Edited by: Rosario Amaya Villar - Unidad de Cuidados Intensivos, Hospital Universitario Virgen del Rocio, Sevilla, España
Last update: December 2023
More infoTo describe the characteristics of patients with acute respiratory distress syndrome (ARDS) due to bilateral COVID-19 pneumonia on invasive mechanical ventilation (IMV), and to analyze the effect of prone position >24 h (prolonged) (PPP) compared to prone decubitus <24 h (PP).
DesignA retrospective observational descriptive study was carried out, with uni- and bivariate analyses.
SettingDepartment of Intensive Care Medicine. Hospital General Universitario de Elche (Elche, Alicante, Spain).
ParticipantsPatients with SARS-CoV-2 pneumonia (2020−2021) on IMV due to moderate-severe ARDS, ventilated in prone position (PP).
InterventionsIMV. PP maneuvers.
Main variables of interestSociodemographic characteristics, analgo-sedation, neuromuscular blockade (NMB), PD duration, ICU stay and mortality, days of IMV, non-infectious complications, healthcare associated infections.
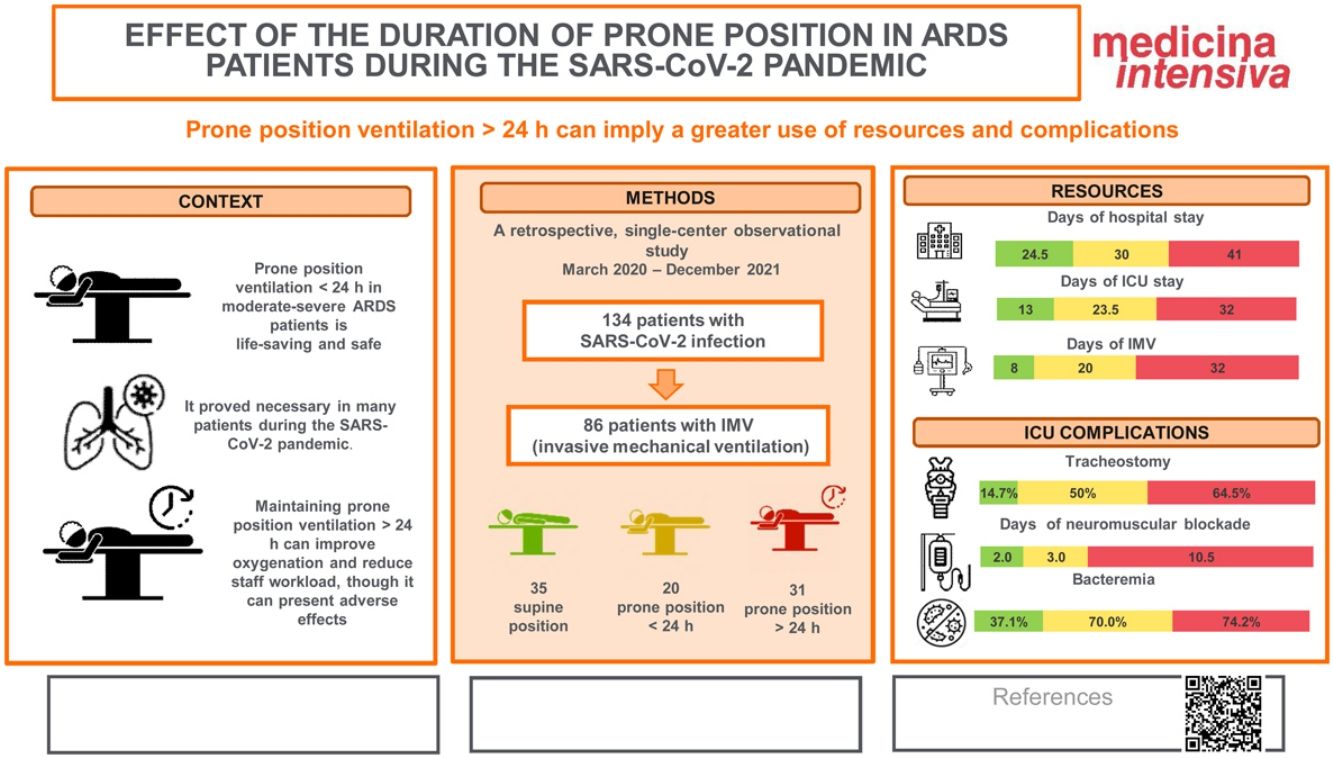
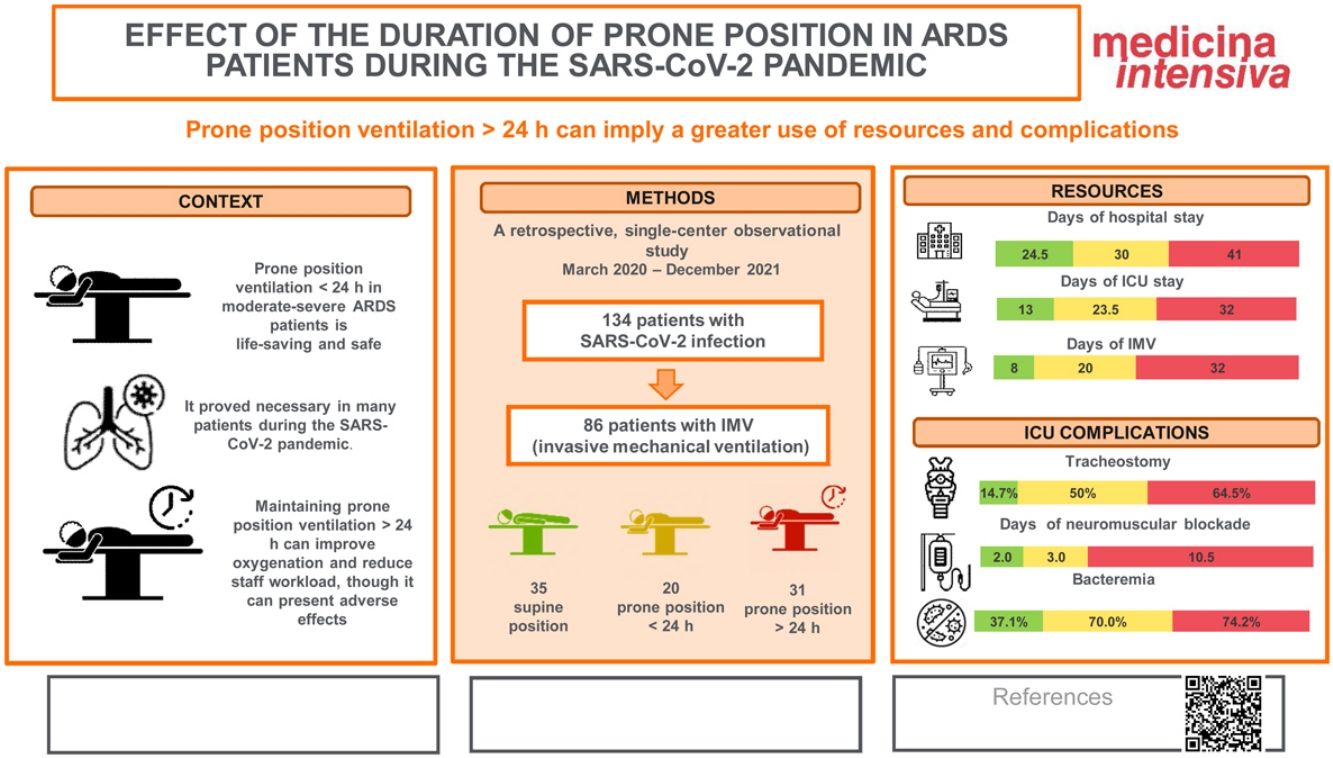
ResultsFifty-one patients required PP, and of these, 31 (69.78%) required PPP. No differences were observed in terms of patient characteristics (gender, age, comorbidities, initial severity, antiviral and antiinflammatory treatment received). Patients on PPP had poorer tolerance to supine ventilation (61.29% vs 89.47%, p = 0.031), longer hospital stay (41 vs 30 days, p = 0.023), more days of IMV (32 vs 20 days, p = 0.032), longer duration of NMB (10.5 vs 3 days, p = 0.0002), as well as a higher percentage of episodes of orotracheal tube obstruction (48.39% vs 15%, p = 0.014).
ConclusionsPPP was associated with greater resource use and complications in patients with moderate-severe ARDS due to COVID-19.
Describir las características de los pacientes con síndrome de distrés respiratorio agudo (SDRA) por neumonía bilateral por COVID-19 en ventilación mecánica invasiva (VMI) y analizar el efecto del decúbito prono prolongado >24 h (DPP) respecto al decúbito prono <24 h (DP).
DiseñoEstudio observacional retrospectivo descriptivo. Análisis uni y bivariante.
ÁmbitoServicio de Medicina Intensiva. Hospital General Universitario de Elche.
ParticipantesPacientes con neumonía por SARS-CoV-2 (2020−21) en VMI por SDRA moderado-severo.
IntervencionesVMI. Maniobras de DP.
Variables de interés principalessociodemográficas; analgosedación; bloqueo neuromuscular (BNM); DP (duración), estancia y mortalidad en UCI, días de VMI; complicaciones no infecciosas; infecciones asociadas a la asistencia sanitaria (IAAS).
Resultados51 pacientes precisaron DP y de ellos 31 (69,78%) requirieron DPP. No se encontraron diferencias en las características iniciales de los pacientes, (sexo, edad, comorbilidades, gravedad inicial, o en el tratamiento antiviral y antiinflamatorio recibido). Los pacientes con DPP presentaron menor tolerancia a la ventilación en decúbito supino (61,29% vs 89,47%, p = 0,031), mayor estancia hospitalaria (30 vs 41 días, p = 0,023), más días de VMI (32 vs 20 días, p = 0,032), mayor duración del tratamiento con BNM (3 vs 10,5 días, p = 0,0002), así como un mayor porcentaje de episodios de obstrucción del tubo orotraqueal (15% vs 48,39%, p = 0,014).
ConclusionesEl DPP se asoció con mayor uso de recursos y complicaciones en pacientes con SDRA moderado-severo por COVID-19.
A previously unknown disease (COVID-19) caused by a new virus of the Coronaviridae family (SARS-CoV-2) first appeared in China in December 2019. In March 2020, the World Health Organization (WHO) declared a worldwide pandemic due to the rapid spread of this virus.1 In Spain, a total of 13,614,807 confirmed cases of COVID-19 and 116,108 deaths have been confirmed to date.2 An important number of affected patients develop acute respiratory distress syndrome (ARDS), requiring invasive mechanical ventilation (IMV) in the Intensive Care Unit (ICU).3
Acute respiratory distress syndrome is characterized by acute and diffuse inflammatory lung damage, resulting in increased pulmonary vascular permeability and lung weight, with a decrease in compliance.4 In patients with severe ARDS, the early use of the prone position (PP) for at least 16 consecutive hours associated to protective ventilation has been shown to reduce early mortality,5 due to improvement of the difference in ventral-dorsal transpulmonary pressure and of the ventilation/perfusion ratio, among other mechanisms.6 This maneuver requires the intervention of experienced professionals and poses risks such as the displacement of invasive devices (orotracheal tube [OTT], vascular catheters, drains, etc.) or hemodynamic instability.5–7 At present, PP for at least 16 h is advised in patients with severe ARDS.8,9 During the pandemic, the number of patients requiring IMV and PP increased,10 and as a consequence of either an excessive care burden in the ICU or increased severity of the clinical condition, some patients remained in PP for longer periods of time.
In contrast to PP with a duration of up to 24 h, the data on the use of prolonged PP (over 24 h: PPP) are limited. While there is evidence that PP for less than 24 h is effective and safe,5 the data on the efficacy and safety of the maneuver for 36−48 h are based on descriptive series.10–16 The present study describes the characteristics of intubated patients with bilateral pneumonia secondary to COVID-19 and subjected to PPP compared with patients in which PP was maintained for less than 24 h.
Patients and methodsA retrospective, descriptive observational study was made of consecutive patients admitted between 15/03/2020 to 31/12/2021 to the Department of Intensive Care Medicine (DICM) of Hospital General Universitario de Elche (HGUE) (Elche, Alicante, Spain) — a hospital with 548 beds (data from 2021). The DICM experienced an increase from 12 simultaneously managed beds to 25 patients at the peak of care during the study period. Not all the patients subjected to IMV due to SARS-CoV-2 pneumonia were attended by the DICM; some patients were admitted to the Department of Anesthesia, conditioned to the availability of space.
The study retrospectively included patients over 18 years of age with SARS-CoV-2 infection confirmed by polymerase chain reaction (PCR) testing, admitted to the DICM and subjected to IMV due to ARDS secondary to bilateral pneumonia. Patients with SARS-CoV-2 infection who were intubated for other reasons, or with contraindications to PP were excluded from the study.
The DICM has no electronic information registry system; the data were therefore compiled from the clinical registries. We collected sociodemographic data (age, gender) and information on treatment for COVID-19 (immunosuppressors, antiretroviral drugs), comorbidities (diabetes mellitus [DM], arterial hypertension [AHT], dyslipidemia [DLP], cardiovascular disease, etc.), the SAPS 3 (Simplified Acute Physiology Score) upon admission,17 the use of IMV and the application of PP maneuvers — with the consideration of PPP, in addition to the total number of cycles required. We also recorded the interval between intubation and the first PP cycle, the PaO2/FiO2 ratio at the start of the first cycle as a severity indicator, the same ratio after the first maneuver, the required maximum positive end-expiratory pressure (PEEP), and posterior tolerance of ventilation in the supine position (SP) — defined as the capacity to maintain PaO2/FiO2 in SP > 150 in the following 24 h. The decision to maintain PP was not protocolized but depended on the situation of the patient and the possibilities of the team to perform the maneuver in a timely and safe manner. The rest of management, both medical and referred to the prevention of nosocomial infection and the protective ventilation parameters, was based on the standard protocols (tidal volume [VT] 6−8 ml/kg ideal body weight, plateau P ≤ 30 cmH2O), which included PP maneuvering in patients with PaO2/FiO2 < 150 with FiO2 ≥ 60% after optimizing the PEEP levels.9 All the patients were ventilated with closed aspiration and passive humidifying systems.
The study outcomes were hospital and ICU stay, the number of days of IMV, mortality in the ICU, the use of sedatives, analgesics and neuromuscular blockers (NMBs), the need for vasoactive drugs, tracheotomy, loss of devices (catheters, tubes, etc.), orotracheal tube (OTT) obstruction, cardiac arrest (CA) during PP, intolerance to enteral nutrition (EN) – defined by a gastric residual volume > 200 ml – and the development of healthcare-associated infections (HAIs).18 The different periods of the pandemic were also taken into consideration.19
The study was approved by the Medicinal Products Research Ethics Committee (mpREC) of Hospital General Universitario of Elche, with registry code PI 103/2021.
A descriptive statistical analysis was made, with the calculation of proportions for categorical variables, and of the median (with percentiles 25 and 75 [p25–p75]) for continuous variables. Comparisons were made of the outcomes of the patients with PP versus those subjected to PPP. The contrasting of hypotheses on proportions was made using the chi-square test. Quantitative variables were contrasted using two-sided nonparametric tests (Mann–Whitney U test for two variables, and the Kruskal–Wallis test in the case of three variables), with an alpha significance level of 5%. The Stats direct v 3.3.5 package (Stats Direct Ltd., Wirral, UK, https://www.statsdirect.com/) was used throughout.
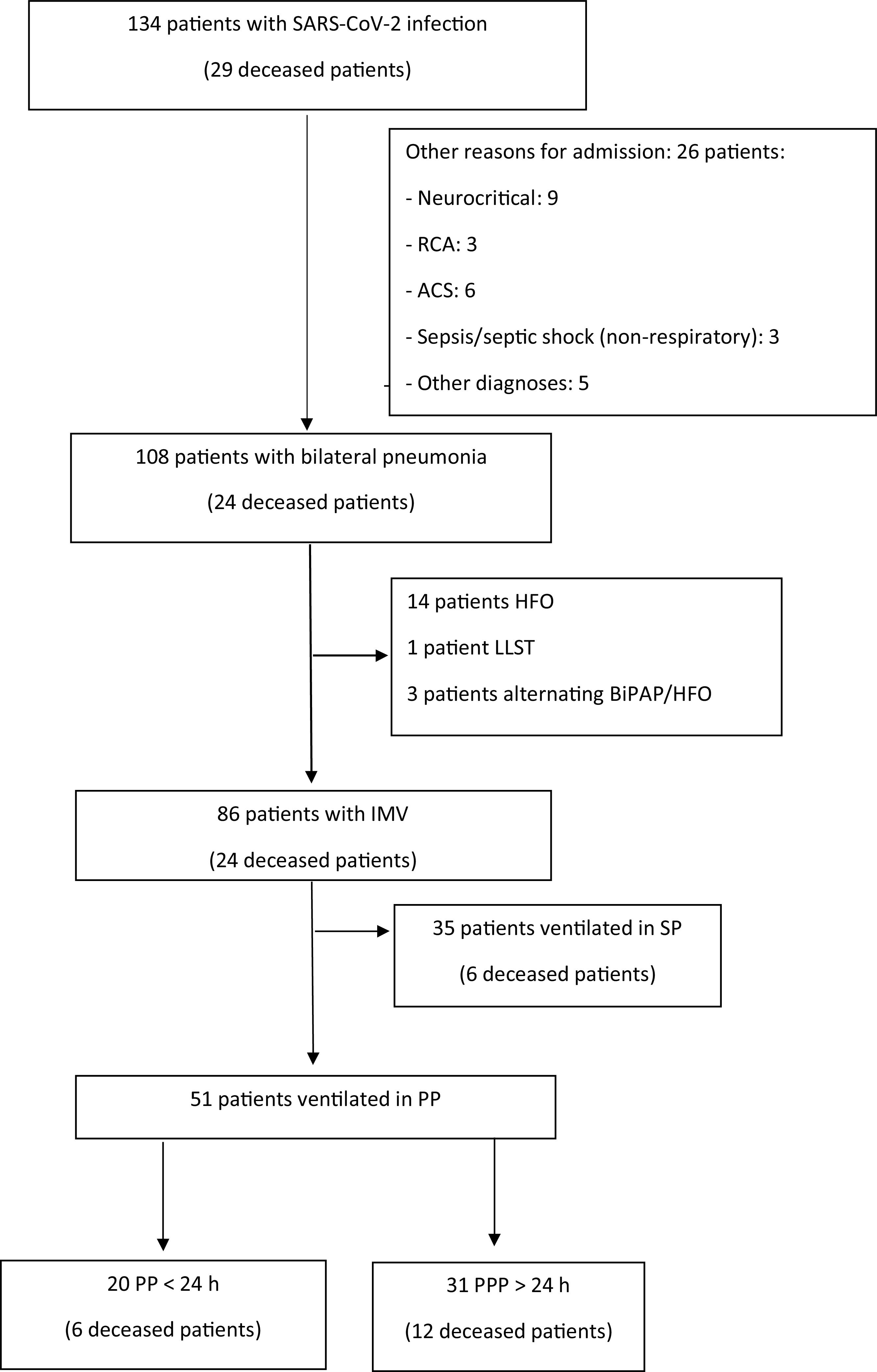
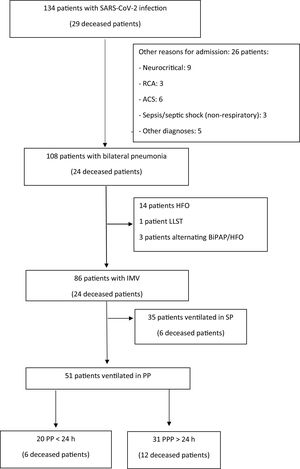
ResultsA total of 134 consecutive patients admitted to the DICM during 2020 and 2021 were analyzed. The flow chart of the patients is summarized in Fig. 1. Of these patients, 86 required IMV. The patients were mostly males (73.26%, 63 patients), with a median age of 65 years (p25–p75: 57–71 years). In total, 51 patients (60%) required PP, all with severe ARDS, and PaO2/FiO2 < 150 mmHg. Their characteristics, according to the duration of PP, are summarized in Table 1. The initial respiratory situation was similar, with parameters indicative of severe ARDS (initial PaO2/FiO2 103 in the patients with PP versus 96 in the patients with PPP; p = 0.885). The patients with PPP received more cycles (up to three cycles, Table 1), with cycling on an early basis in both groups. Most of these maneuvers were carried out during the third wave of the pandemic (Tables 1 and 2).
Flow diagram of the patients included in the analysis.
ACS: acute coronary syndrome; RCA: reanimated cardiac arrest; HFO, high-flow oxygen therapy; LLST: Limitation of life-sustaining treatment; BIPAP: bilevel positive airway pressure ventilation; IMV: invasive mechanical ventilation; SP: supine position; PP: prone position; PPP: prolonged prone position.
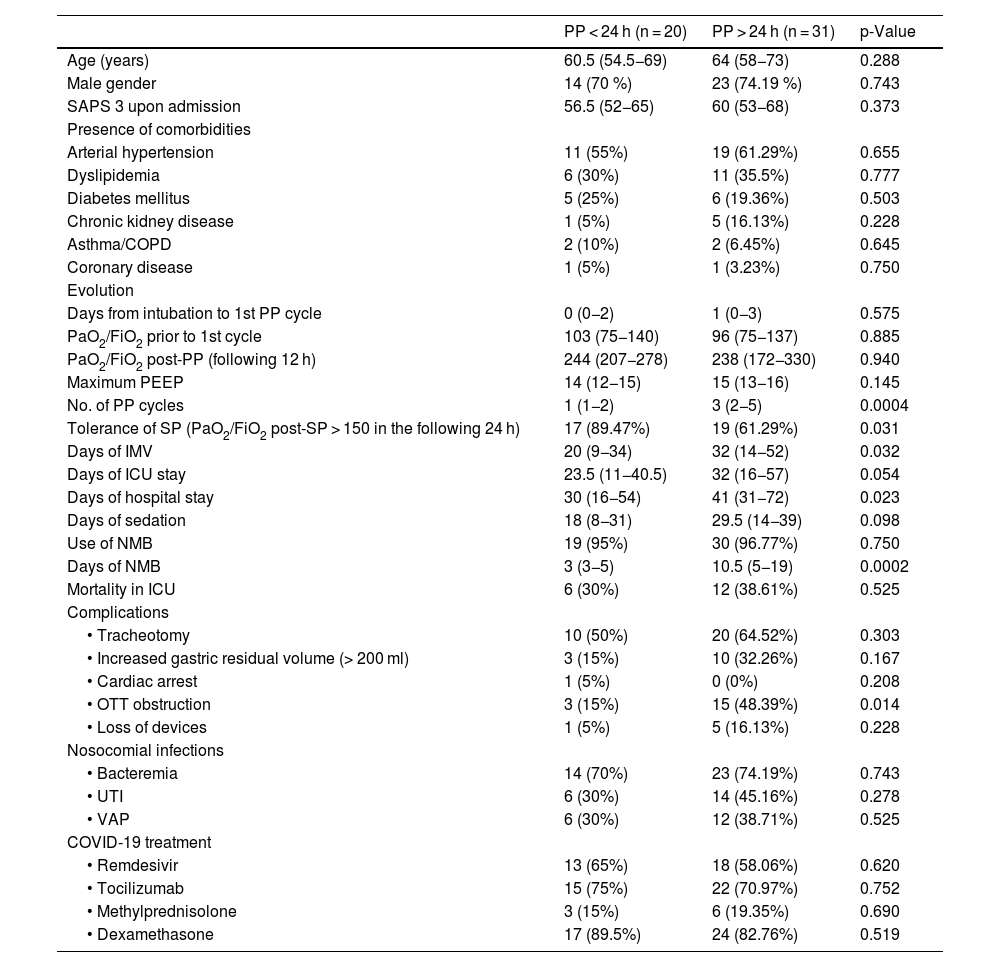
Characteristics and outcomes in patients subjected to IMV, according to the duration of PP.
| PP < 24 h (n = 20) | PP > 24 h (n = 31) | p-Value | |
|---|---|---|---|
| Age (years) | 60.5 (54.5−69) | 64 (58−73) | 0.288 |
| Male gender | 14 (70 %) | 23 (74.19 %) | 0.743 |
| SAPS 3 upon admission | 56.5 (52−65) | 60 (53−68) | 0.373 |
| Presence of comorbidities | |||
| Arterial hypertension | 11 (55%) | 19 (61.29%) | 0.655 |
| Dyslipidemia | 6 (30%) | 11 (35.5%) | 0.777 |
| Diabetes mellitus | 5 (25%) | 6 (19.36%) | 0.503 |
| Chronic kidney disease | 1 (5%) | 5 (16.13%) | 0.228 |
| Asthma/COPD | 2 (10%) | 2 (6.45%) | 0.645 |
| Coronary disease | 1 (5%) | 1 (3.23%) | 0.750 |
| Evolution | |||
| Days from intubation to 1st PP cycle | 0 (0−2) | 1 (0−3) | 0.575 |
| PaO2/FiO2 prior to 1st cycle | 103 (75−140) | 96 (75−137) | 0.885 |
| PaO2/FiO2 post-PP (following 12 h) | 244 (207−278) | 238 (172−330) | 0.940 |
| Maximum PEEP | 14 (12−15) | 15 (13−16) | 0.145 |
| No. of PP cycles | 1 (1−2) | 3 (2−5) | 0.0004 |
| Tolerance of SP (PaO2/FiO2 post-SP > 150 in the following 24 h) | 17 (89.47%) | 19 (61.29%) | 0.031 |
| Days of IMV | 20 (9−34) | 32 (14−52) | 0.032 |
| Days of ICU stay | 23.5 (11−40.5) | 32 (16−57) | 0.054 |
| Days of hospital stay | 30 (16−54) | 41 (31−72) | 0.023 |
| Days of sedation | 18 (8−31) | 29.5 (14−39) | 0.098 |
| Use of NMB | 19 (95%) | 30 (96.77%) | 0.750 |
| Days of NMB | 3 (3−5) | 10.5 (5−19) | 0.0002 |
| Mortality in ICU | 6 (30%) | 12 (38.61%) | 0.525 |
| Complications | |||
| • Tracheotomy | 10 (50%) | 20 (64.52%) | 0.303 |
| • Increased gastric residual volume (> 200 ml) | 3 (15%) | 10 (32.26%) | 0.167 |
| • Cardiac arrest | 1 (5%) | 0 (0%) | 0.208 |
| • OTT obstruction | 3 (15%) | 15 (48.39%) | 0.014 |
| • Loss of devices | 1 (5%) | 5 (16.13%) | 0.228 |
| Nosocomial infections | |||
| • Bacteremia | 14 (70%) | 23 (74.19%) | 0.743 |
| • UTI | 6 (30%) | 14 (45.16%) | 0.278 |
| • VAP | 6 (30%) | 12 (38.71%) | 0.525 |
| COVID-19 treatment | |||
| • Remdesivir | 13 (65%) | 18 (58.06%) | 0.620 |
| • Tocilizumab | 15 (75%) | 22 (70.97%) | 0.752 |
| • Methylprednisolone | 3 (15%) | 6 (19.35%) | 0.690 |
| • Dexamethasone | 17 (89.5%) | 24 (82.76%) | 0.519 |
Data are expressed as median (p25-p75) or n (%). SAPS 3: Simplified Acute Physiology Score; COPD: chronic obstructive pulmonary disease; PaO2: arterial oxygen partial pressure; FiO2: inspired fraction of oxygen; PEEP: positive end-expiratory pressure; PP: prone position; SP; supine position; IMV: invasive mechanical ventilation; ICU: Intensive Care Unit; NMB: neuromuscular blocker; OTT: orotracheal tube; UTI: urinary tract infection; VAP: ventilator associated pneumonia.
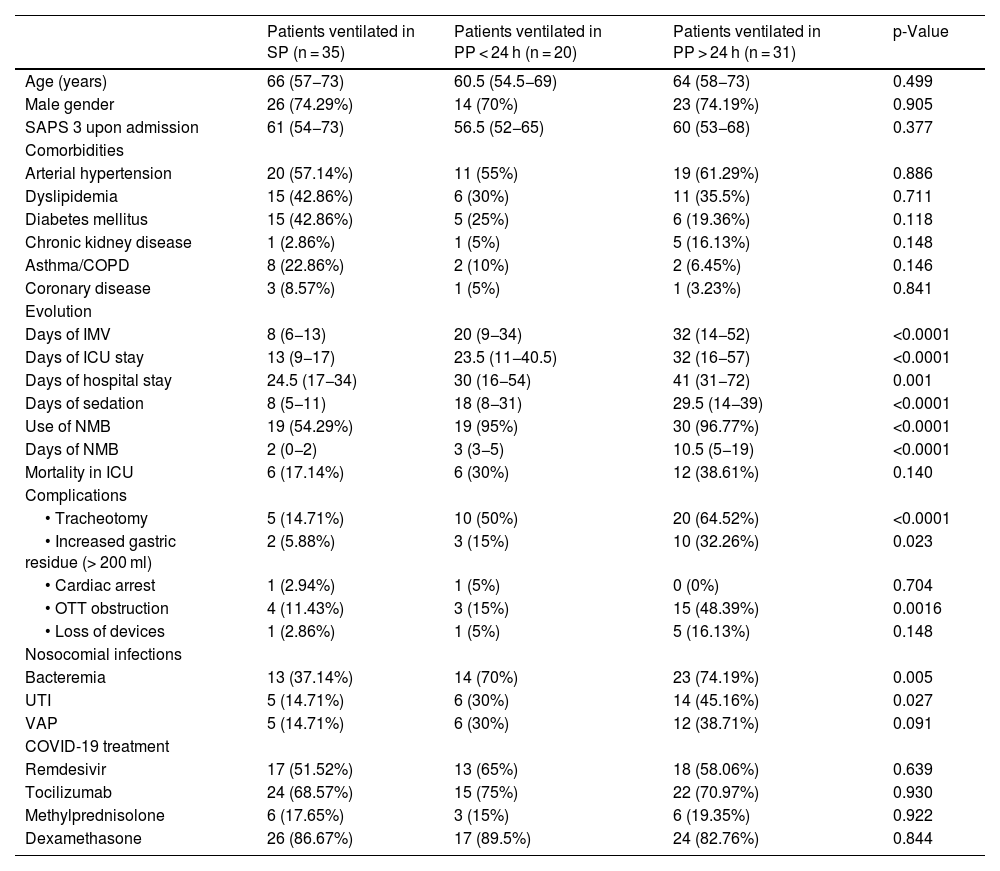
Characteristics and evolution in patients with SARS-CoV-2 pneumonia subjected to IMV.
| Patients ventilated in SP (n = 35) | Patients ventilated in PP < 24 h (n = 20) | Patients ventilated in PP > 24 h (n = 31) | p-Value | |
|---|---|---|---|---|
| Age (years) | 66 (57−73) | 60.5 (54.5−69) | 64 (58−73) | 0.499 |
| Male gender | 26 (74.29%) | 14 (70%) | 23 (74.19%) | 0.905 |
| SAPS 3 upon admission | 61 (54−73) | 56.5 (52−65) | 60 (53−68) | 0.377 |
| Comorbidities | ||||
| Arterial hypertension | 20 (57.14%) | 11 (55%) | 19 (61.29%) | 0.886 |
| Dyslipidemia | 15 (42.86%) | 6 (30%) | 11 (35.5%) | 0.711 |
| Diabetes mellitus | 15 (42.86%) | 5 (25%) | 6 (19.36%) | 0.118 |
| Chronic kidney disease | 1 (2.86%) | 1 (5%) | 5 (16.13%) | 0.148 |
| Asthma/COPD | 8 (22.86%) | 2 (10%) | 2 (6.45%) | 0.146 |
| Coronary disease | 3 (8.57%) | 1 (5%) | 1 (3.23%) | 0.841 |
| Evolution | ||||
| Days of IMV | 8 (6−13) | 20 (9−34) | 32 (14−52) | <0.0001 |
| Days of ICU stay | 13 (9−17) | 23.5 (11−40.5) | 32 (16−57) | <0.0001 |
| Days of hospital stay | 24.5 (17−34) | 30 (16−54) | 41 (31−72) | 0.001 |
| Days of sedation | 8 (5−11) | 18 (8−31) | 29.5 (14−39) | <0.0001 |
| Use of NMB | 19 (54.29%) | 19 (95%) | 30 (96.77%) | <0.0001 |
| Days of NMB | 2 (0−2) | 3 (3−5) | 10.5 (5−19) | <0.0001 |
| Mortality in ICU | 6 (17.14%) | 6 (30%) | 12 (38.61%) | 0.140 |
| Complications | ||||
| • Tracheotomy | 5 (14.71%) | 10 (50%) | 20 (64.52%) | <0.0001 |
| • Increased gastric residue (> 200 ml) | 2 (5.88%) | 3 (15%) | 10 (32.26%) | 0.023 |
| • Cardiac arrest | 1 (2.94%) | 1 (5%) | 0 (0%) | 0.704 |
| • OTT obstruction | 4 (11.43%) | 3 (15%) | 15 (48.39%) | 0.0016 |
| • Loss of devices | 1 (2.86%) | 1 (5%) | 5 (16.13%) | 0.148 |
| Nosocomial infections | ||||
| Bacteremia | 13 (37.14%) | 14 (70%) | 23 (74.19%) | 0.005 |
| UTI | 5 (14.71%) | 6 (30%) | 14 (45.16%) | 0.027 |
| VAP | 5 (14.71%) | 6 (30%) | 12 (38.71%) | 0.091 |
| COVID-19 treatment | ||||
| Remdesivir | 17 (51.52%) | 13 (65%) | 18 (58.06%) | 0.639 |
| Tocilizumab | 24 (68.57%) | 15 (75%) | 22 (70.97%) | 0.930 |
| Methylprednisolone | 6 (17.65%) | 3 (15%) | 6 (19.35%) | 0.922 |
| Dexamethasone | 26 (86.67%) | 17 (89.5%) | 24 (82.76%) | 0.844 |
Data are expressed as median (p25-p75) or n (%). SAPS 3: Simplified Acute Physiology Score; COPD: chronic obstructive pulmonary disease; PaO2: arterial oxygen partial pressure; FiO2: inspired fraction of oxygen; PEEP: positive end-expiratory pressure; PP: prone position; SP; supine position; IMV: invasive mechanical ventilation; ICU: Intensive Care Unit; NMB: neuromuscular blocker; OTT: orotracheal tube; UTI: urinary tract infection; VAP: ventilator associated pneumonia.
Of the total patients, 31 required at least one PPP cycle (36% of 86 patients with IMV). Compared with the patients requiring cycles of PP < 24 h, we observed no significant differences in terms of age, comorbidities, severity upon admission or administered antiinflammatory or antiviral treatment. However, they showed less tolerance to ventilation in SP in the 24 h following a PP cycle (61.29% versus 89.47%; p = 0.031). Despite an initially favorable response to PP in both groups, four patients with PPP (12.90% of 31 patients) failed to improve despite prolongation of the maneuver. Compared with the patients subjected to cycles of PP < 24 h, the patients with PPP had a longer hospital stay (41 versus 30 days; p = 0.023), more days of IMV (32 versus 20 days; p = 0.032), and a longer duration of NMB use (10.5 versus 3 days; p = 0.0002). With regard to the complications, the incidence of OTT obstruction was greater in the patients with PPP than in those subjected to PP < 24 h (48.39% versus 15%; p = 0.014) (Table 1).
On comparing the three situations (SP, PP and PPP), the patients subjected to PPP were seen to have more complications: increased gastric residual volume (5.88% in SP, 15% in PP and 32.26% in PPP; p = 0.023), OTT obstruction (11.43% in SP, 15% in PP and 48.39% in PPP; p = 0.0016), NMB use (54.29% in SP, 95% in PP and 96.77% in PPP; p < 0.0001) over longer maneuvering periods (2 days in SP, 3 in PP, and 10.5 days in PPP; p < 0.0001), a greater duration of IMV and more days of hospital stay (Table 2).
On the other hand, the development of healthcare-associated infections (HAIs) was seen to be more frequent among the patients requiring PP and PPP versus those ventilated in SP: bacteremias (37.14% in SP, 70% in PP, 74.19% in PPP; p = 0.005) and urinary tract infections (14.71% in SP, 30% in PP, 45.16% in PPP; p = 0.027).
Globally, the mortality rate in the ICU among all COVID-19 patients was 21.66% (29/134). The mortality rate in the patients with bilateral (double) pneumonia admitted to the DICM was 22.22% (24/108), and all of them required IMV. Lastly, the mortality rate among the patients ventilated in PP and PPP was 30% and 38.61%, respectively (p = 0.525) (Table 1, Fig. 1).
DiscussionThe special circumstances seen in the ICUs during the pandemic gave rise to changes in the kind of patient care provided, including particularly the use of ventilation in PP for prolonged periods of time. This has generated much interest in knowing the outcomes of this maneuver, such as the impact of oxygenation upon the patients,11,12,14,15 the work load,15,16 the development of pressure ulcers,10,11,14,16 long-term neuropathic consequences,16 and the loss or accidental dysfunction of invasive devices.10
The present study describes the outcomes of 108 patients admitted to the DICM due to SARS-CoV-2 infection between March 2020 and December 2021. As in other studies, most of the patients requiring IMV due to bilateral pneumonia secondary to COVID-19 were males (73%) in the sixth decade of life (median 63.5 years).10–16 Up to 60% required PP, which is in contrast to the data published before the pandemic, when the reported incidence of PP was about 16% in patients with severe ARDS.20 The data referred to the number of prone cycles required by the patients were similar to those reported in other studies10,11,13,14 (median 2 cycles), though the patients with PPP received significantly more cycles. With regard to severity, and considering the PaO2/FiO2 ratio prior to the first PP cycle, the patients were seen to exhibit similar severity, with a similar initial response. The patients subjected to PPP were characterized by poorer tolerance to SP (defined as the possibility of maintaining PaO2/FiO2 > 150 in the supine position over the 24 h following prone maneuvering), a longer duration of IMV, more PP cycles, a longer hospital and ICU stay, and a greater prevalence of complications (Table 1) – though with no greater incidence of HAIs. These same differences were recorded with respect to the patients ventilated in SP, and the number of urinary infections and bacteremias was also higher (Table 2). These outcomes were seen to be progressively more accentuated among the patients ventilated in SP, PP < 24 h and PPP (Table 2).
The published data on the use of PPP during the pandemic vary greatly, with periods of up to 5 days and involving one14 or more cycles.10–12,15,16 The described benefits include particularly sustained improvement of oxygenation11,12,14,15 and the possibility of reducing the professional work overload.10,12,15,16 In our study, we recorded an increased use of resources such as the administration of NMBs or tracheostomy, as well as a longer duration of IMV and of hospital stay. In addition, we documented a progressively greater incidence of adverse events such as OTT obstruction and HAIs, conditioned to an increased need for prolonged PP (Tables 1 and 2).
Increased NMB use has been reported in ventilated patients with severe ARDS due to COVID-19.11,21 The current treatment guidelines suggest that NMB should be limited to those patients in which a lung protective ventilation protocol cannot be applied due to asynchrony or increased breathing effort, and involving periods of no longer than 48 h.22 In our patients ventilated in PP, the use of NMBs was also greater than before the pandemic,20 and the duration of treatment was longer than recommended by the clinical practice guides,22 though not so in the case of the patients ventilated in SP. Among the patients ventilated in PP < 24 h, this treatment lasted 24 h more than recommended (up to 3 days) (Tables 1 and 2). However, it was in the PPP group where the use of NMBs was prolonged to a significant extent (3 versus 10.5 days; p = 0.0002) (Table 1). Possibly, such prolonged use may be more related to a longer duration of IMV than to PPP or a greater total number of PP cycles, though many other confounding factors not considered in this study may also be involved, such as other data referred to organ failure, or deeper sedation protocols.
With regard to the rest of complications associated with PP, mention must be made of OTT obstruction, particularly in the patients subjected to PPP (Tables 1 and 2). We recorded a progressive increase in risk: 11.43% among patients ventilated in SP, 15% in PP, and 48.39% in PPP (p = 0.0016). This circumstance has already been described as a potential complication of IMV in PP, though to a lesser degree (up to 6.4% in the study published by González-Seguel et al.7 and up to 5% in the PROSEVA trial5). Obstruction of the OTT with mucus- and blood-containing secretions has been associated with patients with pneumonia due to SARS-CoV-2, in the context of the hypercoagulability state that characterizes the disease,23 and patients ventilated in PP are reported to suffer a greater incidence of retained respiratory secretions (up to 12% of all patients).16 It should be noted that the same type of humidifying device (passive) was used in all the analyzed patients, and this did not seem to affect the prevalence of airway occlusion compared with the use of active humidifying systems, as indicated by the current evidence.24 These three circumstances may have played a role in the large number of complications in the patients with PPP.
With regard to the difficulties of enteral nutrition (EN), an increase in gastric residual volume has been reported in PP.25 It has been postulated that the use of NMBs may play a role in this respect, though these drugs do not exert a paralyzing effect upon the intestinal muscle. The mentioned difficulties are therefore attributed to the underlying disease condition and to the concomitant use of vasopressor drugs or high sedation doses.25 This same effect was observed in our study, and was progressively greater in patients ventilated in SP, PP < 24 h and PPP, respectively (5.88% in SP, 15% in PP < 24 h, 32.26% in PPP; p = 0.023)(Table 1), though it must be noted that the gastric residual volume was lower than the currently recommended value of 500 ml.26
In relation to HAIs, these problems were seen to be more common in the patients requiring PP than in those ventilated in SP (Table 2), though no differences were observed according to the duration of the maneuver (Table 1). Such infections were thus probably related to the maneuver as such, not to its duration. The protocols for the prevention of nosocomial infections9 were applied within the context of the great difficulties posed by nursing staff turnover and the staff training possibilities. As in another Spanish study, the most frequent infections were bacteremias and urinary tract infections, and the patients requiring PP and greater use of NMBs also exhibited a greater percentage of HAIs.27
The results obtained must be interpreted within the limitations of a retrospective study involving a limited sample size. This is not an interventional study but a descriptive analysis of the characteristics and outcomes of the patients of a single center. The baseline characteristics and situation at the start of the disease (SAPS 3) and the PaO2/FiO2 ratio of the patients were similar — suggesting that the excessive work burden was what complicated routine nursing care (hygiene, humidifying system replacement) or timely patient postural changes. Other factors such as deeper sedation may also have influenced the poorer outcomes recorded in the PPP group, in addition to the greater evolutive severity of the patients that required PPP (poorer tolerance to SP).
Another limitation of our study is the fact that we did not include joint lesions or pressure ulcers among the complications, even though these problems have been widely described. Likewise, we conducted no long-term follow-up, which would have been interesting to gain a perspective over time of the described effects, particularly at neuromuscular level. Nevertheless, we believe that the results obtained serve to highlight potential points for improvement in the complex management of these patients.
ConclusionsProlonged PP was associated with an increased use of resources (ICU and hospital stay, longer duration of IMV) and more complications and procedures (tracheotomy, NMB, problems with enteral nutrition), though no differences were observed in terms of patient survival.
Author contributionsEva de Miguel-Balsa: Conceptualization, data analysis, literature review and review of the manuscript.
Teresa Blasco-Ruso: Data compilation and analysis, drafting of the manuscript.
Norma Gómez-Medrano: Conceptualization, review of the manuscript.
María Mirabet-Guijarro: Data compilation, review of the manuscript.
Alba Martínez-Pérez: Conceptualization, literature review and review of the manuscript.
Adoración Alcalá-López: Data management, review of the manuscript.
Conflicts of interestThe authors declare that they have no conflicts of interest.
AcknowledgementThe authors acknowledge the work of the entire intensive care unit staff and their dedication to the patients during the pandemic.