Contrast-induced nephropathy (CIN) has been considered one of the leading causes of hospital-acquired acute kidney injury (AKI). However, over the last few years, several studies suggest that this risk has been overestimated.1,2 SEMICYUC Working Group for Intensive Nephrologic Care conducted the NEFROCON study. It is a multicenter, prospective, descriptive clinical trial that included patients admitted to the ICU treated with a first radiological assessment with contrast for 3 months between 2012 and 2013.
CIN is defined as increased creatinine levels >0.5 mg/dL or >50% in serum creatinine 72 h after the administration of contrast.3
The study was approved by Hospital General Universitario de Castellón Clinical Research Ethics Committee (CREC). Each CREC from each participant center established the need for patients’ informed consent forms to participate in the study.
Non-parametric tests were used for the univariate analysis. Categorical variables were analyzed using the chi-square test. Prognostic factors were analyzed using the backward stepwise multivariate logistic regression analysis. To study the effect of prophylaxis and the different treatments of CIN, a multivariate model through multiple logistics regression was built. Afterwards, a propensity score was estimated.
A total of 1012 cases from 33 different hospitals were studied. The demographic characteristics of the study population, the type of contrast used, and the presence of risk factors for the development of AKI are shown on Table 1 of the supplementary data. Preventive measures were implemented in 29.4% of these patients (18.6% of coronary patients, and 38.5% of non-coronary patients, P = .001). Following the recommendations published in the KDIGO clinical guidelines,4 fluid therapy was administered (saline solution at 0.9% or bicarbonate) to 76.8% of the cases, although the administration of an IV saline solution (72.1%) was more common. A total of 23.2% of the patients received N-acetylcysteine.
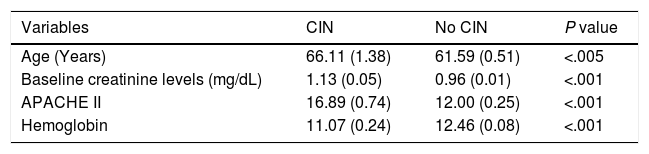
The analysis of the variables associated with the development of CIN is shown on Table 1. In our population, a total of 12.1% of the patients ended up developing CIN with clear differences between coronary patients (8.2%) and the remaining critically ill patients (15.3%), P = .0001. The inter-group different characteristics are shown on Table 2.
Development of CIN associated with the variables analyzed. Univariate analysis.
| Variables | CIN | No CIN | P value |
|---|---|---|---|
| Age (Years) | 66.11 (1.38) | 61.59 (0.51) | <.005 |
| Baseline creatinine levels (mg/dL) | 1.13 (0.05) | 0.96 (0.01) | <.001 |
| APACHE II | 16.89 (0.74) | 12.00 (0.25) | <.001 |
| Hemoglobin | 11.07 (0.24) | 12.46 (0.08) | <.001 |
| Risk factors | Odds ratio | 95%CI |
|---|---|---|
| Hepatic cirrhosis | 3.29 | (IC 1.58–6.84) |
| Heart failure | 2.65 | (IC 1.48–4.77) |
| Transplant | 7.45 | (IC 1.49–37.31) |
| CKD ≥ Grado II | 3.1 | (IC 1.76–5.46) |
| Diuretics | 2.94 | (IC 1.97–4.39) |
| Vasoactive drugs | 3.12 | (IC 2.08–4.67) |
| Shock | 4.26 | (IC 2.86–6.33) |
| AMI | 0.55 | (IC 0.35–0.86) |
95%CI, 95% confidence interval; AMI, acute myocardial infarction; APACHE, Acute Physiology and Chronic Health Evaluation; CIN, contrast-induced nephropathy; CKD, chronic kidney disease.
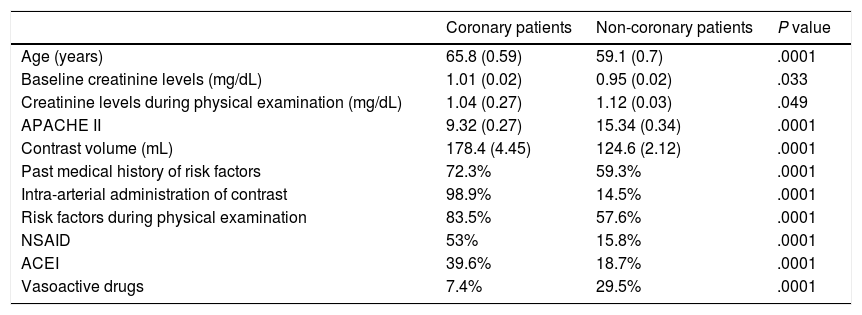
Differential characteristics between coronary and non-coronary patients.
| Coronary patients | Non-coronary patients | P value | |
|---|---|---|---|
| Age (years) | 65.8 (0.59) | 59.1 (0.7) | .0001 |
| Baseline creatinine levels (mg/dL) | 1.01 (0.02) | 0.95 (0.02) | .033 |
| Creatinine levels during physical examination (mg/dL) | 1.04 (0.27) | 1.12 (0.03) | .049 |
| APACHE II | 9.32 (0.27) | 15.34 (0.34) | .0001 |
| Contrast volume (mL) | 178.4 (4.45) | 124.6 (2.12) | .0001 |
| Past medical history of risk factors | 72.3% | 59.3% | .0001 |
| Intra-arterial administration of contrast | 98.9% | 14.5% | .0001 |
| Risk factors during physical examination | 83.5% | 57.6% | .0001 |
| NSAID | 53% | 15.8% | .0001 |
| ACEI | 39.6% | 18.7% | .0001 |
| Vasoactive drugs | 7.4% | 29.5% | .0001 |
ACEI, angiotensin-converting enzyme inhibitors; APACHE, Acute Physiology and Chronic Health Evaluation; NSAID, non-steroidal anti-inflammatory drugs.
In the multivariate analysis, the risk factors independently associated with the development of CIN were the creatinine levels at baseline, the APACHE II score, the hemoglobin levels, the administration of diuretics, and the presence of shock (Table 2 of the supplementary data). However, when propensity score data were analyzed, we could only confirm an increased rate of CIN in patients who received diuretics during the physical examination (OR, 1.89; 95%CI, 1.17–3.04), which was also associated with a significantly higher mortality rate (OR, 1.85; 95%CI 1.13–3.03).
In our series we had a higher rate of CIN in patients with some risk factor to develop AKI (OR, 1.83; 95%CI, 1.18–2.83). In general, these risk factors are characterized by being pathophysiological processes that limit the capacity of kidneys to properly compensate contrast-induced hemodynamic and microcirculatory stress. Among them chronic kidney disease (CKD) is considered the most powerful predictor of CIN.5 The incidence rate is above average with more advanced stages, basically starting at stage II. The effect of risk factors is additive, and the chances of developing NIC are higher with more risk factors. In our series, we had an incidence rate of 13.7% in patients with, at least, 1 risk factor vs 8.4% in patients without any risk factors, being diuretics the most important of all. Furosemide can cause systemic hemodynamic changes (also at kidney level) that can exacerbate contrast-induced changes. A reduced cortical vascular resistance can divert blood from the spinal cord circulation.6 Added to contrast-induced vasoconstriction, this can reduce the oxygen tension of the spinal nephrons down to critical levels causing ischemic damage. This worsening renal function can be associated with a higher mortality rate described, which would justify the recommendation of not using furosemide as prophylaxis or to treat AKI.7
When coronary patients were analyzed (Table 2), it was striking to see that, despite having more risk factors and fewer preventive measures implemented, the rate of CIN is lower. These results may be justified by the different severity profile and the lower APACHE II scores compared to the group of non-coronary patients. In addition to being widely used as a predictor of mortality in critically ill patients, this score can be useful as a predictor of the development of AKI in association with the severity of the patient’s condition.
Regarding preventive measures, the expansion of intravascular volume with IV crystalloids could counteract the effect of contrast-induced renal vasoconstriction, thus reducing its concentration and viscosity in the tubular lumen. However, a certain number of patients still end up developing kidney failure. Consistent with this, several studies recently published failed to show any beneficial effects following the administration of a saline solution, bicarbonate or n-acetylcysteine to prevent CIN.8,9
Maybe one of the most important limitations of the study was the lack of a control group, as well as the lack of clinical follow-up to establish disease progression the in the long-term. Despite the time elapsed until the publication of the study—the types of contrast used were recommended due to their lower nephrotoxicity (iso-osmolar, and hypo-osmolar contrast)—hyper-osmolar contrast was used in 4.2% of the cases only. Other biases are that the analysis was conducted on a very heterogeneous population and in different centers. Although this may undermine the study statistical power, it brings more external validity since the results are more generalizable. Regarding the effect on results, a multilevel analysis was conducted that did not show any center-based effects. Also, coronary, and non-coronary patients were studied separately using a logistic regression model. Since we only analyzed the index event, we cannot determine whether the repetition of successive examinations in some patients may have influenced the mortality outcomes. Finally, the application of prophylaxis was highly heterogeneous and left to the operator’s criterion since it was not a controlled study although this bias was somehow controlled through the propensity score.
Given its pathophysiological complexity, the multifactorial origin of AKI in the intensive care setting,10 and the multiple factors included in its pathogenesis it is expected that the results from the latest studies being conducted will help determine its true toxicity and whether the administration of iodinated contrast is another risk factor that can lead to a worsening renal function in critically ill patients. In this context, diuretics stand as a significant risk factor that increases the rates of CIN and mortality.
FundingNone whatsoever.
Conflicts of interestNone reported.
Please cite this article as: Mas-Font S, Herrera-Gutierrez ME, Gómez-González C, Herrera-Rojas D, Montoiro-Allue R, Sánchez-Morán F, et al. Estudio epidemiológico prospectivo sobre la nefropatía por contraste en pacientes críticos. Estudio NEFROCON. Med Intensiva. 2021;45:e31–e33.