During the new pandemic caused by SARS-CoV-2, there is short knowledge regarding the management of different disease areas, such as coagulopathy and interpretation of D-dimer levels, its association with disseminated intravascular coagulation (DIC) and controversy about the benefit of anticoagulation. Thus, a systematic review has been performed to define the role of D-dimer in the disease, the prevalence of DIC and the usefulness of anticoagulant treatment in these patients. A literature search was performed to analyze the studies of COVID-19 patients. Four recommendations were drawn based on expert opinion and scientific knowledge, according to the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. The present review suggests the presence of higher levels of D-dimer in those with worse prognosis, there may be an overdiagnosis of DIC in the course of the disease and there is no evidence on the benefit of starting anticoagulant treatment based only on isolated laboratory data.
Durante la nueva pandemia causada por SARS-CoV-2, existe poca evidencia en relación a varios aspectos de la enfermedad, como es el caso de la coagulopatía e interpretación de los niveles de dímero D, su asociación con coagulación intravascular diseminada (CID) y controversia en cuanto al beneficio de la anticoagulación. Por ello, se ha realizado una revisión sistemática para definir el rol del dímero D en la enfermedad, la prevalencia y valor pronóstico de la CID y la utilidad del tratamiento anticoagulante en dichos pacientes. Se realizó una búsqueda bibliográfica y análisis de la literatura sobre pacientes con COVID-19. Se elaboraron cuatro recomendaciones basadas en la opinión de expertos y en el conocimiento científico, según el sistema Grading of Recommendations Assesment, Development and Evaluation (GRADE). La presente revisión en pacientes con COVID-19 sugiere la presencia de mayores niveles de dímero D en aquellos con peor pronóstico, que puede haber un sobrediagnóstico de CID en el curso de la enfermedad y que no existe evidencia sobre el beneficio de iniciar tratamiento anticoagulante basándose únicamente en datos aislados de laboratorio.
Since December 2019, with the appearance of the new SARS-CoV-2 coronavirus and the subsequent pandemic1, many seriously ill patients have been admitted to hospitals and Intensive Care Units (ICUs). This has not only represented an important burden for healthcare systems but has also implied great mortality caused by the new disease condition called COVID-192.
In this context of uncertainty, and in the face of a lack of specific treatment for the disease3, healthcare professionals have had to accept the use of therapies based on scant scientific evidence. The indication of early anticoagulation in COVID-19 is an example of this situation.
Recent studies4,5 indicate that mortality due to serious SARS-CoV-2 disease is often associated to the presence of coagulopathy and disseminated intravascular coagulation (DIC), and that high levels of D-dimer (DD), in excess of 1 μg/mL, are associated to increased mortality6. Different publications7,8, as well as a number of local protocols, propose the adoption of different empirical anticoagulation or thromboprophylactic measures involving high doses of low molecular weight heparin (LMWH) based only on the DD level, in the absence of any clear scientific evidence supporting such treatment – with the risk this may pose for our critical patients.
The present systematic literature review was carried out with the purpose of answering four questions of clinical interest in PICO (patient-intervention-comparison-outcome) format:
- 1
Are DD levels associated to the prognosis of patients with COVID-19?
- 2
Is DIC in patients with COVID-19 associated to increased mortality?
- 3
Does the administration of empirical anticoagulation in patients with COVID-19 and elevated DD improve the prognosis?
- 4
Should we provide anticoagulation in patients with COVID-19 and associated DIC?
The Department of Intensive Care Medicine of Hospital Joan XXIII (Tarragona, Spain) carried out this project in April 2020 with a working group of four clinical investigators, with the aim of reviewing the scientific evidence and of developing recommendations of particular interest for the daily clinical management of patients with COVID-19 disease.
Literature searchThe different literature sources were reviewed by two investigators on an independent basis. For the drawing of conclusions, a search was made of articles published from December 2019 to 23 April 2020 in the following databases: Medline (PubMed), Cochrane Library and ScienceDirect. The keywords used individually or in combination for the search were «COVID-19», «coronavirus», «D-dimer», «disseminated intravascular coagulation» and «anticoagulation».
Types of studiesWith regard to the inclusion criteria, and considering the current lack of knowledge about this serious new infectious disease, we reviewed meta-analyses, observational studies, review articles and clinical guides referred to adult patients hospitalized due to COVID-19 disease. Assessment of the quality of evidence was based only on the original articles.
With regard to the exclusion criteria, we excluded studies of pediatric patients, articles published in languages other than English or Spanish, and studies in animals.
Data extraction and analysisInformation was extracted from the publications referred to study design and period, clinical variables, statistical analysis, risk factors and possible bias. Lastly, all the articles were reviewed by the other two clinicians of the working group, with extensive research experience.
Development of the recommendationsWe established four questions of clinical interest in PICO format for the drawing of conclusions, and quality of evidence was assessed based on the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system9. In the event of disagreement, consensus was reached among all the working group members.
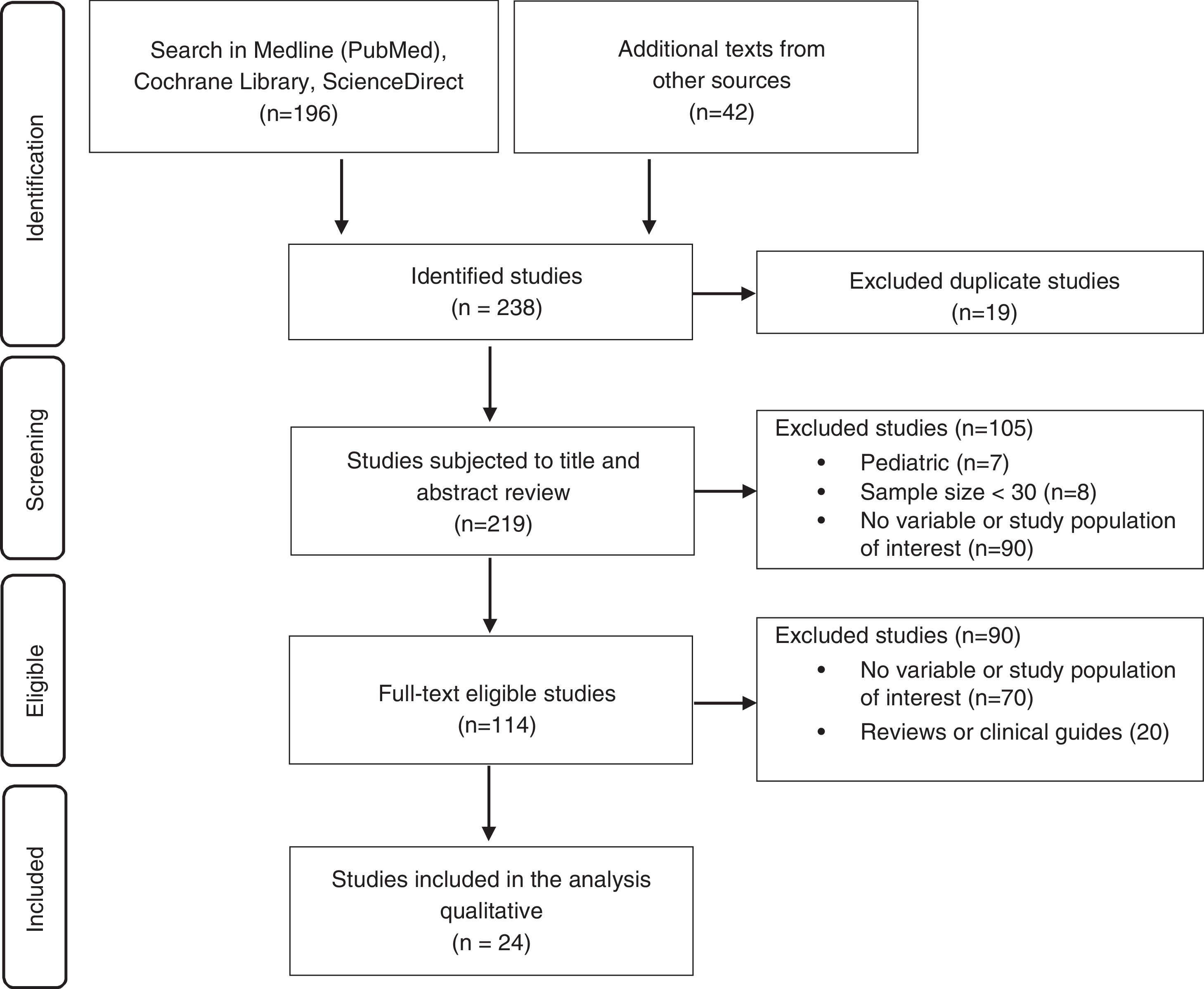
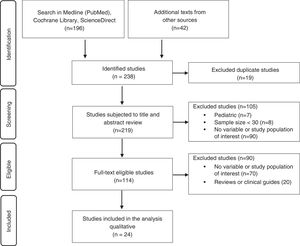
ResultsThe literature search yielded 238 studies, of which 24 were finally included for qualitative analysis and the development of recommendations (Fig. 1).
PICO 1. Are DD levels associated to the prognosis of patients with COVID-19?Conclusion 1: D-dimer in patients with COVID-19 is associated to increased severity, progression of the disease, acute respiratory distress syndrome (ARDS) and death (quality of evidence: low).
Recommendation 1: It is advisable to monitor the D-dimer levels upon admission and every 24–48 h as a tool for evaluation of the prognosis and progression of the disease (strength of recommendation: weakly in favor).
We are increasingly learning more about the relatively frequent coagulation disorders seen in patients with COVID-19, in particular in the more serious cases. Infection due to SARS-CoV-2 appears to induce a blood hypercoagulability state, since there have been reports of coagulation disorders and elevated DD in a large proportion of patients10, with gradual increments related to progression of the disease11. All this could be explained by excessive coagulation cascade and platelet activation - with the consequent formation of intraalveolar fibrin deposits (or systematic fibrin microthrombi). These findings are more characteristic of patients with COVID-19 and ARDS. This is due to the prothrombotic response, which attempts to avoid diffuse alveolar damage and prevent the infectious agent from penetrating into the bloodstream. However, this may give rise to the formation of pulmonary microthrombi, with deleterious effects upon the patient course12. Nevertheless, certain discrepancies may arise from the multiple and sometimes opposite actions of thrombosis on the pulmonary epithelium following sepsis, since on one hand mild lung thrombosis favors repair of the damaged endothelium, while on the other severe thrombosis causes hypoxia and produces pulmonary endothelial damage13.
D-dimer is a fibrin degradation product generated from three reactions: the conversion of fibrinogen into fibrin mediated by thrombin, fibrin reticulation mediated by activated factor XIII, and fibrin degradation mediated by plasmin14. This means that the levels depend on both coagulation and the activation of fibrinolysis. D-dimer has high sensitivity in the presence of thromboembolic disease, but specificity is poor, since DD is also elevated in other situations. Sepsis (in the same way as ARDS)15 is characterized by marked inhibition of fibrinolysis; the DD levels in septic patients therefore probably do not adequately reflect the degree of fibrin formation16,17. In this respect, it may be suggested that the isolated use of DD for the diagnosis of DIC may lead to error. Likewise, DD has been previously studied, with the observation of a high prevalence of elevated DD levels in cases of community-acquired pneumonia18, severe sepsis or septic shock19 - which moreover evidences its role as a predictor of mortality in sepsis20.
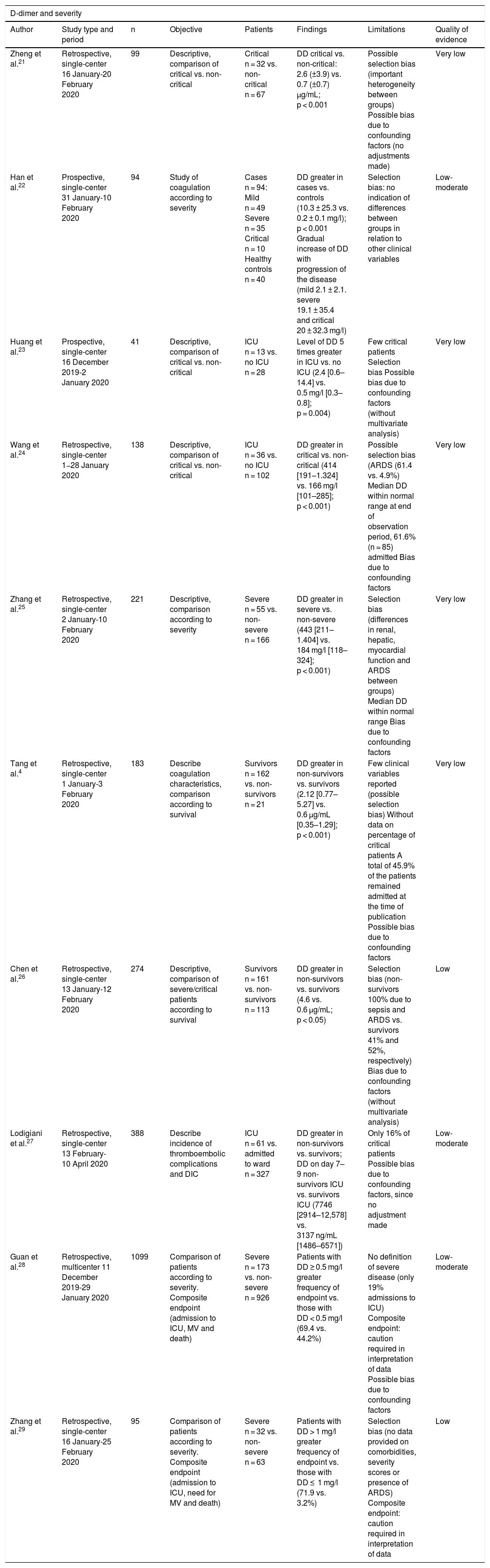
Non-adjusted observational studiesBased on the recent literature, the incidence of DD elevation in patients with SARS-CoV-2 infection is about 46.4%, and is even higher in cases of severe disease (59.6%)12. Different studies have documented significant DD elevation in patients with severe COVID-19 disease compared with individuals presenting milder symptoms21 and healthy subjects22 (Table 1). In this latter study22, a gradual increase in DD was moreover observed with progression of the disease – thus suggesting a possible association of DD to such progression. These findings are consistent with those published by Huang et al.23, who reported that the DD levels upon admission among patients with severe COVID-19 disease was up to 5-fold higher than among those not requiring admission to the ICU. However, it must be noted that the number of patients in the ICU was only 13, and of these, only two required invasive mechanical ventilation. Another two studies24,25also recorded higher DD levels upon admission to the ICU versus non-critical patients, though the median values were within the normal laboratory range, and both studies were characterized by important methodological limitations.
Non-adjusted observational studies related to D-dimer levels and the prognosis of patients with COVID-19 disease.
| D-dimer and severity | |||||||
|---|---|---|---|---|---|---|---|
| Author | Study type and period | n | Objective | Patients | Findings | Limitations | Quality of evidence |
| Zheng et al.21 | Retrospective, single-center 16 January-20 February 2020 | 99 | Descriptive, comparison of critical vs. non-critical | Critical n = 32 vs. non-critical n = 67 | DD critical vs. non-critical: 2.6 (±3.9) vs. 0.7 (±0.7) μg/mL; p < 0.001 | Possible selection bias (important heterogeneity between groups) Possible bias due to confounding factors (no adjustments made) | Very low |
| Han et al.22 | Prospective, single-center 31 January-10 February 2020 | 94 | Study of coagulation according to severity | Cases n = 94: Mild n = 49 Severe n = 35 Critical n = 10 Healthy controls n = 40 | DD greater in cases vs. controls (10.3 ± 25.3 vs. 0.2 ± 0.1 mg/l); p < 0.001 Gradual increase of DD with progression of the disease (mild 2.1 ± 2.1. severe 19.1 ± 35.4 and critical 20 ± 32.3 mg/l) | Selection bias: no indication of differences between groups in relation to other clinical variables | Low-moderate |
| Huang et al.23 | Prospective, single-center 16 December 2019-2 January 2020 | 41 | Descriptive, comparison of critical vs. non-critical | ICU n = 13 vs. no ICU n = 28 | Level of DD 5 times greater in ICU vs. no ICU (2.4 [0.6–14.4] vs. 0.5 mg/l [0.3–0.8]; p = 0.004) | Few critical patients Selection bias Possible bias due to confounding factors (without multivariate analysis) | Very low |
| Wang et al.24 | Retrospective, single-center 1−28 January 2020 | 138 | Descriptive, comparison of critical vs. non-critical | ICU n = 36 vs. no ICU n = 102 | DD greater in critical vs. non-critical (414 [191–1.324] vs. 166 mg/l [101–285]; p < 0.001) | Possible selection bias (ARDS (61.4 vs. 4.9%) Median DD within normal range at end of observation period, 61.6% (n = 85) admitted Bias due to confounding factors | Very low |
| Zhang et al.25 | Retrospective, single-center 2 January-10 February 2020 | 221 | Descriptive, comparison according to severity | Severe n = 55 vs. non-severe n = 166 | DD greater in severe vs. non-severe (443 [211–1.404] vs. 184 mg/l [118–324]; p < 0.001) | Selection bias (differences in renal, hepatic, myocardial function and ARDS between groups) Median DD within normal range Bias due to confounding factors | Very low |
| Tang et al.4 | Retrospective, single-center 1 January-3 February 2020 | 183 | Describe coagulation characteristics, comparison according to survival | Survivors n = 162 vs. non-survivors n = 21 | DD greater in non-survivors vs. survivors (2.12 [0.77–5.27] vs. 0.6 μg/mL [0.35–1.29]; p < 0.001) | Few clinical variables reported (possible selection bias) Without data on percentage of critical patients A total of 45.9% of the patients remained admitted at the time of publication Possible bias due to confounding factors | Very low |
| Chen et al.26 | Retrospective, single-center 13 January-12 February 2020 | 274 | Descriptive, comparison of severe/critical patients according to survival | Survivors n = 161 vs. non-survivors n = 113 | DD greater in non-survivors vs. survivors (4.6 vs. 0.6 μg/mL; p < 0.05) | Selection bias (non-survivors 100% due to sepsis and ARDS vs. survivors 41% and 52%, respectively) Bias due to confounding factors (without multivariate analysis) | Low |
| Lodigiani et al.27 | Retrospective, single-center 13 February-10 April 2020 | 388 | Describe incidence of thromboembolic complications and DIC | ICU n = 61 vs. admitted to ward n = 327 | DD greater in non-survivors vs. survivors; DD on day 7–9 non-survivors ICU vs. survivors ICU (7746 [2914–12,578] vs. 3137 ng/mL [1486–6571]) | Only 16% of critical patients Possible bias due to confounding factors, since no adjustment made | Low-moderate |
| Guan et al.28 | Retrospective, multicenter 11 December 2019-29 January 2020 | 1099 | Comparison of patients according to severity. Composite endpoint (admission to ICU, MV and death) | Severe n = 173 vs. non-severe n = 926 | Patients with DD ≥ 0.5 mg/l greater frequency of endpoint vs. those with DD < 0.5 mg/l (69.4 vs. 44.2%) | No definition of severe disease (only 19% admissions to ICU) Composite endpoint: caution required in interpretation of data Possible bias due to confounding factors | Low-moderate |
| Zhang et al.29 | Retrospective, single-center 16 January-25 February 2020 | 95 | Comparison of patients according to severity. Composite endpoint (admission to ICU, need for MV and death) | Severe n = 32 vs. non-severe n = 63 | Patients with DD > 1 mg/l greater frequency of endpoint vs. those with DD ≤ 1 mg/l (71.9 vs. 3.2%) | Selection bias (no data provided on comorbidities, severity scores or presence of ARDS) Composite endpoint: caution required in interpretation of data | Low |
DIC: disseminated intravascular coagulation; DD: D-dimer; ARDS: acute respiratory distress syndrome; ICU: Intensive Care Unit; MV: mechanical ventilation.
Different studies have also reported higher DD levels among non-survivors versus survivors4,26,27. However, it must be noted that these studies were affected by confounding factors due to a lack of outcome adjustment in the different populations involved. Furthermore, in one study4, almost half of the patients were still admitted at the time of publication; the final data therefore could vary significantly, and the results consequently must be interpreted with caution. Another two studies28,29 found DD elevation to be more frequent in patients with increased disease severity and mortality. The multicenter study published by Guan et al.28 involved a large number of patients. However, the definition of severe disease was not specified. The main limitation of these two studies is that they used a composite endpoint with variables of different impact; caution is therefore required in interpreting the data, since the three end events could be associated to multiple confounding factors, such as severity upon admission, comorbidities or the presence of ARDS.
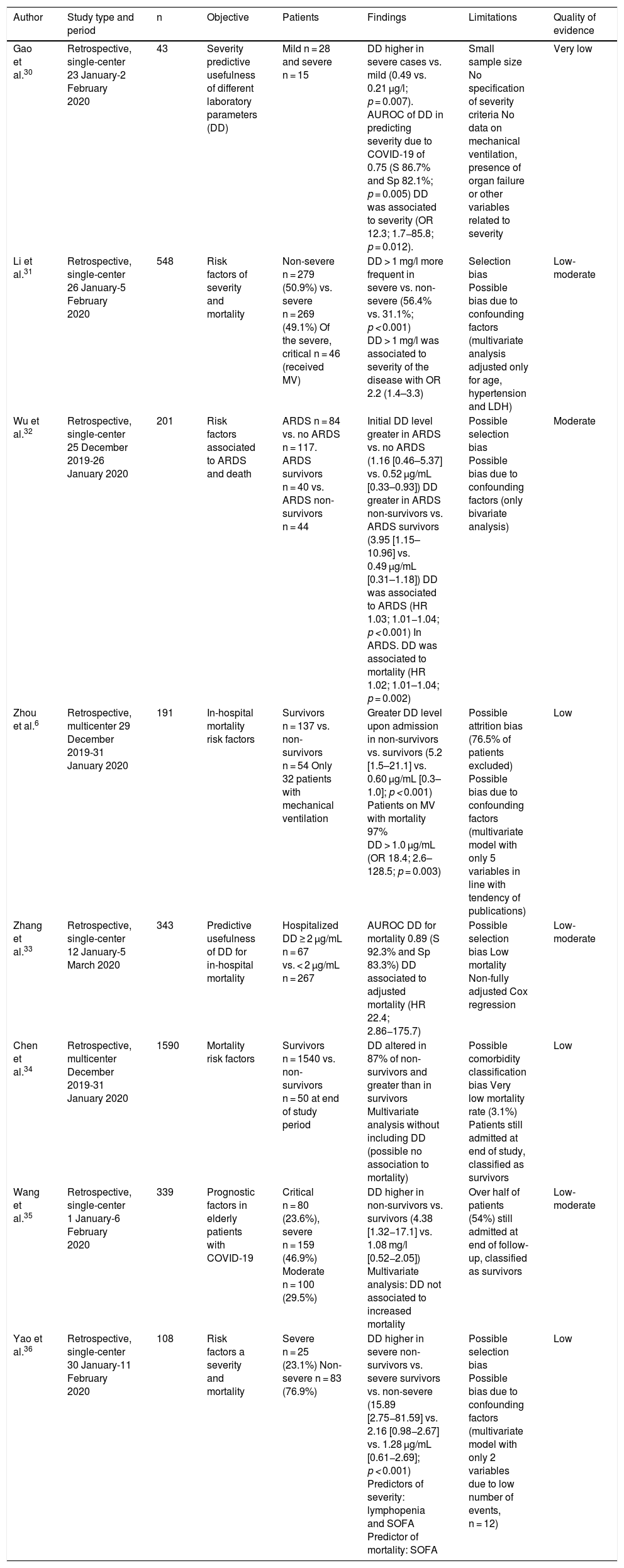
Adjusted observational studiesMultiple studies have included a statistical analysis adjusted for different factors that could explain the association between elevated DD and a poorer patient prognosis (Table 2). Gao et al.30 examined the predictive usefulness of DD based on the area under the receiver operating characteristic (AUROC) curve for the diagnosis of severe COVID-19 in 43 patients classified as having mild or severe disease. D-dimer level was associated to increased severity (odds ratio [OR] 12.3; 95% confidence interval [95%CI]: 1.71–85.8), with an AUROC for predicting the severity of COVID-19 pneumonia of 0.75. However, the model only included the levels of DD and interleukin 6. Similarly, DD > 1 mg/l has also been associated to increased COVID-19 disease severity, with an OR of 2.2 (95%CI: 1.4–3.3)31.
Adjusted observational studies related to D-dimer levels as independent predictor in patients with COVID-19 disease.
| Author | Study type and period | n | Objective | Patients | Findings | Limitations | Quality of evidence |
|---|---|---|---|---|---|---|---|
| Gao et al.30 | Retrospective, single-center 23 January-2 February 2020 | 43 | Severity predictive usefulness of different laboratory parameters (DD) | Mild n = 28 and severe n = 15 | DD higher in severe cases vs. mild (0.49 vs. 0.21 μg/l; p = 0.007). AUROC of DD in predicting severity due to COVID-19 of 0.75 (S 86.7% and Sp 82.1%; p = 0.005) DD was associated to severity (OR 12.3; 1.7−85.8; p = 0.012). | Small sample size No specification of severity criteria No data on mechanical ventilation, presence of organ failure or other variables related to severity | Very low |
| Li et al.31 | Retrospective, single-center 26 January-5 February 2020 | 548 | Risk factors of severity and mortality | Non-severe n = 279 (50.9%) vs. severe n = 269 (49.1%) Of the severe, critical n = 46 (received MV) | DD > 1 mg/l more frequent in severe vs. non-severe (56.4% vs. 31.1%; p < 0.001) DD > 1 mg/l was associated to severity of the disease with OR 2.2 (1.4–3.3) | Selection bias Possible bias due to confounding factors (multivariate analysis adjusted only for age, hypertension and LDH) | Low-moderate |
| Wu et al.32 | Retrospective, single-center 25 December 2019-26 January 2020 | 201 | Risk factors associated to ARDS and death | ARDS n = 84 vs. no ARDS n = 117. ARDS survivors n = 40 vs. ARDS non-survivors n = 44 | Initial DD level greater in ARDS vs. no ARDS (1.16 [0.46–5.37] vs. 0.52 μg/mL [0.33–0.93]) DD greater in ARDS non-survivors vs. ARDS survivors (3.95 [1.15–10.96] vs. 0.49 μg/mL [0.31–1.18]) DD was associated to ARDS (HR 1.03; 1.01−1.04; p < 0.001) In ARDS. DD was associated to mortality (HR 1.02; 1.01–1.04; p = 0.002) | Possible selection bias Possible bias due to confounding factors (only bivariate analysis) | Moderate |
| Zhou et al.6 | Retrospective, multicenter 29 December 2019-31 January 2020 | 191 | In-hospital mortality risk factors | Survivors n = 137 vs. non-survivors n = 54 Only 32 patients with mechanical ventilation | Greater DD level upon admission in non-survivors vs. survivors (5.2 [1.5–21.1] vs. 0.60 μg/mL [0.3–1.0]; p < 0.001) Patients on MV with mortality 97% DD > 1.0 μg/mL (OR 18.4; 2.6–128.5; p = 0.003) | Possible attrition bias (76.5% of patients excluded) Possible bias due to confounding factors (multivariate model with only 5 variables in line with tendency of publications) | Low |
| Zhang et al.33 | Retrospective, single-center 12 January-5 March 2020 | 343 | Predictive usefulness of DD for in-hospital mortality | Hospitalized DD ≥ 2 μg/mL n = 67 vs. < 2 μg/mL n = 267 | AUROC DD for mortality 0.89 (S 92.3% and Sp 83.3%) DD associated to adjusted mortality (HR 22.4; 2.86−175.7) | Possible selection bias Low mortality Non-fully adjusted Cox regression | Low-moderate |
| Chen et al.34 | Retrospective, multicenter December 2019-31 January 2020 | 1590 | Mortality risk factors | Survivors n = 1540 vs. non-survivors n = 50 at end of study period | DD altered in 87% of non-survivors and greater than in survivors Multivariate analysis without including DD (possible no association to mortality) | Possible comorbidity classification bias Very low mortality rate (3.1%) Patients still admitted at end of study, classified as survivors | Low |
| Wang et al.35 | Retrospective, single-center 1 January-6 February 2020 | 339 | Prognostic factors in elderly patients with COVID-19 | Critical n = 80 (23.6%), severe n = 159 (46.9%) Moderate n = 100 (29.5%) | DD higher in non-survivors vs. survivors (4.38 [1.32−17.1] vs. 1.08 mg/l [0.52−2.05]) Multivariate analysis: DD not associated to increased mortality | Over half of patients (54%) still admitted at end of follow-up, classified as survivors | Low-moderate |
| Yao et al.36 | Retrospective, single-center 30 January-11 February 2020 | 108 | Risk factors a severity and mortality | Severe n = 25 (23.1%) Non-severe n = 83 (76.9%) | DD higher in severe non-survivors vs. severe survivors vs. non-severe (15.89 [2.75−81.59] vs. 2.16 [0.98−2.67] vs. 1.28 μg/mL [0.61−2.69]; p < 0.001) Predictors of severity: lymphopenia and SOFA Predictor of mortality: SOFA | Possible selection bias Possible bias due to confounding factors (multivariate model with only 2 variables due to low number of events, n = 12) | Low |
APACHE: Acute Physiology and Chronic Health Evaluation; AUROC: area under the receiver operating characteristic curve; DD: D-dimer; E: specificity; HR: hazard ratio; LDH: lactate dehydrogenase; OR: odds ratio; S: sensitivity; ARDS: acute respiratory distress syndrome; SOFA: Sequential Organ Failure Assessment; ICU: Intensive Care Unit; MV: mechanical ventilation.
Wu et al.32 studied the association of DD to ARDS. In a cohort of 201 patients with COVID-19, they found the initial DD levels to be higher in patients with ARDS than in those without ARDS. Likewise, DD was seen to be higher among those patients with ARDS who died than those who survived. In the bivariate analysis, the authors found DD to be associated to ARDS (hazard ratio [HR] 1.03; 95%CI: 1.01–1.04; p < 0.001) and mortality in patients with ARDS (HR 1.02; 95%CI: 1.01–1.04; p = 0.002), though without taking into account other confounding factors. It should be mentioned that 41.8% of the population developed ARDS, though only one of every four patients was admitted to the ICU, and only 2.5% required mechanical ventilation – this possibly reflecting a population with ARDS different from that seen in our ICUs.
In the multicenter study published by Zhou et al.6, involving 191 patients admitted to hospital due to COVID-19, DD > 1.0 μg/mL was seen to be strongly associated to increased mortality (OR 18.4; 95%CI: 2.6–128.5; p = 0.003). However, significant differences were observed in many other variables that were not included in the multivariate model. The authors acknowledged having randomly selected the 5 variables for inclusion in the model, in line with the tendencies of the studies published to date. This complicates adequate interpretation of the data. Zhang et al.33 conducted a more rigorous study to define the usefulness of DD based on the AUROC for predicting in-hospital mortality in patients with COVID-19. The authors identified a cut-off point of 2 μg/mL for predicting increased mortality, with an AUROC of 0.89. After adjusting for possible confounding factors (age, gender and comorbidities), they found a high DD level to be associated to increased mortality (HR 22.4; 95%CI: 2.86–175.7). However, the global mortality rate was only 3.8% - this possibly reflecting a less seriously ill population. In contrast, some studies34,35,36 in which higher DD levels were recorded among non-survivors than in survivors, observed no independent association between DD level and mortality after adjusting for confounding factors.
In sum, DD level appears to be associated to the prognosis of patients with COVID-19. However, since most of the studies published to date have been carried out in China and involve very heterogeneous populations in terms of disease severity, with possible selection bias and confounding factors, more scientific evidence is needed in order to confirm this association.
PICO 2: Is DIC in patients with COVID-19 associated to increased mortality?Conclusion 2: Few studies report on the incidence of DIC according to the criteria of the International Society on Thrombosis and Hemostasis (ISTH), and there is little evidence on whether its presence is associated to increased mortality (quality of evidence: low).
Recommendation 2: Daily monitoring of the coagulation parameters and of the development of thrombotic or hemorrhagic manifestations is advised for the early diagnosis of DIC according to the criteria of the ISTH (strength of recommendation: weakly in favor).
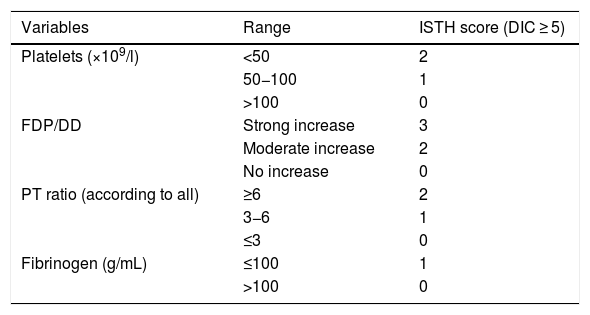
According to the Scientific and Standardization Committee of the ISTH, DIC is defined as an acquired syndrome characterized by intravascular activation of the coagulation systems on a systemic basis, with thrombotic or hemorrhagic phenomena, associated to characteristic laboratory test parameter alterations and accompanied by the development of organ dysfunction as an expression of coagulation activation37 (Table 3). Disseminated intravascular coagulation has been shown to be a predictor of mortality in patients with severe sepsis and septic shock38.
Diagnostic criteria for disseminated intravascular coagulation of the ISHT.
| Variables | Range | ISTH score (DIC ≥ 5) |
|---|---|---|
| Platelets (×109/l) | <50 | 2 |
| 50−100 | 1 | |
| >100 | 0 | |
| FDP/DD | Strong increase | 3 |
| Moderate increase | 2 | |
| No increase | 0 | |
| PT ratio (according to all) | ≥6 | 2 |
| 3−6 | 1 | |
| ≤3 | 0 | |
| Fibrinogen (g/mL) | ≤100 | 1 |
| >100 | 0 |
DIC: disseminated intravascular coagulation; DD: D-dimer; ISTH: International Society on Thrombosis and Hemostasis; FDP: fibrinogen degradation products; PT: prothrombin time.
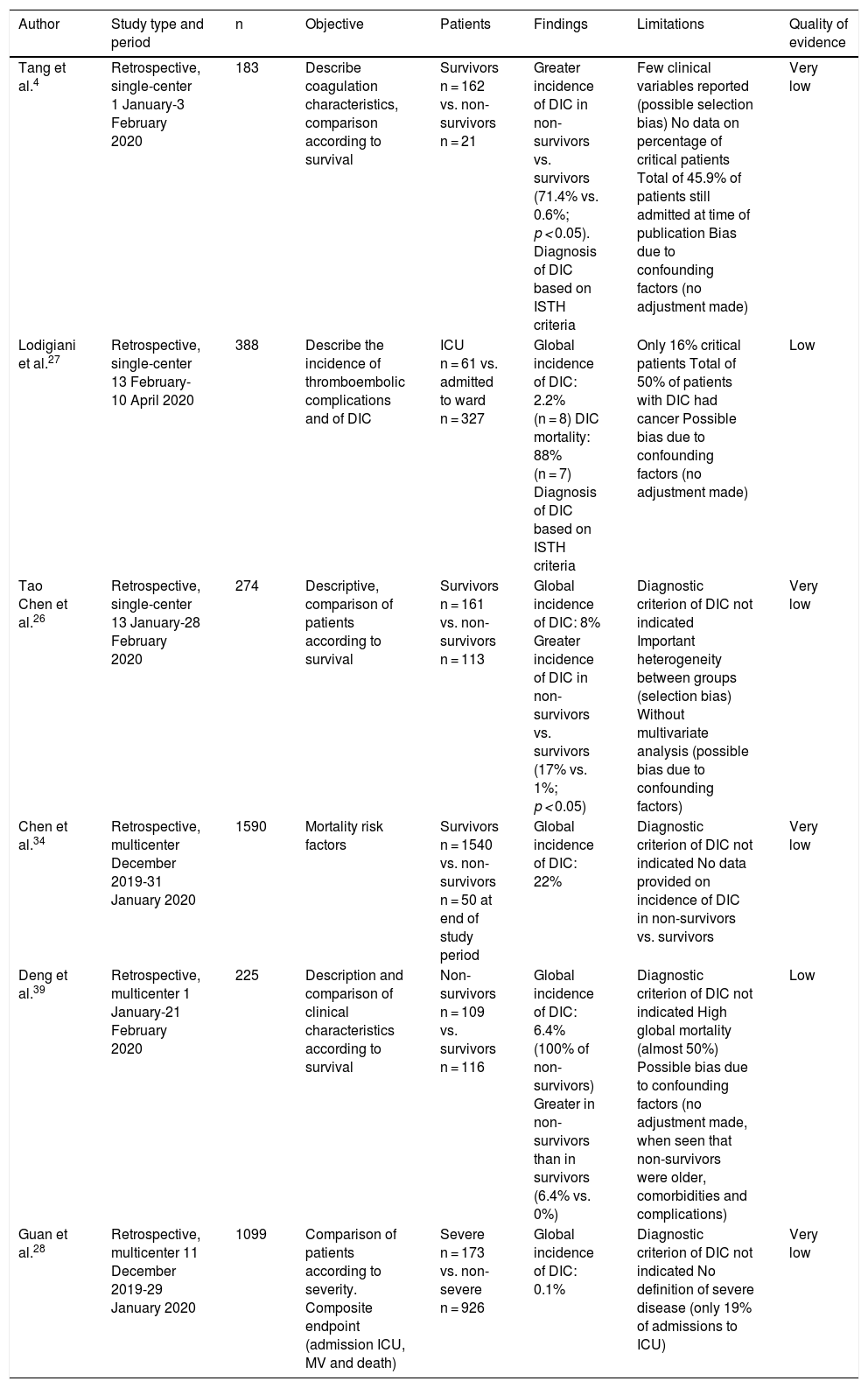
Some studies offer discordant results in relation to the prevalence of DIC and its association to the prognosis of patients with COVID-19 (Table 4). Tang et al.4 found that a large proportion of deceased patients met the international criteria of DIC according to the ISTH (71.4% versus 0.6% of the survivors). This was a low level of evidence study, since the analysis was carried out when many of the patients were still admitted - with no data being reported on the existence of multiorgan failure, sepsis, ARDS or clinical manifestations of DIC (thrombosis or hemorrhage). On the other hand, Lodigiani et al.27 reported a far lower incidence of DIC according to the ISTH criteria (2.2%), with a mortality rate of 88% (n = 7). Other authors26,34,39 have reported incidences of between 6.4–22%, though without specifying the criterion used for establishing the diagnosis. In contrast, Guan et al.28, in their study of 1099 patients, including 173 with severe disease, only reported one case of DIC (incidence 0.1%), likewise without specifying the criterion used for establishing the diagnosis. Some of these studies4,26,39 coincide in reporting a higher incidence of DIC among the patients that died, though without adjusting for confounding factors.
Studies related to the incidence of DIC and its association to the prognosis of COVID-19 disease.
| Author | Study type and period | n | Objective | Patients | Findings | Limitations | Quality of evidence |
|---|---|---|---|---|---|---|---|
| Tang et al.4 | Retrospective, single-center 1 January-3 February 2020 | 183 | Describe coagulation characteristics, comparison according to survival | Survivors n = 162 vs. non-survivors n = 21 | Greater incidence of DIC in non-survivors vs. survivors (71.4% vs. 0.6%; p < 0.05). Diagnosis of DIC based on ISTH criteria | Few clinical variables reported (possible selection bias) No data on percentage of critical patients Total of 45.9% of patients still admitted at time of publication Bias due to confounding factors (no adjustment made) | Very low |
| Lodigiani et al.27 | Retrospective, single-center 13 February-10 April 2020 | 388 | Describe the incidence of thromboembolic complications and of DIC | ICU n = 61 vs. admitted to ward n = 327 | Global incidence of DIC: 2.2% (n = 8) DIC mortality: 88% (n = 7) Diagnosis of DIC based on ISTH criteria | Only 16% critical patients Total of 50% of patients with DIC had cancer Possible bias due to confounding factors (no adjustment made) | Low |
| Tao Chen et al.26 | Retrospective, single-center 13 January-28 February 2020 | 274 | Descriptive, comparison of patients according to survival | Survivors n = 161 vs. non-survivors n = 113 | Global incidence of DIC: 8% Greater incidence of DIC in non-survivors vs. survivors (17% vs. 1%; p < 0.05) | Diagnostic criterion of DIC not indicated Important heterogeneity between groups (selection bias) Without multivariate analysis (possible bias due to confounding factors) | Very low |
| Chen et al.34 | Retrospective, multicenter December 2019-31 January 2020 | 1590 | Mortality risk factors | Survivors n = 1540 vs. non-survivors n = 50 at end of study period | Global incidence of DIC: 22% | Diagnostic criterion of DIC not indicated No data provided on incidence of DIC in non-survivors vs. survivors | Very low |
| Deng et al.39 | Retrospective, multicenter 1 January-21 February 2020 | 225 | Description and comparison of clinical characteristics according to survival | Non-survivors n = 109 vs. survivors n = 116 | Global incidence of DIC: 6.4% (100% of non-survivors) Greater in non-survivors than in survivors (6.4% vs. 0%) | Diagnostic criterion of DIC not indicated High global mortality (almost 50%) Possible bias due to confounding factors (no adjustment made, when seen that non-survivors were older, comorbidities and complications) | Low |
| Guan et al.28 | Retrospective, multicenter 11 December 2019-29 January 2020 | 1099 | Comparison of patients according to severity. Composite endpoint (admission ICU, MV and death) | Severe n = 173 vs. non-severe n = 926 | Global incidence of DIC: 0.1% | Diagnostic criterion of DIC not indicated No definition of severe disease (only 19% of admissions to ICU) | Very low |
DIC: disseminated intravascular coagulation; ISTH: International Society on Thrombosis and Hemostasis; ICU: Intensive Care Unit; MV: mechanical ventilation.
It is therefore possible that DIC may be overdiagnosed, since most of the publications describe the alteration of isolated laboratory parameters as evidence of coagulopathy associated to the disease, without strictly complying with the diagnosis of DIC. Lippi et al.40 published a meta-analysis of 9 studies involving 1779 patients with COVID-19, including 399 with severe disease (22.4%). The analysis showed the platelet count to be significantly lower in the more seriously ill patients, and even lower in those that died. In the four studies (n = 1427) affording data on the incidence of thrombocytopenia, the latter was seen to be associated to a 5-fold higher risk of severe COVID-19 disease (OR 5.1; 95%CI: 1.8–14.6), without referring to other data suggestive of DIC. In relation to the coagulation times, Huang et al.23 recorded longer prothrombin times in critical patients. However, two other studies22,24 reported DD and fibrinogen levels in the more seriously ill patients, though without evidencing alterations in coagulation time. Analyses have also been made of coagulation anomalies based on traditional tests and thromboelastometry profiles in a group of 22 cases admitted to the ICU due to COVID-19 versus healthy controls5. The cases presented significantly higher DD and fibrinogen levels than the controls (p < 0.0001). Furthermore, thromboelastometry profiles evidencing hypercoagulability were recorded, reflected by shorter clot forming times and greater maximum clot firmness values (p < 0.001). It therefore was concluded that patients with COVID-19 presenting hyperfibrinogenemia (resulting in increased fibrin formation and polymerization, which may predispose to thrombosis) present severe hypercoagulability instead of consumption coagulopathy as in the context of DIC.
Likewise, as reported by the American Society of Hematology41, in contrast to the pattern seen in classical DIC secondary to bacterial sepsis or trauma, the coagulopathy observed in patients with COVID-19 is characterized by the elevation of fibrinogen and DD, which is correlated to a parallel increase in inflammatory markers, and prolongation of prothrombin time and activated partial thromboplastin time (aPTT), while thrombocytopenia, if seen, is usually mild to moderate. Furthermore, in both sepsis and ARDS, we observe an increase in procoagulating activity, with pulmonary vascular microthrombosis (immunothrombosis) and a decrease in fibrinolytic activity that contributes to fibrin formation due to endothelial dysfunction following the excessive proinflammatory response to the viral infection. These pulmonary fibrin microthrombi have been found both in the presence and in the absence of DIC15. Therefore, it is possible that the laboratory test findings in patients with COVID-19, such as the increase in degradation products of fibrinogen/DD, should not always be attributed to DIC.
In consequence, coagulopathy associated to COVID-19 disease appears to be associated with a hypercoagulability profile different from that of consumption coagulopathy. Some patients with severe SARS-CoV-2 infection may develop coagulopathy meeting DIC criteria according to the ISTH, with the fulminant activation of coagulation and the consumption of coagulation factors, moderate to severe thrombocytopenia, the prolongation of prothrombin time and activated partial thromboplastin time, marked DD elevation and decreased fibrinogen. However, DIC involves a complex clinical and laboratory test diagnosis that cannot be established only from the isolated laboratory test data42. Consequently, based on the data available at this time, it is not possible to establish its incidence or association to the prognosis of patients with SARS-CoV-2 pneumonia.
PICO 3: Does the administration of empirical anticoagulation in patients with COVID-19 and elevated DD improve the prognosis?Conclusion 3: There is no evidence that empirical anticoagulation at full or intermediate doses results in improved outcomes in patients with COVID-19 and elevated DD (quality of evidence: none).
Recommendation 3: It is not advisable to prescribe empirical anticoagulation in patients with COVID-19 according to the DD levels. Such treatment should only be administered in the context of a controlled clinical trial (strength of recommendation: strongly against).
In coagulopathy, and independently of its cause, treatment of the underlying condition is essential. In the case of COVID-19 infection, given the lack of a specific treatment shown to afford benefit3, supportive care is currently the only option for improving the course of the disease. The recent literature indicates that COVID-19 may predispose to the development of arterial and venous thromboembolic complications as a consequence of the excessive inflammation, hypoxia, patient immobilization and the possible development of DIC. Klok et al.7, in a series of 184 critical patients with SARS-CoV-2 pneumonia, reported a cumulative incidence of such complications of 31%, including venous thromboembolism and arterial thrombosis.
It has been reported that microvascular thrombosis is implicated in hypoxemic respiratory failure in some patients with COVID-19. The necropsy studies to date are limited, but some point to the existence of microvascular thrombosis in the pulmonary circulation43,44. In consequence, it has been postulated that benefit may be obtained from the administration of anticoagulation in the management of critical patients with high DD levels or altered coagulation parameters (coagulopathy or DIC)8,45. A clear example of this is provided by the recommendations of the Spanish Society of Intensive and Critical Care Medicine and Coronary Units (Sociedad Española de Medicina Intensiva, Crítica y Unidades Coronarias [SEMICYUC])46, which include the consideration of anticoagulation in patients with elevated DD (>2000 ng/mL). Similarly, the Cardiovascular Thrombosis working group of the Spanish Society of Cardiology (Sociedad Española de Cardiología)47 has developed a consensus document based on author opinions or small case series, recommending anticoagulation in patients with severe COVID-19 and a high thromboembolic risk – the latter being defined as high DD levels or elevated proinflammatory markers, among other parameters.
However, there is presently no scientific evidence to support such treatment. In fact, our literature search only identified the study published by Tang et al.48, involving 449 patients, which compared patients who received heparin (7 days of low molecular weight heparin or unfractionated heparin) versus those who did not. The mortality rate was 29.8%, with no differences being observed after 28 days between the heparin and non-heparin groups (30.2% versus 29.7%). The authors used the sepsis-induced coagulopathy score (SIC)49 instead of the DIC score of the ISTH37. With this classification, 21.6% of the patients met criteria for an SIC score ≥4, and in these subjects the administration of heparin was associated to lesser mortality (40.0% versus 64.2%; p = 0.03), though not so in the patients with an SIC score <4. Similarly, in the patients with DD >3 μg/mL (6 times the upper limit of normal), the administration of heparin was associated to a 20% decrease in mortality rate. However, this study had important limitations, since no analysis was made of the use of anticoagulation therapy; instead, it compared the use of heparin as prophylaxis (use recommended in hospitalized patients, and regarded as good clinical practice) versus no such use (malpractice). Likewise, this was a retrospective study, with possible selection bias, that did not report the characteristics of the compared groups regarding patient severity, and no multivariate analysis was made to assess SIC and mortality. In view of the above, these results must be viewed with great caution.
According to most of the studies published to date50, there is no indication for full dose empirical anticoagulation in patients with COVID-19 disease, unless clinical thrombosis or thromboembolism has been documented51 or there is some other classical indication for its use (mechanical valves, atrial fibrillation, etc.). In fact, to date there is no published evidence justifying an increase in heparin dose in patients with severe COVID-19 disease; such practice therefore should only be applied in the context of controlled clinical trials. Doing otherwise would be regarded as clinical malpractice. In effect, new treatments must be evaluated in randomized controlled trials to truly understand both their benefits and their associated risks52. Many of the failed hypotheses in clinical research over the last 30 years have reemerged with the hope of affording new therapies for COVID-19. Maintaining the principles of evidence based medicine in critical patient care, as has been demonstrated in randomized multicenter trials, will improve the outcomes of patients with severe COVID-19 disease.
Similarly, many institutional protocols, including those of the Spanish Society of Hematology (Sociedad Española de Hematología)53, have advocated intermediate intensity thromboprophylaxis (i.e., the standard daily prophylactic dose of low molecular weight heparin twice a day), and have proposed its use in the case of patients with a high risk of thrombosis7, even in the absence of supporting scientific evidence. In fact, both the World Health Organization (WHO)54 and different societies50 continue to recommend standard pharmacological thromboprophylaxis doses.
It is clear that thromboprophylaxis should be administered to all patients admitted to hospital, in accordance with the current clinical practice guides51,55. In the case of patients hospitalized with COVID-19, with an increased risk of thrombosis because of their condition, and due to the procoagulant state associated to the disease, the current recommendation to use heparin at standard prophylactic doses (daily low molecular weight heparin corrected for body weight and renal clearance or fondaparinux, proposed in preference of unfractionated heparin in order to reduce contact) should also be maintained in order to prevent thrombotic events46,56. A panel of medical experts from China and Europe have developed a consensus document based on the evidence regarding the prevention and management of thromboembolic disease associated to COVID-19 that confirms this57. Thromboprophylaxis should be maintained, despite anomalous coagulation test results, in the absence of active bleeding, and it should only be suspended if the platelet count drops to below 25–30 × 109/l. Mechanical thromboprophylaxis is to be used when pharmacological thromboprophylaxis is contraindicated41,46.
PICO 4: Should we provide anticoagulation in patients with COVID-19 and associated DIC?Conclusion 4: There is no evidence to justify the use of anticoagulation in DIC associated to COVID-19 (quality of evidence: none).
Recommendation: It is not possible to recommend the administration of anticoagulation therapy in patients with DIC associated to COVID-19, except in cases with confirmed thrombotic phenomena (strength of recommendation: weakly against).
In the case of confirmed DIC associated to SARS-CoV2 disease, and in the same way as with DIC of any other cause, the effectiveness of anticoagulation is subject to controversy, despite the conduction of multiple randomized controlled trials49. Resolution of the triggering event or underlying disease is crucial for management of the syndrome, without anticoagulation therapy having been shown to improve the prognosis58. Consequently, and in the absence of any other indication for anticoagulation (documented ischemic phenomena or purpura fulminans), these patients should only receive thromboprophylaxis50.
ConclusionsFollowing the systematic review of the literature published to date, it can be concluded that DD level may be a predictor of severity, and even of mortality. However, the evidence supporting this association is of low quality, and further studies adjusting for confounding factors are needed to confirm it. In turn, COVID-19 disease may be associated to DIC, though its incidence in the current studies is variable; if DIC occurs, it could have an impact upon the patient prognosis. It therefore would be advisable to monitor hemostasis in order to allow the early identification of DIC in accordance with the international diagnostic criteria. In relation to empirical or intermediate-dose anticoagulation, there is no evidence to support its use in our patients, even in the presence of DD elevation, for in the same way as in sepsis or respiratory distress, this parameter may be altered without the existence of DIC. Thromboprophylaxis is indicated in all patients admitted to hospital due to COVID-19 disease (except where contraindicated), and full-dose anticoagulation should only be administered in the case of classical indications, documented arterial or venous thromboembolic disease and, in the case of DIC, provided it is associated to ischemic phenomena or purpura fulminans.
Authorship/collaborationAR, MB, RC and GM contributed to study conception and design, data acquisition, and data analysis and interpretation.
RC and GM participated in drafting of the article and in the critical review of its intellectual content.
AR and MB contributed to final approval of the submitted version of the manuscript.
Financial supportNone.
Conflicts of interestNone.
Thanks are due to all the healthcare professionals that continue the struggle against this pandemic each day.
Please cite this article as: Moreno G, Carbonell R, Rodríguez M, Rodríguez A. Revisión sistemática sobre la utilidad pronóstica del dímero-D, coagulación intravascular diseminada y tratamiento anticoagulante en pacientes graves con COVID-19. Med Intensiva. 2021;45:42–55.