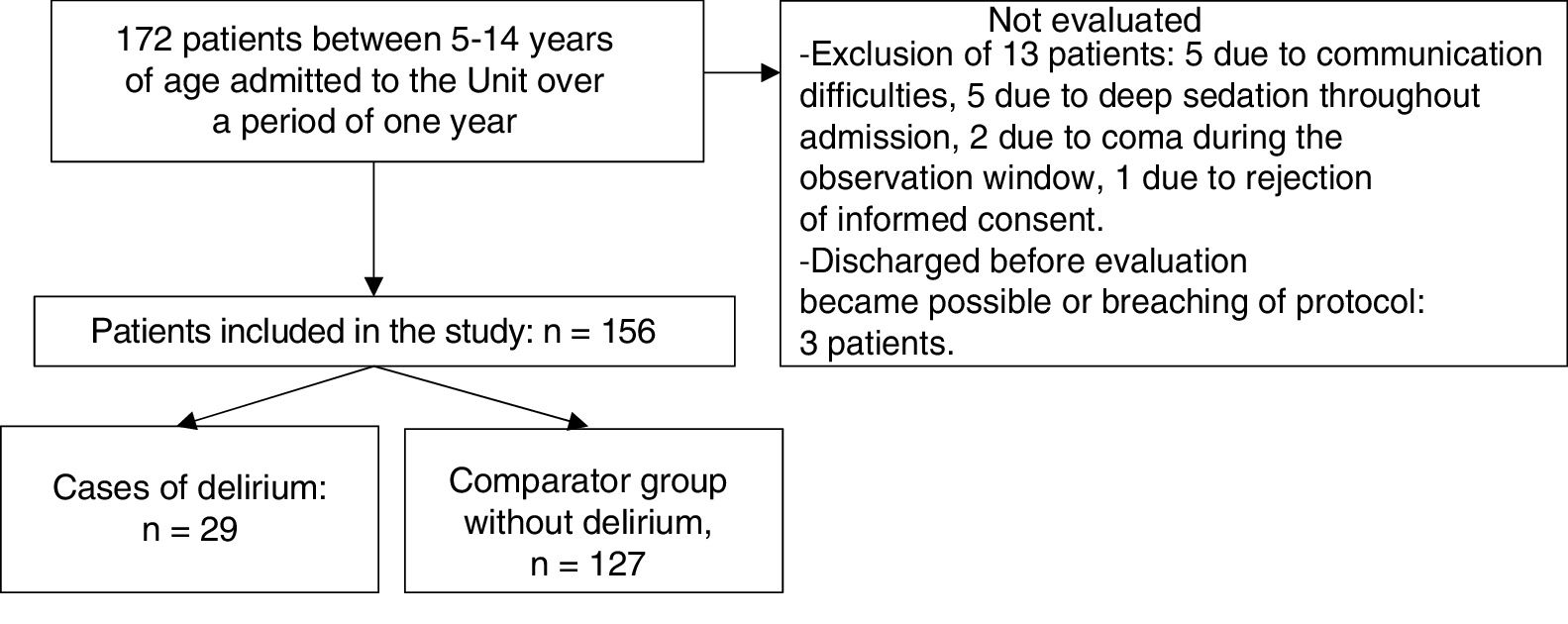
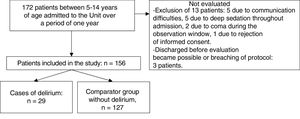
ResultsSample characteristicsA total of 172 patients aged between 5 and 14 years of age were admitted in the course of the study period. The study sample consisted of 156 of these patients (90.7%). Thirteen children presented some exclusion criterion and were not evaluated, while another three were excluded because they had been discharged before evaluation proved possible, or due to breaches in protocol. Fig. 1 shows the patient flow in the study.
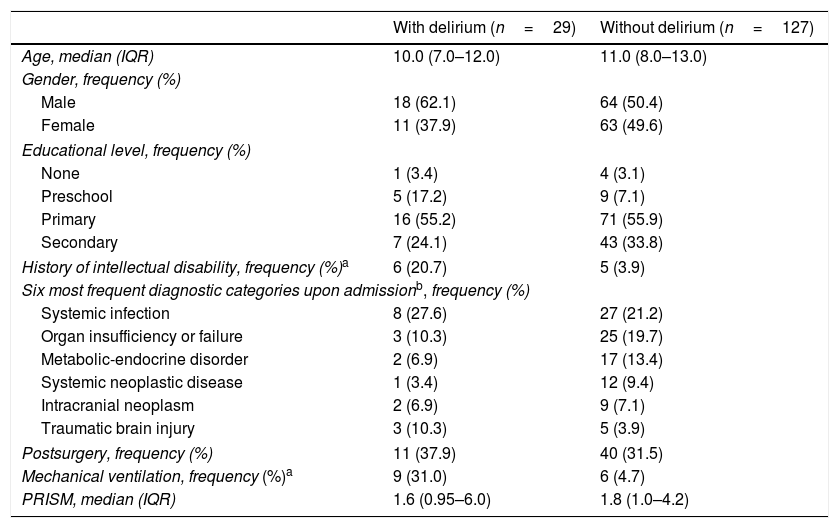
Twenty-nine of the 156 patients included in the study developed delirium (18.6%). The median age of the global sample was 11.0 years (range: 5–14). The median PRISM score was 1.8 (range: 0.3–62.0). Table 1 shows the sociodemographic and clinical characteristics of the 156 patients evaluated. Eighteen received deep sedation upon admission (11.5%), and the median time elapsed to reach RASS≥3 was three days (IQR: 1.0–5.2). Only mechanical ventilation and a history of cognitive impairment were more frequent in the group of patients with delirium. There were no differences between the two groups in terms of the PRISM score.
Sociodemographic and clinical characteristics of the 156 children with and without delirium in the critical care unit.
| With delirium (n=29) | Without delirium (n=127) | |
|---|---|---|
| Age, median (IQR) | 10.0 (7.0–12.0) | 11.0 (8.0–13.0) |
| Gender, frequency (%) | ||
| Male | 18 (62.1) | 64 (50.4) |
| Female | 11 (37.9) | 63 (49.6) |
| Educational level, frequency (%) | ||
| None | 1 (3.4) | 4 (3.1) |
| Preschool | 5 (17.2) | 9 (7.1) |
| Primary | 16 (55.2) | 71 (55.9) |
| Secondary | 7 (24.1) | 43 (33.8) |
| History of intellectual disability, frequency (%)a | 6 (20.7) | 5 (3.9) |
| Six most frequent diagnostic categories upon admissionb, frequency (%) | ||
| Systemic infection | 8 (27.6) | 27 (21.2) |
| Organ insufficiency or failure | 3 (10.3) | 25 (19.7) |
| Metabolic-endocrine disorder | 2 (6.9) | 17 (13.4) |
| Systemic neoplastic disease | 1 (3.4) | 12 (9.4) |
| Intracranial neoplasm | 2 (6.9) | 9 (7.1) |
| Traumatic brain injury | 3 (10.3) | 5 (3.9) |
| Postsurgery, frequency (%) | 11 (37.9) | 40 (31.5) |
| Mechanical ventilation, frequency (%)a | 9 (31.0) | 6 (4.7) |
| PRISM, median (IQR) | 1.6 (0.95–6.0) | 1.8 (1.0–4.2) |
Discrete variables compared with the χ2 test and continuous variables with the Mann–Whitney U-test.
PRISM: Pediatric Risk of Mortality Score; IQR: interquartile range.
Most of the 29 patients with delirium (n=16; 55.2%) were hypoactive, while 7 were mixed cases (24.1%) and 6 were hyperactive (20.7%). There were no patients without motor alterations. In the hypoactive cases the median RASS score was –1 (IQR: –1 to –1; range: –3 to 0), versus –2 in the mixed cases (IQR: –3 to –2; range: –3 to –1) and +2 in the hyperactive patients (IQR: 0 to +2.25; range: 0 to +3).
Table 2 shows that there were no significant differences in the frequency of the acute form of onset, in symptoms fluctuation or in alterations of the RASS (score other than 0) among the three motor types of delirium. Likewise, there were no significant differences in the median of the two clinical characteristics (attention and thought process).
Characteristics evaluated with the pCAM-ICU according to the motor type of delirium in the 29 children admitted to the critical care unit.
| Characteristic | Hypoactive (n=16) | Mixed (n=7) | Hyperactive (n=6) |
|---|---|---|---|
| Clinical presentation according to pCAM-ICU, frequency (%) | |||
| Change versus baseline | 12 (75.0) | 7 (100.0) | 6 (100.0) |
| Fluctuation | 15 (93.7) | 6 (85.7) | 6 (100.0) |
| Attention alteration according to pCAM-ICU, median (IQR) | 1.0 (0.0–6.0) | 0.0 (0.0–2.0) | 4.5 (2.2–5.2) |
| Alteration in RASS, frequency (%) | 15 (93.7) | 7 (100.0) | 6 (100.0) |
| Alteration of the thought process according to pCAM-ICU, median (IQR) | 1.5 (0.0–4.7) | 0.0 (0.0–2.0) | 2.5 (0.7–3.5) |
No differences among the 3 groupsa.
pCAM-ICU: Pediatric Confusion Assessment Method for the Intensive Care Unit; RASS: Richmond Agitation Sedation Scale; IQR: interquartile range.
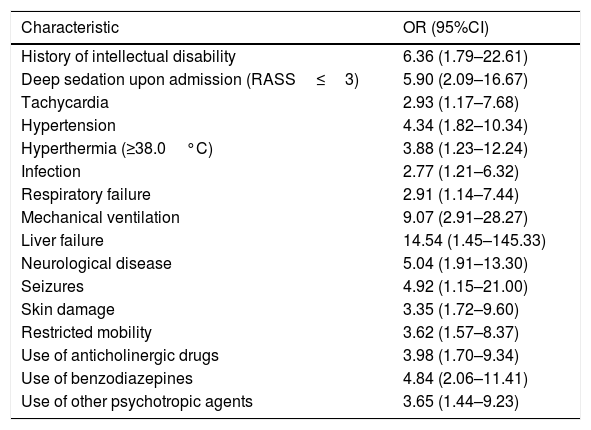
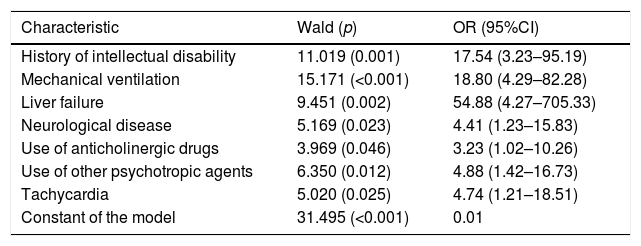
Table 3 reports the variables found to be associated to delirium in the univariate analysis, while Table 4 shows the multivariate logistic regression model (χ2=10.567; p=0.103 for the Hosmer and Lemeshow goodness of fit test).
Characteristics related to the diagnosis of delirium in 29 of 156 children admitted to the critical care unit. Univariate analysis.
| Characteristic | OR (95%CI) |
|---|---|
| History of intellectual disability | 6.36 (1.79–22.61) |
| Deep sedation upon admission (RASS≤3) | 5.90 (2.09–16.67) |
| Tachycardia | 2.93 (1.17–7.68) |
| Hypertension | 4.34 (1.82–10.34) |
| Hyperthermia (≥38.0°C) | 3.88 (1.23–12.24) |
| Infection | 2.77 (1.21–6.32) |
| Respiratory failure | 2.91 (1.14–7.44) |
| Mechanical ventilation | 9.07 (2.91–28.27) |
| Liver failure | 14.54 (1.45–145.33) |
| Neurological disease | 5.04 (1.91–13.30) |
| Seizures | 4.92 (1.15–21.00) |
| Skin damage | 3.35 (1.72–9.60) |
| Restricted mobility | 3.62 (1.57–8.37) |
| Use of anticholinergic drugs | 3.98 (1.70–9.34) |
| Use of benzodiazepines | 4.84 (2.06–11.41) |
| Use of other psychotropic agents | 3.65 (1.44–9.23) |
RASS: Richmond Agitation Sedation Scale.
Multivariate logistic regression model of factors related to delirium in 29 of 156 children admitted to the critical care unit.
| Characteristic | Wald (p) | OR (95%CI) |
|---|---|---|
| History of intellectual disability | 11.019 (0.001) | 17.54 (3.23–95.19) |
| Mechanical ventilation | 15.171 (<0.001) | 18.80 (4.29–82.28) |
| Liver failure | 9.451 (0.002) | 54.88 (4.27–705.33) |
| Neurological disease | 5.169 (0.023) | 4.41 (1.23–15.83) |
| Use of anticholinergic drugs | 3.969 (0.046) | 3.23 (1.02–10.26) |
| Use of other psychotropic agents | 6.350 (0.012) | 4.88 (1.42–16.73) |
| Tachycardia | 5.020 (0.025) | 4.74 (1.21–18.51) |
| Constant of the model | 31.495 (<0.001) | 0.01 |
In the univariate analysis, a history of intellectual disability, deep sedation upon admission, hypertension, liver failure, neurological impairment, mechanical ventilation and the use of benzodiazepines yielded the highest ORs.
Seven of the 16 variables found to be significant in the univariate analysis also proved significant in the univariate model, though deep sedation, hypertension and the use of benzodiazepines were excluded. In contrast, other variables entered the model, such as the presence of tachycardia and the use of anticholinergic agents or other psychotropic drugs.
DiscussionApproximately one-fifth of the children evaluated in the first 24–72h of admission to the PICU, or evaluated as soon as it became possible, presented delirium. Over one-half of the cases were hypoactive, and the three motor groups or types did not differ in any of the individual features of delirium evaluated by the pCAM-ICU. Of the large series of clinical (past or current alterations) and pharmacological variables studied, mechanical ventilation, the administration of different psychotropic agents and the use of anticholinergic drugs were associated to delirium according to the multivariate analysis.
PrevalenceAccording to a systematic review published in 2014, the data on the prevalence of delirium in critically ill children have historically varied by over 20 percentage points (from 5.0 to 28.0%). The reason for such variability could be the fact that some of the diagnostic methods used are scantly reliable (e.g., based on subjective suspicion on the part of the clinicians) or pose validity problems (e.g., utilization of the Pediatric Anesthesia Emergence Delirium [PAED], designed for the detection of emergent delirium and scantly sensitive to hypoactive cases).18 In more recent studies carried out with tools for the PICU setting, such as the pCAM-ICU19 or the Cornell Assessment of Pediatric Delirium (CAP-D),6,20,21 the prevalence – in coincidence with our own series – has consistently been found to be about 20%, independently of the age range considered. The largest prevalence study is an international survey published in 2017, and which reported a figure of 23.3%.20
Clinical characteristicsThree tools are available for the standardized clinical diagnosis of delirium in critically ill children. On one hand we have the PAED, designed to explore 5 behavioral aspects in emergent delirium, which is generally hyperactive (eye contact, movements with a certain purpose, noticing the surroundings, restlessness and inconsolability).22 In turn, the CAP-D is based on the PAED with the addition of two further items to evaluate hypoactive cases (scantly active, with few movements while awake, and slow response to stimuli).23 Lastly, we have the version of the pCAM-ICU for patients over 5 years of age (published in 2011), which has been available in Spanish since 2013. This instrument is based on the clinical evaluation of the onset and course of the symptoms and the degree of alertness, as well as on the quantification of symptoms of two of the three nuclear domains: attention (cognitive) and the thought process (higher order thought).12,13 The Preschool-CAM-ICU (psCAM-ICU) for patients under 5 years of age appeared in 2016.24
In this study and in others based on the application of standardized tools, approximately one-half of all the cases seen in the PICU are hypoactive.19,20 This type of delirium is also the most common presentation among adults admitted to intensive care.25 In both critical children6 and adults,25 hypoactive presentations imply a poorer prognosis than the other motor types. Furthermore, in contrast to hyperactive or mixed delirium, hypoactive presentations do not imply disruptive behavioral disorders, and in many cases are not detected in pediatric units.26 In this respect it is advisable to establish routine assessment with appropriate tools for detecting all motor types of delirium.
The phenotypes of delirium have been widely studied in the literature referred to adult patients,2,3,15,27–29 making it possible to identify their nuclear domains (cognitive, higher thought control and circadian)2 and to design research criteria fundamented upon the phenotype.3 In adults it has been seen that cognitive performance is similar independently of the motor type involved.30 In children, the pCAM-ICU allows us to quantify two symptoms of two different nuclear domains (attention, which in all cases must be altered in order to establish the diagnosis with the algorithm, and the thought process), contextualized within the clinical course of the patient and the alteration of alertness.12,13 It should be noted that the intensity of the items attention and thought process was very low in all the patients with delirium, independently of the motor type involved (ranges between 0/10 and 4.5/10 and between 0/10 and 2.5/5, respectively). Longitudinal studies are needed, evaluating more features of the three domains of delirium and their evolution over time in larger samples of PICU patients. Such studies would allow us to further our knowledge of the semiologic characteristics of the disorder and to monitor its possible neuropsychological consequences.
Associated factorsThe univariate analysis showed different preventable and non-preventable factors to be related to delirium. Seven variables were finally retained in the multivariate model. Characteristics such as respiratory failure, seizures or systemic infections, which proved significant in the univariate analysis but not in the logistic regression model in our PICU, in which the highest PRISM score was 62.0, possibly may be more relevant in Departments with even more seriously ill patients or in concrete monographic Units.
Cognitive disability, mechanical ventilation, liver failure and tachycardia were the clinical conditions with the strongest correlation to delirium in this model.
Both previous studies and our own series associate intellectual disability with the syndrome.6,20,21 Likewise, previous neurocognitive disorder (dementia) is an important predisposing factor for delirium in adults.31 A parallelism can be established between cognitive dysfunction in both basal functional defect conditions in children (intellectual disability) and in adults (dementia), where brain vulnerability makes it potentially possible for different causal factors with an impact upon metabolism to give rise to the acute cerebral failure known as delirium. In this same line, it is noteworthy that the existence of some neurological disease was also seen to be a factor associated to delirium in the multivariate analysis. It is necessary to explore whether a differential risk exists according to the neurological diagnosis, since children with critical conditions affecting brain areas implicated in delirium (such as the frontal and parietal cortex or thalamus)32 could be at greater risk than other neurological patients.
In this study, mechanical ventilation was seen to be associated to delirium. This has also been evidenced by two other recent studies.6,21 Although aspects such as immobilization, or factors related to oxygenation, contribute to explain the relationship between mechanical ventilation and delirium, the use of different types of psychotropic agents – also found to be significant in our sample and in another risk factor study6 – offers a more plausible explanation.33 In an attempt to improve the wellbeing of patients subjected to mechanical ventilation, more psychotropic agents than necessary can be administered.34 In this respect, a useful measure for reducing the risk of delirium could be optimization of the prescription of psychotropic agents in children both with and without mechanical ventilation.
As evidenced in our study of pediatric patients, there is a close correlation between altered liver function and delirium. Hepatic encephalopathy is very characteristic of liver failure, and delirium is often its first manifestation. Furthermore, since delirium and encephalopathy have a number of neurocognitive features in common, the clinical difficulty of establishing the limits between both conditions makes it necessary to resort to validated tools such as the pCAM-ICU to screen for delirium.35 The association between these disorders could be due to that fact that both share a number of physiopathological characteristics, such as impaired neuronal respiration and oxidative stress (a consequence of inflammatory factors), or excess dopaminergic action together with a decrease in gabaergic transmission.32,35
Diminished cholinergic activity is fundamental in the physiopathology of delirium. The imbalance in dopaminergic (relative increase) and cholinergic activity (relative decrease) at central level constitutes the common neuronal pathway for the different etiologies.16,32 The association between anticholinergic drugs and delirium observed in this study confirms the data reported in both critically ill adults36 and children.6 Many drugs, or their metabolites, exert anticholinergic effects that may exert cumulative actions when several of them are used.37 Rationalization of drug use is therefore an obviously needed measure.
Lastly, regarding the causes of sinus tachycardia, which in this study was found to be associated to delirium, mention must be made of increased physical activity, the effects of drugs such as anticholinergic agents, or the physiological response to fever, disease or stress.38 Accordingly, the relationship between delirium and tachycardia may be dual. On one hand, the increase in motor activity characteristic of hyperactive or mixed delirium may increase the heart rate, and on the other hand different health and treatment conditions of the patients may also lead to tachycardia. In this way, tachycardia within the logistic regression model could represent an epiphenomenon related to different causes of delirium and to the increase in motor activity of some patients with this disorder. A longitudinal study could clarify whether the presence of tachycardia may be useful as a clinical predictor of delirium in children admitted to the PICU.
Limitations and conclusionsThe results of this study should be interpreted taking a number of limitations into account: (a) We explored the relationship between different clinical factors and delirium on a cross-sectional basis. This design does not allow us to assess the time relationship between the variable of interest (delirium) and the studied factors. On the other hand, the problem addressed in our article has been little studied to date, and this first approach considers associations between different variables that need to be replicated through subsequent longitudinal studies. (b) Although we evaluated patients during one year on a consecutive basis and the prevalence of delirium was high, a sample size limited to 29 cases does not allow us to draw firm conclusions regarding the relative importance of the factors related with delirium – the risk of both qualitative (variable independently associated to the disorder) and quantitative (magnitude of the association) error being high. Larger studies are therefore needed. (c) The lack of instruments for evaluating patients under 5 years of age and the absence of patients over 14 years of age prevented us from studying these population groups. The results obtained therefore cannot be extrapolated to them. (d) According to the PRISM score, we did not study patients in the uppermost third of mortality risk. The policy of the hospital favors the early admission of critical patients to the Unit, in order to guarantee the opportune interdisciplinary care. Although the evaluated patients presented a broad range of health problems, the different types of PICUs require the conduction of studies in Units with other operating protocols, of a monographic nature or with a greater prevalence of concrete disease conditions, with more patients receiving deep sedation and subjected to mechanical ventilation, as well as with groups presenting an increased mortality risk.
It should be underscored that although our sample of patients with delirium was small, a detailed account of the individual neuropsychiatric characteristics was provided, evaluated with the pCAM-ICU according to the motor types of the DRS-R98. This allowed us to contribute to the clinical understanding of the syndrome, which has been little studied in the PICU setting to date.
In conclusion, delirium is very prevalent in pediatric intensive care, and because of its health consequences needs to be dealt with on a routine basis. The characteristics of the syndrome are similar in all motor types of the disease, and hypoactive delirium is the most common presentation. As a result, clinical suspicion needs to be emphasized, since hypoactive forms of the disease are often overlooked. Different factors related to the physiopathology of delirium are related to the diagnosis in children admitted to the PICU. Some factors may be preventable through the rationalization of medical care.
Financial supportThis study was financed by the Dirección de Investigación e Innovación (CIDI) of the Universidad Pontificia Bolivariana (project: 434B-08/15-45). The CIDI did not participate in the design of the study or in data compilation, analysis or interpretation. Likewise, it did not intervene in the drafting of the article or in the decision to submit it for publication.
AuthorshipCarmenza Ricardo-Ramirez and Matilde Ligia Álvarez-Gómez participated in conception of the study, project design, patient evaluation, data compilation, data analysis and drafting of the manuscript. Camilo Andrés Agudelo-Vélez and José Gabriel Franco-Vásquez participated in critical review of the project, patient evaluation, data compilation, data processing and analysis and drafting of the manuscript. Susana Zuluaga-Penagos, Ricardo Antonio Consuegra-Peña, Katerine Uribe-Hernández, Isabel Cristina Mejía-Gil and Eliana María Cano-Londoño participated in critical review of the project, patient evaluation, data compilation and critical review of the article. Mussatye Elorza-Parra participated in patient evaluation, data compilation and critical review of the article. All the authors reviewed and approved submission for publication of the final manuscript.
Conflicts of interestThe authors declare that they have no financial or personal conflicts of interest in relation to the contents of this article.
Please cite this article as: Ricardo Ramirez C, Álvarez Gómez ML, Agudelo Vélez CA, Zuluaga Penagos S, Consuegra Peña RA, Uribe Hernández K, et al. Características clínicas, prevalencia y factores asociados al delirium en niños de 5 a 14 años de edad ingresados en cuidados intensivos. Med Intensiva. 2019;43:147–155.