Hyperleukocytosis can cause respiratory failure due to pulmonary leukostasis. Additionally, gas tests can confirm the presence of pseudo-hypoxemia and the patient may end up receiving unnecessary treatments if undiagnosed.
We hereby present the case of a 25-year-old male with chronic myeloid leukemia (CML) diagnosed 10 years ago and with poor adherence to medical treatment. The patient was admitted to the ER with asthenia and adynamia of 2-week duration complicated with dyspnea over the last 24hours. The patient remained apyretic and lucid during the physical examination with polypnea at 29rpm, diffuse crepitant rattle, and pulse oximetry (SpO2) values no less than 90% with oxygen therapy through a low-flow face mask. Hemodynamically stable. Distended abdomen, hepatomegaly, and grade V splenomegaly. The blood test sample showed the following values: hemoglobin, 3.6g/dL; leukocytosis, 688000cells/μL; platelets 321000cells/μL. Peripheral blood swab sample consistent with CML in chronic stage. The chest X-ray confirmed the presence of right para hilar region discreet opacity. The patient was admitted to the intensive care unit (ICU) with low respiratory work and non-invasive mechanical ventilatory support (MVS) was initiated. Thirty (30) minutes later, the patient started feeling confused, polypneic (30–35rpm) and with thoracoabdominal asynchrony. Even though the SpO2 was 95%, the arterial-blood gas test conducted (Radiometer ABL800 FLEX, Denmark) confirmed the presence of severe respiratory failure: PaO2=50mmHg, PaCO2=38mmHg, values of arterial blood oxygen saturation (SaO2) of 86% (fraction of inspired oxygen=0.6, pressure support ventilation=8cmH2O, positive end-expiratory pressure=9cmH2O). We proceeded with orotracheal intubation and invasive MVS. On suspicion of leukostasis-induced respiratory failure, cytoreductive therapy was started with the administration of hydroxyurea and leukapheresis associated with the molecular therapy of CML with dasatinib. Red-cell concentrates were transfused to solve the anemia.
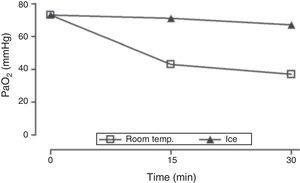
Upon admission to the ICU, there was a clear discrepancy between the pulse oximetry levels (SpO2≥95%) and the arterial-blood gas test levels (SaO2<90%, PaO2≤50mmHg). The levels of methemoglobin (0%) and carboxyhemoglobin (1.4%), and p50 (25.9mmHg) were within normal ranges. Due to severe hyperleukocytosis, pseudo-hypoxemia or spurious hypoxemia were considered and discussed due to an excessive consumption of oxygen following the acquisition of the specimen. To put this hypothesis to the test, arterial-blood gas tests were performed simultaneously from specimens kept at room temperature or in ice (in an effort to slow metabolic consumption) for 30min (leukocytosis, 688000cells/μL when the sample was collected). Whereas the PaO2 and SaO2 levels remained relatively constant in the blood that was preserved in ice, they dropped dramatically at room temperature which is consistent with the pseudo-hypoxemia hypothesis (Fig. 1). Both the ice preservation of the sample and its quick processing minimized this preanalytical mistake in further decisions.
PaO2 measurements in arterial blood samples kept at room temperature or in ice. Both specimens were acquired at the same time (FiO2 1, leukocytosis, 688000cells/μL). One arterial-blood gas test was performed immediately after the collection of the specimen and two more at 15 and 30minutes. FiO2, fraction of inspired oxygen; PaO2, partial pressure of oxygen in arterial blood; room temp., room temperature.
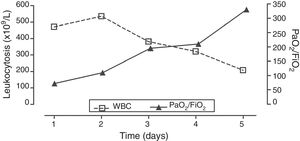
The patient received six session of leukapheresis associated with treatment with hydroxyurea showing favorable progression with gradual reduction of leukocytosis and gradual improvement of gas exchange (Fig. 2). The patient did not show any other infectious complications, organ dysfunctions or elements suggestive of tumor lysis syndrome (TLS) or disseminated intravascular coagulation (DIC). The MVS was removed six days after admission and the patient remained lucid and free from neurological alterations. After hospitalization in the internal medicine ward the patient was discharged from the hospital, remained asymptomatic, and with a significant improvement of his hematologic alterations (hemoglobin, 7.7g/dL; leukocytosis, 60700cells/μL and platelets, 213000cells/μL).
Hyperleukocytosis is defined as white blood cell counts (WBC) above 100000cells/μL and is associated with an increase of morbimortality in patients with different types of leukemia.1 Its main clinical manifestations are leukostasis-related (organ infiltration by leukemic cells), TLS and/or DIC.2 At pulmonary level, leukostasis can cause respiratory failure due to microvascular obstruction by altering the ventilation/perfusion relationship and the diffusion of gases.3 Regardless of this, the arterial-blood gas tests run in patients with hyperleukocytosis can confirm the presence of pseudo-hypoxemia or spurious hypoxemia, that is, in patients who don’t really show decreased PaO2 levels. The main explanation to this phenomenon was given back in 1979 and has to do with the high metabolic consumption of blood cells following the collection of the sample.4,5 As a matter of fact, its magnitude is somehow associated with the WBC.6 If not properly estimated it can lead to errors of interpretation through diagnoses of respiratory failure in patients who don’t have this condition or by overestimating the severity of this respiratory condition.7 This can have a negative impact on the patients when PaO2 is considered as the main criterion to define the need for diagnostic or therapeutic procedures such as MVS. Therefore, the physician should be aware of this phenomenon to be able to recognize it, being the SpO2 and SaO2 discrepancy a key observation here.
Several authors have reported that keeping the sample in ice and processing it immediately can minimize this error.4,5,8 However, this finding was not consistent with other studies that confirmed a certain reduction of PaO2 yet despite the icing of the blood specimen.6 On the other hand, the addition of sodium fluoride or potassium cyanide to the specimen can inhibit metabolic consumption and stop the development of spurious hypoxemia.5,9 In this type of patients pulse oximetry follow-up is especially relevant but only as long as the presence of methemoglobinemia and carboxyhemoglobinemia is ruled out since it may affect the measurement of SpO2.7
In sum, hyperleukocytosis can cause true and/or spurious hypoxemia. It is essential to keep a high index of suspicion here and rule-out the existence of the latter. If undiagnosed it can condition the prescription of unnecessary and potentially dangerous therapies for patients.
Please cite this article as: Angulo M, Machado D, Larrosa L, Biestro A. Hipoxemia real y espuria en un paciente con hiperleucocitosis extrema. Med Intensiva. 2019;43:435–436.