To estimate the prevalence of frailty in patients admitted to the Intensive Care Unit (ICU) and its impact upon ICU mortality at 1 and 6 months.
DesignA prospective observational cohort study was carried out.
SettingSpanish ICU.
InterventionNone.
Patients and methodsPatients≥65 years of age admitted to the ICU for >24h. Variables were registered upon admission, and functional status was assessed by telephone calls 1 and 6 months after discharge from the ICU.
Main study variablesAge, gender, frailty (FRAIL scale), functional status (Barthel, Lawton, Clinical Dementia Rating and NUTRIC score), days of mechanical ventilation (MV), functional score (APACHE II and SOFA), ICU mortality, and mortality 1 and 6 months after ICU discharge.
ResultsA total of 132 patients were evaluated, of which 46 were frail (34.9%). Age of the frail versus non-frail patients: 78.8±7.2 and 78.6±6.4 years, respectively (P=.43); male gender: 43.8% versus 56.3% (P=.10); SOFA score: 4.7±2.9 versus 4.6±2.9 (P=.75); MV: 33.3% versus 66.7% (P=.75); days of MV: 5.6±15 versus 4.3±8.1 (P=.57); ICU mortality 13% versus 6% (P=.14), mortality at 1 month 24% versus 8% (P=.01), mortality 6 months 32% versus 15% (P=.03). Frailty is associated with mortality at one month (OR=3.5, P<.05, 95% CI (1.22–10.03) and at 6 months after discharge from the ICU (OR=2.62, P<.05, 95% CI (1.04–6.56).
ConclusionsFrailty was present in 35% of the patients admitted to the ICU, and was associated with mortality.
Estimar la prevalencia de fragilidad en pacientes ingresados en cuidados intensivos (UCI) y su impacto sobre la mortalidad intra UCI, al mes y a los 6 meses.
DiseñoEstudio de cohorte prospectiva.
ÁmbitoUCI polivalentes españolas.
IntervenciónNinguna.
Pacientes y métodosPacientes≥65 años ingresados en UCI>24horas. Se recogieron las variables al ingreso y la situación basal por teléfono al mes y a los 6 meses del alta de UCI.
Variables de interés principalEdad, sexo, fragilidad (escala FRAIL), situación basal (Barthel, Lawton, Clinical Dementia Rating y Nutric Score), días de ventilación mecánica (VM), escalas de gravedad (APACHE II y SOFA), mortalidad UCI, al mes y a los 6 meses del alta.
ResultadosCiento treinta y dos pacientes, 46 frágiles (34,9%). Pacientes frágiles vs. no frágiles: 78,8±7,2 vs. 78,6±6,4 años (p=0,43), varones 43,8% vs. 56,3% (p=0,10), SOFA 4,7±2,9 vs. 4,6±2,9 (p=0,75), VM 33.3% vs. 66,7% (p=0,75), días de VM 5,6±15 vs. 4,3±8,1 (p=0,57), mortalidad UCI 13% versus 6% (p = 0,14), mortalidad al mes 24% versus 8% (p = 0,01), mortalidad 6 meses 32% versus 15% (p = 0,03). La fragilidad se asocia con la mortalidad al mes (OR = 3,5; p <0,05, IC del 95% (1,22-10,03) y a los 6 meses del alta de UCI (OR = 2,62; p <0,05, IC del 95% (1,04-6,56).
ConclusionesLa fragilidad está presente en el 35% de los pacientes ingresados en UCI, asociándose a la mortalidad.
At present, 19% of the Spanish population is 65 years old or older (Spanish National Statistics Institute [Instituto Nacional de Estadística, INE] 2018). This population group in turn accounts for 55% of all hospital admissions (with stays that are longer than in other age groups), and 44% of all hospital discharges.1 A consequence of the aging of the population is that the patients admitted to the Intensive Care Unit (ICU) are also becoming older. Patient age traditionally has been regarded as one of the main determinants of admission to the ICU2 because of its relation to the prognosis, and constitutes a variable to be considered in the severity indices of the patients admitted to our Units. However, if age is excluded from the calculation of these indices, the resulting scores are not significantly modified – a fact that indicates that other factors are responsible for the prognosis.3–6
On the other hand, many studies indicate that the prognosis of the critically ill is more related to the prior functional condition of the patient than to age.7–11 The poorer the baseline condition, the greater the mortality rate and the decisions referred to limitation of life support. The prior situation of the patient has also been related to recovery after passing through intensive care.
In relation to the importance of the baseline condition in terms of the patient prognosis, mention must be made of the concept of frailty, initially described by Fried et al.12 Frailty is a physiological syndrome related to old age, and is characterized by a reduced functional reserve and lessened resistance to stressors that result in increased vulnerability and limited recovery capacity after injury or disease. Frailty is different from disability, because it can affect healthy individuals yet implies a greater risk of fatal outcomes in the presence of any of the following characteristics: recent hospital admission, recurrent falls, comorbidity (osteoarthrosis, fractures, depression, hearing and vision problems, incontinence, cardiovascular events), polypharmacy, muscle weakness, mobility and balance problems, limited physical exercise, cognitive impairment or adverse social conditions. Since the studies of Fried and Rockwood, the measurement of frailty has been widely described in the literature.12–18
Frailty has been associated to increased mortality in all the scenarios in which it has been studied – including the critically ill.6,10,11,18 Hence the identification of frail patients upon admission to the ICU could help predict the clinical outcome and serve to intensify patient care at discharge from the Unit.
Few studies have been made of frailty in patients admitted to the ICU in Spain. The present study was carried out to describe the demographic and clinical characteristics of patients over 65 years of age with criteria of frailty admitted to the ICU, comparing them versus patients without frailty, and estimating the impact of the diagnosis of frailty and/or the baseline condition of the patient upon the prognosis at one and 6 months after admission to the ICU.
Patients and methodsA prospective, multicenter observational study was carried out in four polyvalent ICUs (Hospital Universitario de Getafe, Hospital 12 de Octubre, Hospital Universitario General de Cataluña, Hospital Universitario Sagrat Cor) between 1 June 2016 and 1 June 2017. We included all critical patients aged 65 years or older admitted to the ICU during the mentioned study period and with an expected stay of over 24h. Patients who rejected participation in the study were excluded, as were those with limitation of life support measures or expected mortality in the first 48–72h of admission.
In the first 24h of admission, and after obtaining informed consent from the patient or a relative (if the patient was disabled, sedated or subjected to orotracheal intubation), an interview was held to determine patient frailty and baseline condition (functional and cognitive condition), prior to admission to the ICU. Frailty was measured using the Frail Scale (Morley et al.) based on 5 items: fatigability, resistance, walking, comorbidity and weight loss. Each item was scored as one point. The sum of the individual scores ranged from 0 to 5 (0=best, 5=worst), and indicated frail health (3–5 points), pre-frailty (1–2 points) and robustness (0 points). In our study, patients with a score of 0–2 points were regarded as not frail, while those with three or more points were considered to be frail. Fatigue was measured by asking the interviewee how long he/she felt tired in the last four weeks – the answers “all the time” and “most of the time” each being scored as one point. Resistance in turn was evaluated by questioning about difficulties in walking up 10 steps alone without stopping and without help, while walking was assessed by asking the interviewee if he/she had difficulties walking 200m or one block alone and without help. The answer “yes” was scored as one point. Disease was scored as one point in the case of patients reporting 5 or more disorders out of a total of 11 conditions: dementia, heart disease, depression, osteoarthrosis, asthma, bronchitis/emphysema, diabetes, arterial hypertension, osteoporosis and stroke. Weight loss was scored as one point in the case of patients with an unintended weight loss of 5% or more in the last 12 months.
The patient baseline condition was evaluated using the Barthel index for the assessment of basic activities of daily living (dependency being defined as a score of <60 points), while the Lawton and Brody index was used to score instrumental activities (involving a score without a cut-off point, where the maximum score is 8 and defines total independence, while a score of 0 is indicative of dependency). Cognitive function in turn was evaluated by means of the Clinical Dementia Rating (CDR) scale, where 0.5 indicates cognitive impairment and scores of over 2.5 are suggestive of dementia.
In addition, we prospectively compiled patient age and gender, reason for admission, Charlson comorbidity index (presence of comorbidity being defined by a score>3), nutritional risk (based on the Nutric score, with the presence of comorbidity being defined by a score>5), severity upon admission (assessed by means of the SOFA and APACHE II scores), number of falls in the last 6 months, number of hospital admissions in the last year, duration of stay in the ICU and in hospital, days of mechanical ventilation, need for tracheostomy, complications in the ICU (see definition in annex 1 online), limitation of life support, mortality in the ICU, mortality at one and 6 months, and patient destination at hospital discharge (home, home but with need for a caregiver, home for the elderly, functional rehabilitation center). One and 6 months after discharge from the ICU, we again assessed the baseline condition of the patient (Barthel index, Lawton index and CDR) through a telephone interview.
A descriptive study of the patients was made, followed by univariate comparison between frail and non-frail patients. Given the importance attributed to age, we also classified the patients into two groups: elderly patients aged 65–80 years and very elderly individuals (>80 years of age), with comparison of both groups to assess possible differences attributable to age.
The study was approved by the Clinical Research Ethics Committees of each of the participating hospitals.
Statistical analysisNormal data distribution was assessed using the Smirnov–Kolmogorov test. The data were reported as the mean and standard deviation (SD) or as the median and interquartile range (IQR), depending on the type of distribution, and as absolute values and percentages. Comparisons were made with the chi-squared test, the parametric Student t-test and the nonparametric Mann–Whitney U-test and Kruskal–Wallis test, according to the distribution of the variable. Statistical significance was considered for p<0.05.
A multivariate analysis was performed, using logistic regression models and taking as dependent variables mortality in the ICU and mortality at one and 6 months after discharge from the ICU. In turn, the independent variables were defined as those variables that were identified in the univariate analysis as being associated to the outcomes of interest (mortality) with a value p<0.10, as well as those variables of clinical interest. Since the number of variables to be entered in the multivariate model was limited by the prevalence of the outcome, we were only able to include one to two variables. For this reason fitting of the model was limited to the APACHE II score as the variable summarizing global severity upon admission, and to patient age as the variable of clinical interest, in compliance with the objectives of our study.
The SPSS version 21 statistical package was used throughout.
ResultsWe included a total of 132 patients, of which 46 (35%) met criteria of frailty. In the telephone follow-up we recorded a loss of 8 patients at one month and 14 patients at 6 months.
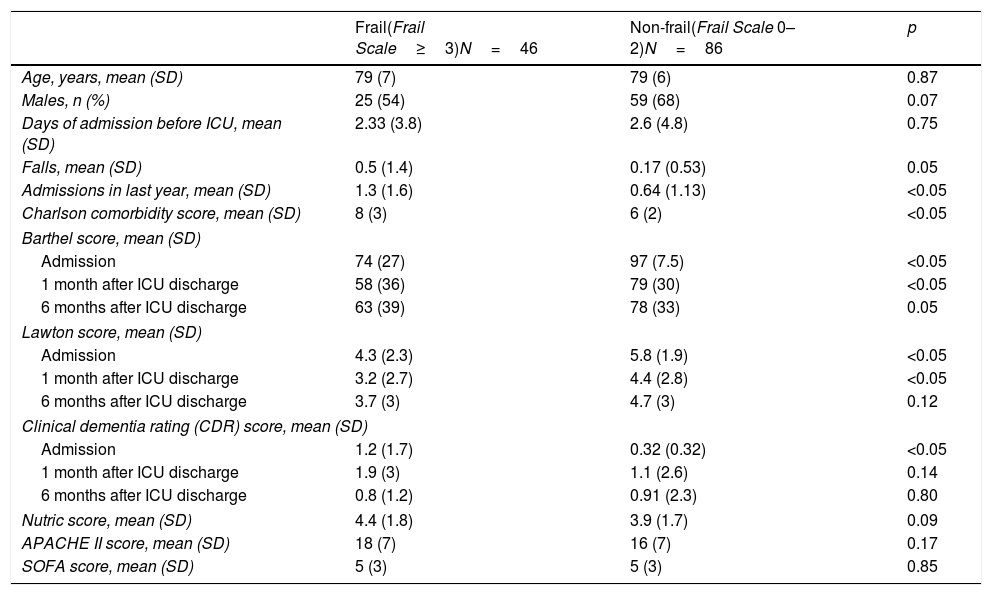
The frail patients suffered more falls and required more hospital admissions in the previous year, and moreover suffered greater comorbidity (Charlson comorbidity index). Age and severity at the time of admission to the ICU (APACHE II, SOFA) were similar in both groups (Table 1).
Comparison of the baseline characteristics of the frail and non-frail patients.
| Frail(Frail Scale≥3)N=46 | Non-frail(Frail Scale 0–2)N=86 | p | |
|---|---|---|---|
| Age, years, mean (SD) | 79 (7) | 79 (6) | 0.87 |
| Males, n (%) | 25 (54) | 59 (68) | 0.07 |
| Days of admission before ICU, mean (SD) | 2.33 (3.8) | 2.6 (4.8) | 0.75 |
| Falls, mean (SD) | 0.5 (1.4) | 0.17 (0.53) | 0.05 |
| Admissions in last year, mean (SD) | 1.3 (1.6) | 0.64 (1.13) | <0.05 |
| Charlson comorbidity score, mean (SD) | 8 (3) | 6 (2) | <0.05 |
| Barthel score, mean (SD) | |||
| Admission | 74 (27) | 97 (7.5) | <0.05 |
| 1 month after ICU discharge | 58 (36) | 79 (30) | <0.05 |
| 6 months after ICU discharge | 63 (39) | 78 (33) | 0.05 |
| Lawton score, mean (SD) | |||
| Admission | 4.3 (2.3) | 5.8 (1.9) | <0.05 |
| 1 month after ICU discharge | 3.2 (2.7) | 4.4 (2.8) | <0.05 |
| 6 months after ICU discharge | 3.7 (3) | 4.7 (3) | 0.12 |
| Clinical dementia rating (CDR) score, mean (SD) | |||
| Admission | 1.2 (1.7) | 0.32 (0.32) | <0.05 |
| 1 month after ICU discharge | 1.9 (3) | 1.1 (2.6) | 0.14 |
| 6 months after ICU discharge | 0.8 (1.2) | 0.91 (2.3) | 0.80 |
| Nutric score, mean (SD) | 4.4 (1.8) | 3.9 (1.7) | 0.09 |
| APACHE II score, mean (SD) | 18 (7) | 16 (7) | 0.17 |
| SOFA score, mean (SD) | 5 (3) | 5 (3) | 0.85 |
SD: standard deviation; n (%): number of patients (percentage).
The frail patients also presented poorer baseline functional (Barthel and Lawton scores), cognitive (CDR) and nutritional conditions (Nutric score) than the non-frail patients (Table 1) upon admission to the ICU. Functional condition was poorer in frail patients than in non-frail patients (without reaching statistical significance 6 months after discharge from the ICU). Cognitive function one month after discharge from the ICU tended to be worse in frail patients than in non-frail individuals (p=0.14), though the difference was no longer noticeable 6 months after discharge from the ICU.
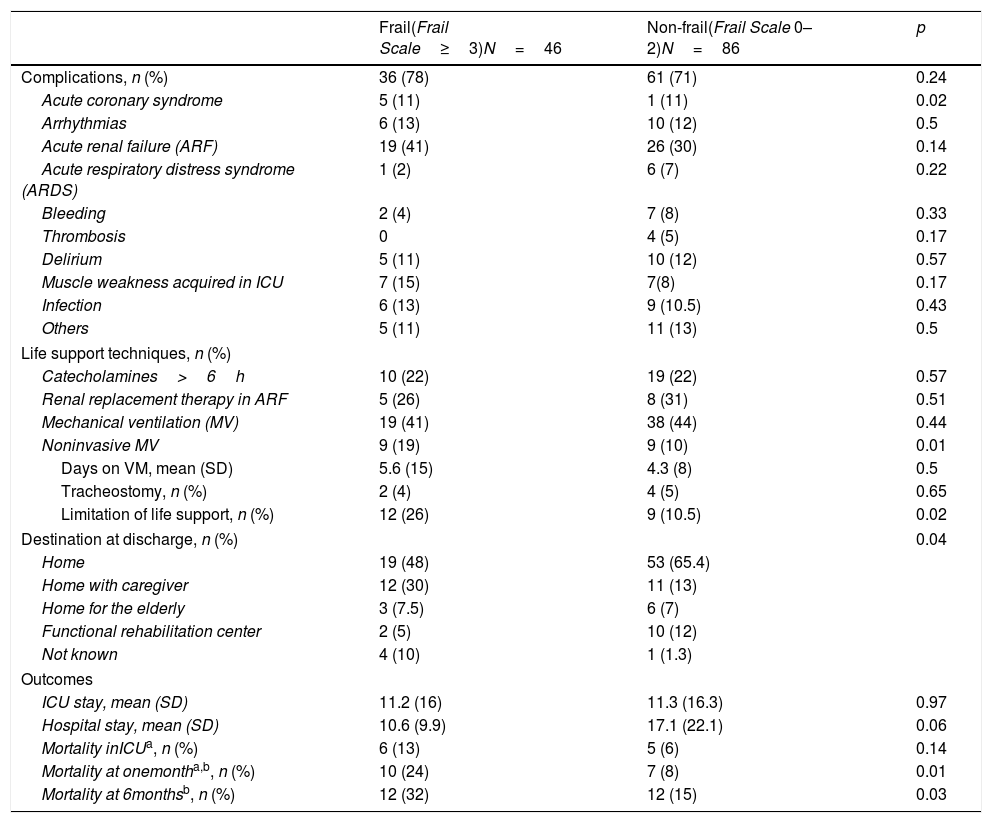
The need for mechanical ventilation and tracheostomy was slightly greater among the non-frail patients. However, noninvasive mechanical ventilation was more frequent in frail patients (p=0.01). The duration of stay in the ICU was similar in frail patients and in non-frail individuals, while hospital stay tended to be longer among the latter (p=0.06). Complications were more frequent in frail patients, particularly acute renal failure and weakness acquired in the ICU (Table 2).
Comparison of the variables related to ICU stay.
| Frail(Frail Scale≥3)N=46 | Non-frail(Frail Scale 0–2)N=86 | p | |
|---|---|---|---|
| Complications, n (%) | 36 (78) | 61 (71) | 0.24 |
| Acute coronary syndrome | 5 (11) | 1 (11) | 0.02 |
| Arrhythmias | 6 (13) | 10 (12) | 0.5 |
| Acute renal failure (ARF) | 19 (41) | 26 (30) | 0.14 |
| Acute respiratory distress syndrome (ARDS) | 1 (2) | 6 (7) | 0.22 |
| Bleeding | 2 (4) | 7 (8) | 0.33 |
| Thrombosis | 0 | 4 (5) | 0.17 |
| Delirium | 5 (11) | 10 (12) | 0.57 |
| Muscle weakness acquired in ICU | 7 (15) | 7(8) | 0.17 |
| Infection | 6 (13) | 9 (10.5) | 0.43 |
| Others | 5 (11) | 11 (13) | 0.5 |
| Life support techniques, n (%) | |||
| Catecholamines>6h | 10 (22) | 19 (22) | 0.57 |
| Renal replacement therapy in ARF | 5 (26) | 8 (31) | 0.51 |
| Mechanical ventilation (MV) | 19 (41) | 38 (44) | 0.44 |
| Noninvasive MV | 9 (19) | 9 (10) | 0.01 |
| Days on VM, mean (SD) | 5.6 (15) | 4.3 (8) | 0.5 |
| Tracheostomy, n (%) | 2 (4) | 4 (5) | 0.65 |
| Limitation of life support, n (%) | 12 (26) | 9 (10.5) | 0.02 |
| Destination at discharge, n (%) | 0.04 | ||
| Home | 19 (48) | 53 (65.4) | |
| Home with caregiver | 12 (30) | 11 (13) | |
| Home for the elderly | 3 (7.5) | 6 (7) | |
| Functional rehabilitation center | 2 (5) | 10 (12) | |
| Not known | 4 (10) | 1 (1.3) | |
| Outcomes | |||
| ICU stay, mean (SD) | 11.2 (16) | 11.3 (16.3) | 0.97 |
| Hospital stay, mean (SD) | 10.6 (9.9) | 17.1 (22.1) | 0.06 |
| Mortality inICUa, n (%) | 6 (13) | 5 (6) | 0.14 |
| Mortality at onemontha,b, n (%) | 10 (24) | 7 (8) | 0.01 |
| Mortality at 6monthsb, n (%) | 12 (32) | 12 (15) | 0.03 |
SD: standard deviation; n (%): number of patients (percentage).
Mortality among the frail patients was greater in all three periods considered, though statistical significance was only reached one month and 6 months after discharge from the ICU.
A significantly greater proportion of frail patients were subjected to limitation of life support, while a greater proportion of non-frail patients were discharged home (Table 2).
The logistic regression models showed frailty to be associated to mortality at one month (odds ratio [OR]=3.5; p<0.05, 95% confidence interval [95%CI] 1.22–10.03) and 6 months after discharge from the ICU (OR=2.62; p<0.05. 95%CI 1.04–6.56).
On the other hand, as can be seen in Table 1 of the supplementary material, on comparing elderly patients (65–80 years of age) versus very elderly individuals (>80 years of age), no significant differences were observed in the incidence of frailty or in terms of other variables, except comorbidity (p<0.05). In this subanalysis, the very elderly patients tended to present functional dependency after both one and 6 months, with discretely greater mortality – though statistical significance was not reached in any of the periods considered.
DiscussionThe prevalence of frailty in our study was similar to that found in the literature.6,11,18 Although frailty has been associated to elderly patients (>80 years),6 it can also be found in younger individuals.11 In our series the incidence was similar in elderly patients and in very elderly individuals. Likewise, no significant differences were observed on comparing different patient variables by age groups – this seeming to confirm the existence of variables more important than age in defining the prognosis of patients admitted to the ICU. Frailty should be taken into account in identifying patients most likely to benefit from admission to critical care, since it is associated to mortality.
No association was observed in our study between the baseline condition and mortality, in contrast to what is found in the literature, where functional condition is identified as an independent mortality risk factor.10 However, the frail patients had a poorer baseline functional and cognitive condition, and this made them more vulnerable. They suffered more falls, with more previous admissions and greater comorbidity. The functional scores (Barthel and Lawton indices) were low upon admission to the ICU, and dependency persisted without recovery over time. In contrast, the cognitive scores improved after discharge from the ICU. To our knowledge, such improvement in the CDR score is related to the characteristics of the test used to assess cognitive impairment, and constitutes a subjective appraisal of the condition of the patient in different areas (memory, orientation, interpersonal relations and self-care capacity).
The mortality risk scales (APACHE II and SOFA) showed no differences between the two groups of patients, though the literature describes a higher score on the SAPS 3 scale as being associated to increased mortality.10 Under equal severity conditions upon admission, the frail patient requires less orotracheal intubation, vasopressor drug use and renal replacement therapy. Even the increased use of noninvasive ventilation in the group of frail patients may be conditioned by an attempt to not resort to mechanical ventilation in these individuals. Accordingly, treatment in frail patients may be less intense because of their baseline condition, with more frequent limitation of life support. The use of resources in the ICU is thus limited as a result. At discharge from our Units, frail patients are referred to a home for the elderly or to their own home with the need for a caregiver, while non-frail patients are referred to a functional rehabilitation center, with more intensive use of post-ICU management resources.
A lack of physical exercise has been found to be a risk factor for the appearance of frailty, with secondary sarcopenia and muscle weakness.19–25 This may explain why weakness acquired in the ICU was a frequent complication in frail patients in our study, giving rise to poorer recovery over the long term.
Le Maguet et al.18 measured frailty using two scales: Frailty Phenotype (FP, frailty≥3) and the Clinical Frailty Score (CFS≥5). Mortality in the ICU was better predicted by the FP, while the CFS was better in predicting mortality after 6 months. Flaatten et al. in turn found the CFS to be associated to short-term mortality. In our study, the Frail scale showed good performance in predicting mortality after one month and 6 months, but not in predicting mortality in the ICU. In any case, and independently of the scale used, it can be concluded that frailty is associated to mortality and is a factor to be taken into account in the prognosis of our patients.
Our study has limitations. Despite its multicenter nature, the number of included patients was not very large. Furthermore, follow-up was only made during the 6 months after discharge, without examining whether there is longer term recovery among the patients referred to functional rehabilitation centers. Furthermore, there were losses to telephone follow-up, which conditions extrapolation of the results, particularly as regards mortality at one month and 6 months.
Frailty is evidenced in 35% of the patients over 65 years of age admitted to the ICU, and determines mortality among the seriously ill. Frail patients have poorer baseline conditions at the time of admission to intensive care, with scant recovery at discharge from the ICU. This requires more intense and earlier post-ICU care26,27 in an attempt to improve the prognosis.
Contribution of the authorsDesign: S. López-Cuenca and A. Artigas-Raventós.
Data compilation: S. López-Cuenca, L. Oteiza-López, N. Lázaro-Martín, M.M. Irazabal-Jaimes and M. Ibarz-Villamayor.
Analysis and preparation of the manuscript: S. López-Cuenca and J.A. Lorente-Balanza.
Financial supportThe study was supported by a research aid from the Fundación del Enfermo Crítico (2017). Sociedad Española de Medicina Intensiva y Unidades Coronarias (SEMICyUC), Spain.
Conflicts of interestNone.
Please cite this article as: López Cuenca S, Oteiza López L, Lázaro Martín N, Irazabal Jaimes MM, Ibarz Villamayor M, Artigas A, et al. Fragilidad en pacientes mayores de 65 años ingresados en cuidados intensivos (FRAIL-ICU). Med Intensiva. 2019;43:395–401.