Sickle cell disease (SCD) is an inherited pathology based on structural alteration of hemoglobin and characterized by vaso-occlusive crises1 with a prevalence of one million worldwide. Acute chest syndrome (ACS) is the main life-threatening complication. Until now ACS diagnostic criteria was based on clinical evaluation and chest X-ray (CXR). These patients often require non-invasive ventilation (NIV) to lung recruitment and oxygenation improvement. NIV parameters are indicated according to clinical variables, oxygen requirements and sequentially CXR data. Although lung ultrasound (LUS) could provide information for both diagnosis and evolution, it has never been tested in pediatrics population.
We present two cases of LUS of SCD admitted to pediatric intensive care unit (PICU) due to ACS with respiratory failure.
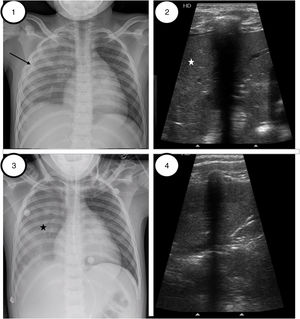
Case 1A 7-year-old girl with SCD underwent a minor elective surgery (inguinal hernia). At 24h, presented a subsequent clinical deterioration determined by persistent cough, progressive tachypnea, inspiratory crackles, with right basal hypoventilation and fever despite preventing measures according to SCD local protocol. CXR showed bilateral and right superior lobe infiltrates with minor pleural effusion. Serial LUS were performed during PICU admission. First ultrasound showed a global consolidation of the entire right lung with an associated pleural effusion of 0.5cm. Fig. 1 shows both images. NIV was initiated with maxim parameters of IPAP 18 and EPAP 8 cmH2O, and a FiO2 up to 60%, requiring red cell transfusion. During the successive ultrasound controls, progressive lung improvement was observed with an increase in lung aeration to almost complete resolution. Serial CXR changes were less evident. Both images can be compared in Fig. 1. NIV support was gradually decreased being suspended after 7 days.
Patient 1. (1) Initial chest radiography preceding alveolar recruitment, right coalescent opacities with right pleural effusion (black narrow). (2) Lung ultrasound image preceding lung recruitment, exhibiting diffuse abnormal pulmonary aeration with condensation pattern (white star) on the right anterior, lateral and posterior regions. (3) Chest-X radiography after VNI recruitment. Similar coalescent opacities in right lung (black star) without pleural effusion. (4) Lung ultrasound reveals the presence of sonographic air bronchograms representing air-filled bronchi with linear, hyperechoic branching echoes toward pleural lung. The consolidated lung parenchyma has improved with respect to Image 2.
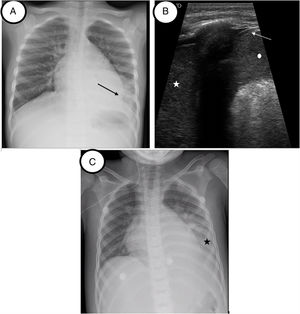
An 8-year-old girl with SCD was admitted to the emergency department due respiratory failure and fever with a pathologic CXR in which left lower lobe infiltration was observed. Due to ACS suspicion, patient was transferred to the PICU for early respiratory support. On admission, LUS showed left lung global collapse with 0.3cm pleural effusion. Both images can be compared in Fig. 2. NIV was initiated to optimize respiratory support, requiring CPAP up to 8cmH2O and FiO2 up to 50%. Clinical and ultrasonographic evolution was favorable, allowing the suspension of support in the following 48h. CXR image after VNI lung recruitment can be compared in Fig. 2.
Patient 2. (A) Initial chest radiography before lung recruitment. Coalescent opacities in left lower lobe (black arrow). No pleural effusion observed. (B) Lung ultrasound image before VNI lung recruitment. Tissular pattern suggesting consolidation (white star). No air bronchogram is observed. The lung partner is similar to the spleen pattern (white point). White arrow shows the doble-rail image of the diaphragm. (C) Chest-X radiography after recruitment. Area of hazy with increased lung opacity (black star).
Thanks to lung ultrasound bedside controls, PEEP parameters were increased until an improvement in lung aeration in both patients. Moreover, while the echographic and clinical improvement was simultaneous, CXR did not reveal major changes.
ACS is the main cause of PICU admission in patients with SCD.2 Its diagnosis is based on the confluence of fever and/or respiratory symptoms accompanied by new infiltrate on the CXR. It is essential to have a high index of suspicion for early diagnosis and treatment support.3 Respiratory process ethiology can be several: infectious (bacterial, viral), fatty embolism in the context of a vasoocclusive crisis, or by pulmonary vascular micro infarcts due to hypoventilation secondary to atelectasis. Clinical presentation can be wide from a moderate respiratory process to an acute respiratory distress syndrome (ARDS) and even leading to a multiorgan failure. There is already experience in the support of ACS in adults on extracorporeal oxygenation membrane (ECMO).4
Some of the radiological signs which can be found are both lobular and multilobar presence consolidations, or pulmonary collapse, in some cases associated with pleural effusion. However, CXR may be normal in early stages in up to 66% of the cases,5 so that the suspicion of ACS was because of the hypoxemia.3
LUS use has increased in recent years in the diagnosis of multiple lung processes. However, it has not been previously described the LUS use for diagnosis and follow-up of ACS in pediatrics. An adults study published in 20156 compared the usefulness of CXR versus LUS in the diagnosis of ACS, using CT-based tomography as the reference standard. In this study, they described three patterns in the lung parenchyma: normal, ground-glass opacity and consolidation. In accordance with these patterns, LUS was evaluated in 12 lung regions (6 in each hemithorax), associating a score of 0–2, respectively. Normal pattern was defined by the presence of non-coalescing B lines or by lung sliding with A lines, ground-glass opacities were defined by the presence of coalescing B lines and consolidation by punctate hyperechogenic images. According to this score, they evaluated the lung aeration of each patient and saw that a higher score assessed by LUS was associated with worse posterior evolution. They also proved that LUS was superior to radiography for diagnosis consolidations and pleural effusion in these patients, presenting a greater sensitivity (72% versus 44%), especially in the assessment of the post-basal regions.
In our patients, ultrasound shows a higher degree of lesion than that assessed by radiography, as in the study by Rahmouni et al.6 of 2016.
There is currently no experience in pediatrics for diagnosis ACS using LUS. Moreover there are no studies about lung recruitment maneuvers guided by ultrasound in children. Ultrasound-controlled lung recruitment is well-defined in adults with several articles emphasizing its usefulness as a bedside tool7–10 in ARDS, even though in the evaluation of aeration of ventilated patients.
In our patients, LUS allowed to confirm the ACS and lung collapse diagnosis. It was also useful to follow-up the lung recruitment improvement, helping to assess the NIV parameters. However, more studies are needed in pediatric population to confirm our experience.