Cardiogenic shock (CS) is a highly complex clinical condition that requires a management strategy focused on early resolution of the underlying cause and the provision of circulatory support. In cases of refractory CS, mechanical circulatory support (MCS) is employed to replace the failed cardiocirculatory system, thereby preventing the development of multiorgan failure. There are various types of MCS, and patients with CS typically require devices that are either short-term (< 15 days) or intermediate-term (15–30 days). When choosing the device the underlying cause of CS, as well as the presence or absence of concomitant conditions such as failed ventricle, respiratory failure, and the intended purpose of the support should be taken into consideration. Patients with MCS require the comprehensive care indicated in complex critically ill patients with multiorgan dysfunction, with an emphasis on device monitoring and control. Different complications may arise during support management, and its withdrawal must be protocolized.
El shock cardiogénico (SC) es un cuadro clínico de alta complejidad que requiere un manejo centrado en resolver de forma precoz la causa condicionante y asegurar un soporte circulatorio. En caso de shock refractario, los sistemas de soporte circulatorio mecánico (SCM) permiten suplir al sistema cardiocirculatorio fracasado para evitar el desarrollo de fracaso multiorgánico. Existen diferentes tipos de SCM y en los pacientes en SC se suelen contemplar dispositivos de corta (menos de 15 días) o intermedia duración (15–30 días). Para su elección se debe tener en cuenta la causa que ha condicionado el SC y aspectos como el ventrículo fracasado, la presencia/ausencia de insuficiencia respiratoria y el propósito del soporte. Los pacientes con SCM requieren un cuidado integral de enfermo crítico complejo con disfunción multiorgánica, haciendo hincapié en la monitorización y el control del dispositivo. Durante su manejo puede aparecer diferentes complicaciones y su retirada debe estar protocolizada.
Cardiogenic shock (CS) is a highly complex clinical condition that requires progressive dynamic management. This approach is focused on early detection and resolution of the possible underlying causes while also ensuring adequate circulatory support to avoid multiorgan failure (MOF).1 This requires comprehensive and expeditious hemodynamic monitoring2 to ensure preliminary optimization, guided by hemodynamic objectives.3–6 Patients who continue to experience shock despite the initial measures7 will require invasive and advanced echocardiographic monitoring to further assess the underlying etiology, select the optimal therapeutic strategy, monitor the response to the adopted management measures, and plan an adequate mechanical circulatory support (MCS) strategy.8
The fundamental goal of MCS is to compensate for the failure of the cardiocirculatory system in order to prevent the development of MOF. In this regard, MCS must provide oxygen transport (DO2) matched to organ/tissue requirements (VO2); reduce myocardial oxygen consumption to facilitate the recovery of ventricular function; and allow time for cardiac function to improve, facilitate urgent heart transplantation, or decide whether the patient is a candidate for transplantation or longer-term mechanical support.9
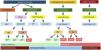
Classification and types of MCSTechnological advances have led to a notable diversification of MCS. The existing techniques are classified according to various criteria, including the level of support provided (either partial or global), the ventricle supported (either uni- or bi-ventricular), the access route used (either peripheral or central), the type of implantation (either percutaneous or surgical), the hemodynamic and/or cardiorespiratory effects produced, and the anticipated duration of the support (short <15 days, intermediate 15–30 days, or long >30 days) (Fig. 1).10
Classification of the mechanical circulatory support systems.
IABP: intra-aortic balloon pump; TH: TandemHeart ®; V-AECMO: venoarterial extracorporeal membrane oxygenation; RV: right ventricle; LV: left ventricle; PD: ProtekDUO ®; DL: Dual Lumen ®; RA-P: right atrium-pulmonary; LV-Ax: left ventricle-axillary; LV-Ao: left ventricle-aorta.
*: percutaneous or surgical, according to availability and urgency.
In patients with CS, the management strategy focuses on the use of short or intermediate-term devices that compensate for the pathophysiological and anatomical characteristics of the patient and support the potential recovery of failed organ-specific cardiac and/or pulmonary function. Furthermore, the existing experience and scientific evidence allow for the complementary use of such devices and the adequate planning of therapeutic escalation and de-escalation.11
Left partial supportIntra-aortic balloon pumpThe intra-aortic (counterpulsation) balloon pump (IABP) remains a widely used MCS option due to its expedient and straightforward placement.12 The mechanism by which the device provides circulatory support is well established. During insufflation of the balloon, an increase in diastolic arterial pressure is observed, which improves blood flow to the coronary, cerebral and renal regions. Conversely, during deflation of the ballon, the afterload, end-diastolic pressure and myocardial work are reduced. Nevertheless, the technique only increases cardiac output to a limited extent (0.5 l/min), and the level of support it provides it typically inadequate for patients with refractory CS.
The effect of the IABP on mortality in CS is subject to controversy, as the IABP SHOCK II trial failed to demonstrate a 30-day mortality benefit with this technique.13,14 The subsequent meta-analyses likewise evidenced no improvement in short- and middle-term survival.15 In fact, the current clinical guidelines do not recommend its generalized use in post-acute myocardial infarction CS.16 Despite this, the IABP is still widely used, mainly in centers where no other types of MCS are available, as a bridging measure for transferring the patient to another center where devices capable of greater hemodynamic support are accessible, The IABP remains useful in certain perioperative scenarios, such as post-cardiotomy shock, mechanical complications of acute myocardial infarction (AMI)(acute mitral valve insufficiency and ventricular septal rupture) or high-risk revascularization surgery.17 Furthermore, it has generated growing interest as a left ventricle unloading strategy in patients subjected to venoarterial extracorporeal membrane oxygenation (V-AECMO), since it appears to reduce mortality, with fewer complications than other devices.18
IMPELLA® (Abiomed, Danvers, MA, USA)This transvalvular microaxial flow univentricular MCS system is implanted percutaneously or surgically and is positioned through the aortic valve in the left ventricle (LV). It impels the blood directly from the ventricular cavity towards the aorta with a continuous flow. This leads to an increase in cardiac output and mean blood pressure, as well as improvements in both systemic and coronary perfusion. Moreover, the optimal emptying of the LV, accompanied by a reduction in left ventricle end-diastolic pressure (LVEDP), contributes to cardiac recoverability, as LV wall stress is diminished, thereby minimizing myocardial oxygen demand.19
Among the devices currently available for use in clinical practice, mention must be made to the IMPELLA® CP and IMPELLA® 5/5.5. The former is implanted percutaneously using a 14 F introducer through the femoral artery (FA), advancing its extremity through the aortic valve and positioning it at 3.5−4 cm from the latter, affording a theoretical flow of up to 3.5−4 l/min. The second device is larger and it´s surgically implanted, preferably through the axillary artery. It can afford a theoretical maximum flow of 5–5.5 l/min, and in contrast to the IMPELLA® CP, it has an assist time of up to 30 days. However, despite their easy implantation and good results, these devices are contraindicated in patients with severe aortic insufficiency or stenosis, mechanical aortic prosthesis, severe peripheral arterial disease, or the presence of a thrombus in the valve or the LV.20 A controversial contraindication is post-infarction ventricular septal defect (VSD), where several series have reported good results.21
Taking into account the results of the latest studies, axial flow MCS devices can be the first option in cases of CS secondary to AMI or single left ventricular failure.22 Escalation to other devices is required if improvement is not observed, if the clinical condition of the patient continues to worsen, or in the case of concomitant alteration of the right ventricle (RV) and/or lungs. Recent systematic reviews have published results comparable to those of extracorporeal membrane oxygenation (ECMO),23 and their concomitant use after escalating to ECMO affords better results than ECMO alone.24,25
TandemHeart® (Livanova)This is a left ventricle MCS device with a percutaneously implanted centrifugal system that extracts blood from the left atrium (LA) and returns it to the FA. Implantation is through the femoral vein with a 21 F cannula that is placed in the LA via the trans-septal route. Although the device offers excellent performance, the technical difficulties of implantation and the easy migration of the LA cannula make its use limited in our setting.10
CentriMag® (Abbott)This is a surgically cannulated MCS device that uses a magnetic levitation centrifugal pump to extract blood from a heart chamber and transport it to the appropriate circulatory system. It is a very widely used device with a theoretical maximum flow of up to 10 L/min. It can be used in univentricular (left, right) or biventricular mode; an oxygenator can be interposed to provide ECMO functions; and it is approved for use beyond 30 days.26 In the case of LV support, although the device can be implanted through a sternotomy at the level of the LA and aorta, a mini-invasive approach can also be used, in which the LV is drained directly from the apex through a mini-thoracotomy and a return to the patient is established using the axillary artery (Supplementary Material 1). In the case of RV support, blood inflow to the system is through the femoral vein, and return to the patient is via the pulmonary artery through a Dacron tube (via sternotomy).
Right partial supportMechanical circulatory support of the right ventricle (RV) is significantly less frequent than MCS of the LV, though its use has increased in recent years, and implantation is via the percutaneous route - with the exception of CentriMag®, which involves surgical implantation.27
IMPELLA RP®(Abiomed): This is a micro-axial assist device that is implanted percutaneously via the femoral route into the right atrium and propels blood into the pulmonary artery.28
ProtekDUO® (LivaNova): This device has a dual-lumen cannula (29 F–31 F) that is implanted percutaneously via the transjugular route in the right atrium (proximal) and pulmonary artery (distal). It is equipped with a centrifugal pump that impulses the blood, affording a flow rate of up to 5 l/min. Moreover, an oxygenator can be interpositioned to provide veno-pulmonary ECMO functions.15,29
Dual Lumen System® (Spectrum Medical): This device has a dual lumen cannula (24 F, 27 F or 31 F) that is implanted through the internal jugular vein and is connected to a centrifugal pump with or without an oxygenator. A circuit is established between the RV and the pulmonary artery, with a maximum flow of 5 l/min.30
Global supportExtracorporeal membrane oxygenation (ECMO)In its venoarterial (V-A) configuration, ECMO is a global MCS system that provides cardiorespiratory support and can be used initially in any context characterized by uni- or biventricular dysfunction with or without respiratory failure.31,32 Since the device can be implanted on an emergent basis, it may be used in the context of cardiorespiratory arrest (CRA) as a rescue resuscitation option (ECMO-CPR), usually placed percutaneously via a peripheral route.33
The device consists of a centrifugal pump that extracts the blood from the venous system of the patient using a cannula – usually of a 19 F–27 F diameter via the femoral vein – and transports it through a circuit to an oxygenator where gas exchange takes place. The oxygenated blood is then returned to the arterial system of the patient using a femoral or axillary 15 F–21 F cannula. Central cannulation via sternotomy or thoracotomy is also possible but poses more complications and is, therefore, less widely recommended.34,35 The size of the cannulas (particularly of the venous cannula) is the main flow limitating factor, and must be taken into account when planning estimated circulatory support and the vascular access to be used for implantation.
Extracorporeal membrane oxygenation reduces cardiac metabolic demand and improves the patient hemodynamics, with a decrease in RV preload; however, due to its characteristics, ECMO may lead to a potential LV pressurization problem. As more flow becomes necessary to assist the patient, LV afterload increases, impeding adequate ventricular emptying and elevating LVEDP. In the more serious cases, this may lead to a worsening of ventricular dysfunction, the closing of the aortic valve, dilatation of the ventricular cavity, lung edema, the appearance of spontaneous echo-contrast, and even intracavitary thrombosis.30 Such situations can be minimized or avoided by the deployment of an IABP, by venting of the LV through the left atrium or the LV, or by using an IMPELLA® device.36 Despite the potential complications, this combined use, known as ECMELLA or ECPELLA (Supplementary Material 2), has been shown to offer advantages in retrospective studies, with lesser patient mortality than when using V-AECMO alone.37,38
Table 1 summarizes the main MCS devices, their characteristics and principal indications.
Characteristics of the main mechanical circulatory support devices.
| IABP | Impella CP®/5.5® | Impella RP® | Centrimag® | V-A ECMO | |
|---|---|---|---|---|---|
| Mechanism | Insufflated in diastole and deflated in systole | Continuous axial flow LV to aorta | Continuous axial flow IVC/RA to PA | Continuous centrifugal flow | Continuous centrifugal flow with oxygenator. |
| Assisted ventricle | LV | LV | RV | RV, LV and/or BiV | BiV |
| Flow | 0.5−1 l/min | 2.5−4 l/min/5.5 l/min | 2−4 l/min | >5 l/min | >5 l/min |
| Support mechanism | Pneumatic | Axial flow | Axial flow | Centrifugal flow | Centrifugal flow |
| Effects | ↑ coronary perfusion↓ afterloadFacilitates unloading of LV | ↑ systemic perfusion↓ LVEDPDirect unloading ofLV | ↓ pressures RV↓ preload RV | ↓ filling and distension of drained cavities | ↑ afterload and ↑ LVEDP↓ preload RVRespiratory support |
| Cannula sizeAccess | 7−8FFA | 13−21 F / 24 FFA/AxA | 22FFV | 19−27 F drainage / 15−21 F returnRV: RA-PA LV: LV – Ao/AxA | 19−27 F drainage / 15−21 F returnFV – FA / FV – AxA/RA -Ao |
| AnticoagulationBleeding risk | − | + | + | ++ | +++ |
| Bleeding risk | + | ++ | ++ | ++ | +++ |
| Risk of ischemia in LL | + | ++ | ++ | − / + (UL) | +++ |
| Notes:Contraindications | Potential use with ECMO for unloading of LV Severe AoI, aortic dissection, peripheral arterial disease | Potential use with ECMO for active unloading of LV (ECMELLA)Severe AoI, aortic dissection, peripheral arterial disease, LV thrombus, mechanical aortic valve | Improves CI and reduces CVP in isolated RV failure.Severe pulmonary insufficiency, IVC/RA/RV thrombus | Can be combined with (Protek-DUO® /Dual Lumen® +/- ECMOInherent to thoracic / heart surgery | Allows global cardiopulmonary assistance. No unloading of the LVSevere AoI, aortic dissection, severe peripheral arterial disease |
IABP: intra-aortic balloon pump; VA-ECMO: venoarterial extracorporeal membrane oxygenation; IVC: inferior vena cava; RA: right atrium; PA: pulmonary artery; LV; left ventricle; RV: right ventricle; BiV: biventricular; LVEDP: left ventricle end-diastolic pressure; FA: femoral artery; FV: femoral vein; Ao: aorta; AxA: axillary artery; LL: lower limbs; UL: upper limbs; IJV: internal jugular vein; AoI: aortic insufficiency; CI: cardiac index; CVP: central venous pressure.
Indications (Table 2) and implantation strategies for MCS vary according to the underlying cause of cardiogenic shock and patient characteristics. Before implanting an MCS, several aspects must be considered to obtain a detailed perspective of the patient, with particular attention to the possible options if no myocardial improvement is achieved after several days of circulatory support (Supplementary Material 3).39 Furthermore, certain situations in which the use of such devices would prove futile must also be considered (Table 3). Two scenarios with different management and prognostic conditions can be considered:
Causes of cardiogenic shock amenable to mechanical circulatory support.
| Cause |
|---|
| Acute coronary syndrome (ACS) |
| Mechanical complications of ACS (VSD, cardiac rupture, valve rupture) |
| Postcardiotomy syndrome (includes incapacity to wean from extracorporeal circulation) |
| Myocarditis |
| Primary graft dysfunction following TxC |
| Refractory ventricular arrhythmias |
| Complicated severe intoxication or infection with myocardial dysfunction |
| Severe hypothermia (<28 °C) |
| Massive PTE |
| Severe hypoxic pulmonary vasoconstriction |
| Exacerbation of chronic heart failure |
| Myocardiopathies |
| Acute valve disease |
| High-risk percutaneous procedures |
ACS: acute coronary syndrome; VSD: ventricular septal defect; TxC: cardiac transplantation; PTE: pulmonary thromboembolism.
Contraindications of mechanical circulatory support.
| Absolute |
|---|
| Metastatic cancer |
| Prolonged CPR without adequate tissue perfusion |
| Non-witnessed cardiac arrest |
| Irreversible severe brain damage |
| Irreversible severe multiorgan failure |
| Short-term lethal chromosomal abnormalities |
| Untreatable aortic dissection or insufficiency |
| Untreatable cardiac tamponade |
| Relative |
|---|
| Advanced age |
| Severe bleeding problems |
| Morbid obesity |
| Severe peripheral arterial disease (only for peripheral cannulation) |
| Previous disease conditions with poor long-term prognosis |
CPR: cardiopulmonary resuscitation.
Multiple disease conditions can originate cardiogenic shock (CS), which is characterized by high morbidity and mortality. The leading cause is acute myocardial infarction (AMI), with a variable prevalence (44–77%) and important temporal and geographical differences. It should be taken into account that the prognosis differs depending on the underlying cause of CS. Although different studies have reported greater survival among patients with AMI, others suggest that pulmonary thromboembolism, fulminant acute myocarditis or acute valve disease have a better prognosis – with malignant ventricular arrhythmias presenting poorer survival rates. These are retrospective studies, however, and the distribution of the different assist devices employed differs from that of current practice; it is therefore difficult to consider etiology as a prognostic factor.40–42
Different risk scales have been proposed in an attempt to predict mortality among patients with CS subjected to ECMO. The most widely used instrument is the SAVE score, which stratifies patients into 5 risk groups associated with different survival rates.38 The ENCOURAGE scale in turn was designed based on a population of patients with AMI, while the REMEMBER scale was developed for patients with post-cardiotomy shock following coronary surgery.43,44 These latter two instruments are less widely used in routine practice.
Exacerbated advanced heart failureIn patients with advanced heart failure (AHF) refractory to conventional therapy, ventricular assist devices (VADs) have shown to improve survival and quality of life.45 The INTERMACS scale can be used to determine the optimum timing of implantation. Although the initial studies reported better survival rates when implantation was carried out in more stable patients (INTERMACS 4–7), at present barely 15% of all cases correspond to these scores.46 Patients with AHF may suffer exacerbations with characteristics and prognoses that differ from those of patients with de novo cardiogenic shock.47 When such exacerbation gives rise to cardiogenic shock, ECMO has been associated with a survival rate of 42% at one year, with lactate concentration and a higher SOFA score before implantation being the main determinants of mortality.48
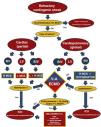
Initial strategyTo select the most adequate MCS strategy, we must identify the dysfunctional ventricle and determine whether there is pulmonary involvement or not. In the case of biventricular dysfunction with respiratory failure and established circulatory problems, ECMO is usually the first choice, seeking the best cannulation strategy49 and considering the frequent need for drainage of the left-side cavities.28,29,50 In patients with univentricular dysfunction, in the absence of respiratory disorders, the choice of MCS will depend on the degree of support or flow required, the reversibility of the disease that will condition the duration of the support, the access options, and even the availability of the technique in the center (Fig. 2).49,51
Algorithm for initial management of cardiogenic shock with mechanical circulatory support (modified from Lorusso R et al.).
MCS: mechanical circulatory support; RV: right ventricle; LV: left ventricle; BiV: biventricular; V--ECMO: venoarterial extracorporeal membrane oxygenation; LVEDP: left ventricle end-diastolic pressure; IABP: intra-aortic balloon pump.
α: according to availability and aim of therapy.
Management of patients with MCS includes both comprehensive care measures required by complex critically ill patients with multiorgan dysfunction,2 as well as monitoring and control of the implanted device.52,53 The use of protocols to ensure consistency of care and quality and safe patient management is very useful.54
CardiovascularContinuous electrocardiographic, invasive blood pressure (BP) and oxygen saturation (SatO2) monitoring is required. Advanced hemodynamic monitoring is performed using the pulmonary artery catheter, which provides information for the management of vasoactive drugs, MCS flow and weaning. Pulse wave analysis or thermodilution techniques are usually not reliable in this scenario, due to the continuous flow provided by MCS. Central venous pressure is a useful parameter, though its trend rather than an absolute value should be assessed, since the negative suction pressure may interfere with it. The ideal access for blood pressure monitoring is the right radial artery. This also allows monitoring of native gas exchange.55
Near-infrared spectroscopy (NIRS) monitoring allows us to observe regional changes (cerebral, cannulated extremity, etc.) in perfusion and oxygenation, detect and control the development of harlequin syndrome, and optimize blood pressure, oxygenation and/or assist flow in the event of decreased NIRS recordings.56,57
Frequent echocardiograms are required as part of the daily patient evaluation to assess cardiac evolution, the occurrence of complications (especially left ventricular dilatation in V-AECMO), and the possibility of weaning from the device.56,58
Hemodynamic management is based on adjustment of the MCS flow, fluid supply and vasoactive medication to the hemodynamic situation of the patient, seeking adequate tissue perfusion and oxygenation, and adequate venous SatO2, native cardiac index, mean blood pressure and lactate clearance.6
The use of inotropic agents is subject to controversy, and individualization of the dose for each situation is advised. Such drugs are useful for improving stroke function (ejection) and avoiding ventricular distension and blood stasis. However, the minimum dose should be administered, with the consideration of suspension as soon as possible.59
In the presence of low systemic vascular resistance, vasoconstrictors are required, with noradrenaline (NA) being the optimal pharmacological agent.60 In patients with refractory arterial hypotension, despite increasing noradrenaline doses, the use of vasopressin, which has been shown to be beneficial in various contexts, should be considered.61,62 While the scientific evidence is more limited, other potential options for refractory cases include intravenous methylene blue, angiotensin II, corticosteroids at stress doses, and metabolic coadjuvants (hydroxycobalamine, thiamine and ascorbic acid).63,64
RespiratoryThe recommendations are those commonly applicable to shock patients, employing “lung protection” strategies with low tidal volumes (<6 ml/kg), a low respiratory rate, optimal positive end-expiratory pressure (PEEP), the avoidance of high plateau pressures, and the minimum oxygen concentration necessary to prevent hyperoxygenation. It is recommended that invasive mechanical ventilation (IMV) be avoided whenever feasible.65
AnticoagulationWith the exception of IABP, mechanical circulatory support devices require anticoagulation at therapeutic doses. Anticoagulation should be monitored on a multimodal basis, and protocols should be adopted in each center. The most widely used drug is unfractionated heparin in continuous infusion, and monitoring is carried out based on different methods: ACT (activated clotting time) between 160–180 seconds, aPTT (activated partial thromboplastin time) ratio 1.5–2, and/or anti-Xa factor levels of 0.3−0.7 IU/mL. Direct thrombin inhibitors (bivalirudin and argatroban) are increasingly being used due to their safety and more stable action, and because they do not cause immune thrombocytopenia. In contrast to heparin, however, they have the inconvenience of lacking an antidote.66
In the event of bleeding, the anticoagulation dose should be adjusted. Alternatively, anticoagulation can be temporarily halted, while maintaining high pump flows to prevent thrombosis. In postsurgical patients, the start of anticoagulation can be deferred until the cessation of immediate bleeding and even until the removal of drains. The administration of low-dose heparin may be initiated, with subsequent dose increases contingent upon confirmation of the absence of bleeding.66 The occurrence of thrombotic events is monitored via ultrasound in both the cardiac cavities and in the cannulated vessels or components of the MCS device.58
InfectionsThese patients are vulnerable to infections associated with cannulation, invasive monitoring and mechanical ventilation, among other factors. Peri-procedure antibiotic prophylaxis is not recommended on a systematic basis – only in cases of emergent implantation where asepsis is lost; if a vessel with previous catheterization is used; in cases of prolonged cannulation due to complications; or in situations of central cannulation.67 Measures for preventing infections are required in patients subjected to MCS (Supplementary Material 4). The diagnosis of infection is complicated by the inflammatory response induced by the circulatory assist process itself, the under-evaluation of fever due to MCS-induced hypothermia, or the use of heat exchangers. Infection should be suspected in the presence of unexplained hemodynamic changes, the appearance of metabolic alterations (acidosis, hyper- or hypoglycemia, hypercapnia), or an increase in acute phase reactant.
RenalWorsening of kidney function is inherent to cardiogenic shock with MCS, since the kidneys are very susceptible to non-pulsatile flow, though other aspects such as previous disease conditions, systemic inflammatory response, dysregulation of the renin-angiotensin-aldosterone axis, increased intrathoracic/intraabdominal pressure, nephrotoxic drugs, severe hypercapnia/hypoxemia, hemolysis, hypercoagulability, etc., also exert an influence.
Close monitoring of kidney function and water balance and hemolysis parameter control is required. It is recommended that the degree of acute renal failure (ARF) during MCS be monitored based on scales such as the KDIGO.68
Neurological and rehabilitationNeurological monitoring should be performed on a multimodal basis, aimed at controlling sedoanalgesia and ensuring the early detection of complications. Periodic sedation vacations are indicated, with selection of the most adequate sedoanalgesic strategy. In patients subjected to ECMO, drugs that adhere to the circuit or to the membrane are to be avoided, with administration of the optimum dose.69
Daily neurological exploration is advised (pupil response with luminescence and/or pupillometry), with NIRS monitoring, bispectral index (BIS) monitoring, and the measurement of cerebral blood flow using transcranial Doppler exploration. A basal electroencephalogram (EEG) is recommended in some situations (after cardiac arrest, prolonged surgery or in cases of peri-implantation hemorrhage), and should be later repeated in the event of signs of intracranial hypertension, myoclonus or seizures, awakening difficulties, refractory shock and reduced BIS or NIRS readings. Other complementary tests (somatosensory evoked potentials, optic nerve sheath diameter, computed tomography [CT] or CT angiography) may be considered if complications are suspected.70
Delirium should be prevented using non-pharmacological measures or treating it early to avoid complications such as reintubation, or accidental decannulation or extubation.69
The rehabilitation of patients should commence at the time of MCS implantation, with an initial assessment of the potential for the patient to sit and/or walk, as well as engage in moderate physical exercise. This is of particular importance in heart transplantation or surgery candidates, as it has been demonstrated to improve the immediate postoperative period, reducing the mechanical ventilation time and the duration of stay in the Intensive Care Unit (ICU) and in hospital.71
Gastrointestinal and nutritionalLiver dysfunction is observed in 15% of the patients; hepatocellular function therefore must be monitored, with the adjustment of anticoagulation and hepatotoxic drugs.
Early and complete nutritional support is required, prioritizing enteral nutrition, with the calculation of the nutritional needs on an individualized basis. It is also important to provide vitamins, minerals, iron and other micronutrients, according to the individual patient requirements.72
ComplicationsThe complications observed in patients undergoing MCS can be attributed to a complex interplay of factors, encompassing both the underlying disease state and comorbidities (e.g., etiology of shock, immunosuppression, etc.) and the mechanical assist procedure itself (e.g., invasiveness, duration and type of support, use of anticoagulation, etc.).
InfectionsAs with any intravascular device, the use of MCS is associated with an increased risk of infections, including those at the insertion site, bacteremia and even endocarditis. These infections have the potential to be serious and to reduce patient survival.73 Prevention is essential in this respect. The implantation technique must be conducted in a sterile manner, and the insertion site must be cared for on a daily basis. Infections require optimal antibiotic therapy, including the selection of the appropriate drug, dosage and duration. In some cases, the removal of the assist device may be warranted, depending on the underlying cause and severity of the infection. These patients are at an increased risk of developing nosocomial infections, including ventilator-associated pneumonia and bacteremia, with incidence rates that vary depending on the series and device involved.73
VascularThe use of MCS has been linked to the development of complications at the insertion site, including hematomas, bleeding and localized pain and thrombosis. These adverse effects are attributed to alterations in vascular flow resulting from cannulation procedures. Other less frequent complications include stenosis following device removal and the formation of arteriovenous fistulas.
EmboligenicThe susceptibility to thrombus formation in cannulated or peri-cannulated blood vessels may lead to thromboembolic phenomena with occlusion of small vessels and in other areas during the circulatory support process and after weaning. There is also an increased susceptibility to thrombus formation in the cardiac cavities as a result of decreased blood flow or dilatation of the cavities.
Echocardiographic and vascular ultrasound monitoring is recommended to ensure early detection of thrombotic phenomena, optimize anticoagulation and avoid embolization. In the case of deep vein thrombosis (DVT) after weaning from ECMO, the occurrence of pulmonary thromboembolism (PTE) should be avoided by maintaining therapeutic anticoagulation, although its duration is controversial.58
Bleeding and hemolysisThe risk of both local (at the insertion site) and systemic bleeding increases with the use of MCS and its associated medications (anticoagulation, antiplatelet agents). Management of bleeding requires quantification and assessment of the consequences, determination of the etiology through complementary tests (endoscopy, CT angiography, fibrobronchoscopy), identification of promoting factors (thrombopenia, coagulation factor deficiency, acquired von Willebrand disease), and use of tests (thromboelastogram or rotational thromboelastometry [ROTEM]) to guide transfusion therapy.
The incidence of hemolysis is high, with variations depending on the type of assist device used. The determination of free hemoglobin is the most specific diagnostic test. Hemolysis can also be suspected from an increase in lactate dehydrogenase (LDH), a decrease in haptoglobin, anemization with an increase in bilirubin concentration and the presence of schistocytes, and hemoglobinuria. Treatment of the underlying cause (hypovolemia, malpositioning of the assist device, existence of thrombi in the MCS circuit) is indicated, with prevention of the associated consequences, which may include hydration and urine alkalinization with a urinary pH of > 6.5–7.
RenalAcute renal failure (ARF) is defined as an increase in creatinine levels following the initiation of extracorporeal membrane oxygenation (ECMO) therapy or the necessity for renal replacement therapy (RRT). It has a prevalence of 24–63 %. The management of this condition should be based on the use of diuretics in the case of volume overload, RRT, and monitoring of the plasma levels of drugs (antibiotics, etc.) with nephrotoxic effects.74 In the event of RRT usage, up to 50% of patients may undergo the procedure via catheterization or, when feasible, through the MCS circuit. In the case of ECMO, there are several connection options, each with its own set of advantages and disadvantages. It is essential to be aware of these factors in order to make an informed decision and adapt the choice to the specific circumstances of each case.
Left ventricular distensionIn some cases, blood cannot be adequately ejected into the pulmonary circulation in patients with CS and severely impaired left ventricular function. This situation may be exacerbated by an increase in afterload due to parallel MCS (V-AECMO) or the use of vasoactive drugs. A decrease in left ventricular output is observed, with an increase in LVEDP, an increase in pulmonary capillary pressure (PCP) with acute lung edema (ALE), and pulmonary hemorrhage. In turn, the inability to open the aortic valve favors the formation of intracardiac and aortic root thrombi. Lastly, worsened oxygenation, with the poorly oxygenated blood supply to the brain and coronary circulation, may lead to neurological deterioration and greater myocardial dysfunction.75
Optimal LV unloading is essential for myocardial recovery in patients with CS and is one of the advantages of certain devices (Impella®, Centrimag®, etc.) over V-AECMO. Detection of impaired unloading is based on ultrasound parameters (distended LV, severe mitral valve regurgitation), clinical variables (ALE, arrhythmias) and/or hemodynamic parameters (pulse pressure < 10 mmHg, PCP > 18 mmHg), and requires early management.76
Stepwise management is required, starting with non-invasive measures (adjustment of ECMO flow, use of inotropes, reduction of vasoactive agents, and even the administration of vasodilators), followed by non-active invasive measures (IABP) and finally active interventions (Impella CP®, percutaneous septostomy, apical LV drainage) if the previous measures prove insufficient.76 The identification of the optimal threshold for unloading and the best method remain controversial, and the choice is usually based on the experience and capacity of each center. It should be noted that prophylactic and systematic unloading is not indicated.77
Respiratory failureThe cause of respiratory failure in patients undergoing MCS can be multifactorial (cardiogenic pulmonary edema, pulmonary embolism, mechanical ventilation-related injury, acute respiratory distress syndrome secondary to comorbidities) and management is aimed at treating these factors. In the case of MCS with Centrimag® type devices, a membrane can be placed in the circuit, and in the case of Impella CP® it would be necessary to escalate to V-AECMO or even to V-VECMO.60
Harlequin or North/South syndromeThis syndrome is typically seen in patients on femoro-femoral V-AECMO, with myocardial recovery and established lung injury. It is characterized by a difference in oxygenation between the upper half of the body (hypoxemic) and the lower half of the body (normal oxygenation), and can have important consequences for organ perfusion (especially the brain). The prevalence varies from 8.8% to 13.3%, depending on the literature source, and can be detected by cerebral NIRS or differential pulse oximetry. The syndrome can be managed by tapering the pump flow until the cause is corrected and/or optimizing mechanical ventilation. In refractory cases, a Y-connection return cannula can be added to the jugular vein (V-VA) or axillary artery (V-AA), the type of peripheral configuration can be changed (femoro-femoral to femoro-axillary), or a switch can be made from peripheral to central.78
Right ventricular failureRight ventricular failure in patients with ventricular assist devices/MCS is a diagnostic and therapeutic challenge.79 If failure is present from the onset of cardiogenic shock (myocarditis, graft rejection, right ventricular infarction), V-AECMO would generally be indicated because it provides biventricular support. In cases of CS with left MCS, failure may occur or worsen after implantation due to increased right ventricular preload. Another cause of right-sided dysfunction is thrombosis or pulmonary thromboembolism. Management includes hemodynamic and echocardiographic monitoring, and the administration of inotropes and/or pulmonary vasodilators (inhaled nitric oxide) may be beneficial. In refractory cases, the implantation of a right MCS device (Impella RP®, etc.) or conversion to global MCS may be considered.80
De-escalationThe goal of MCS for CS is to stabilize the patient until the underlying cause is corrected or definitive treatment is initiated. At this point weaning or de-escalation of the device should be considered.60 Weaning requires the evaluation of several parameters that indicate a greater likelihood of success, and although each center usually has its own protocol, a minimum clinical condition and a prior weaning trial are included in all cases.80 If the minimum clinical-hemodynamic conditions for MCS weaning are not met, escalation to another device of longer duration should be considered, or even cardiac transplantation may be indicated, provided there are no contraindications. In the case of multiorgan dysfunction (>2 organs) and/or no chance of clinical recovery, the adjustment of measures or the withdrawal of the MCS should be evaluated before considering the possibility of organ and/or tissue donation (Fig. 3).
Algorithm for mechanical circulatory support withdrawal in patients with cardiogenic shock.
CS: cardiogenic shock; MOD: multiorgan dysfunction; MBP: mean blood pressure; SBP: systolic blood pressure; DP: differential pressure; PCP: pulmonary capillary pressure; CVP: central venous pressure; CI: cardiac index; CW: cardiac work; PAPi: pulmonary artery pulsatility index; SvO2: venous oxygen saturation; LVEF: left ventricular ejection fraction; FS: fractional shortening; TAPSE: tricuspid annular plane systolic excursion; VTI: velocity-time integral; LVOT: left ventricular outflow tract; LV: left ventricle; RV: right ventricle; MCS: mechanical circulatory support; IABP: intra-aortic balloon pump; V-AECMO: venoarterial extracorporeal membrane oxygenation; TxC: cardiac transplantation; LVAD: long-term left ventricular assist device; LLST: limitation of life support therapy.
*: If ECMELLA, first suspend ECMO, and then Impella CP®.
Mechanical circulatory support is an essential therapeutic tool, and despite the limited evidence, it has represented an important step forward in the management of refractory cardiogenic shock. The use of MCS requires advanced hemodynamic and clinical evaluation to determine the best time and type of circulatory support. The clinical course of these patients is variable and not without complications, requiring multidisciplinary assessment and constant reevaluation of the use and indication of the circulatory support. Further studies are needed to clarify various aspects of the use of MCS in patients with cardiogenic shock, with intensive care being a key supportive element to be evaluated and developed.
Author contributionsLMV, AAG, JMGJ, JLPV and MPFG designed the study, performed the literature review and wrote the manuscript. LMV, JMGJ, AAG, JLPV and MPFG participated in preparing the tables and figures. LMV, MPFG, JLPV, JMGJ and AAG reviewed the final version of the manuscript. All authors read and approved the final manuscript.