Mortality among asthmatic patients has decreased substantially thanks mainly to advances in drug treatment. Nevertheless, in some cases and despite optimum pharmacological treatment, invasive ventilatory support proves necessary. The risk of barotrauma, which can have fatal consequences, is directly related to pulmonary insufflation secondary to air trapping and an increase in plateau pressure (Pp) – prolonged expiratory times and even disconnection from the respiratory being needed to lower the intrinsic positive end-expiratory pressure (PEEP).1 The mortality risk among patients with severe asthma requiring invasive mechanical ventilation is 6.5–10.3%.2
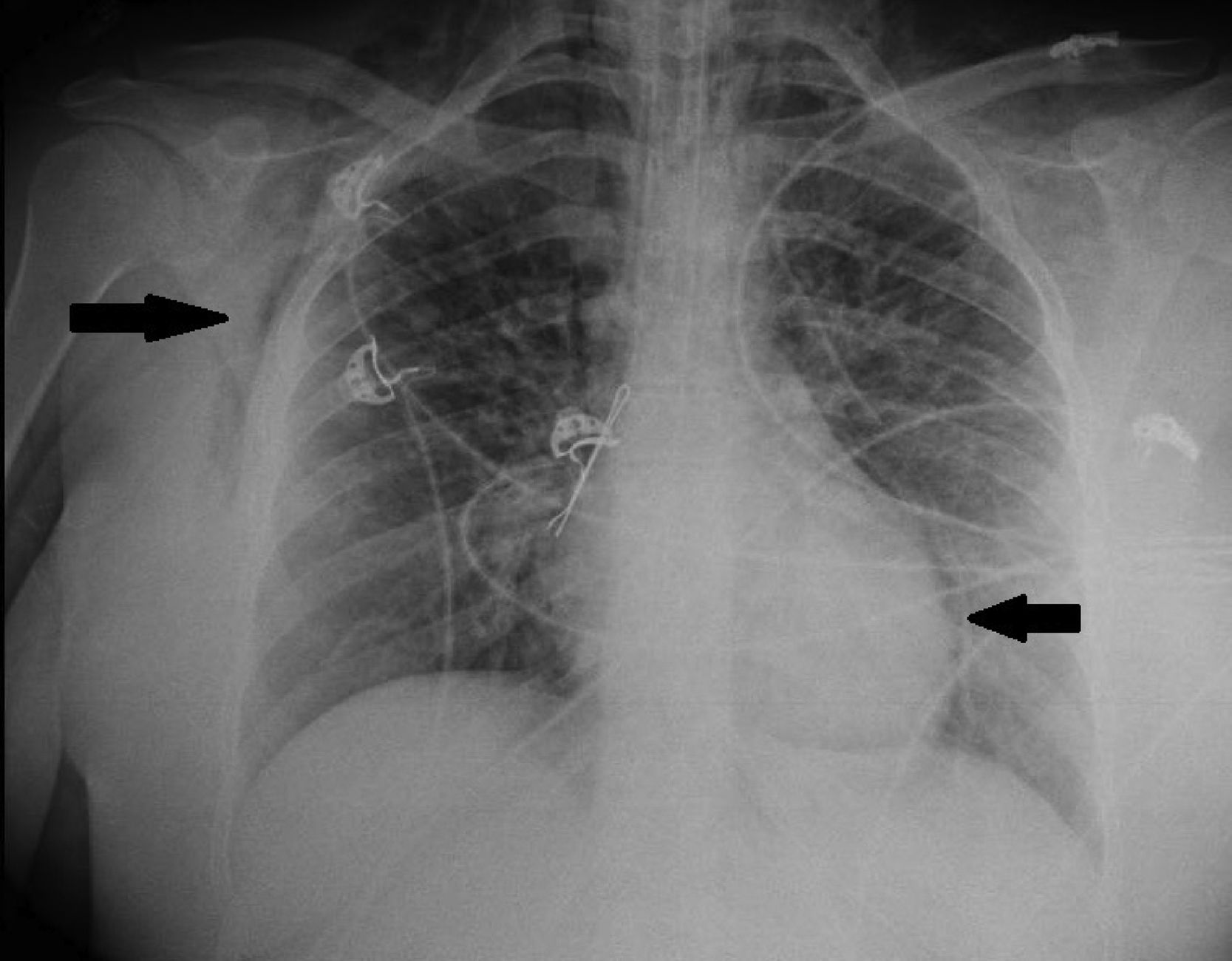
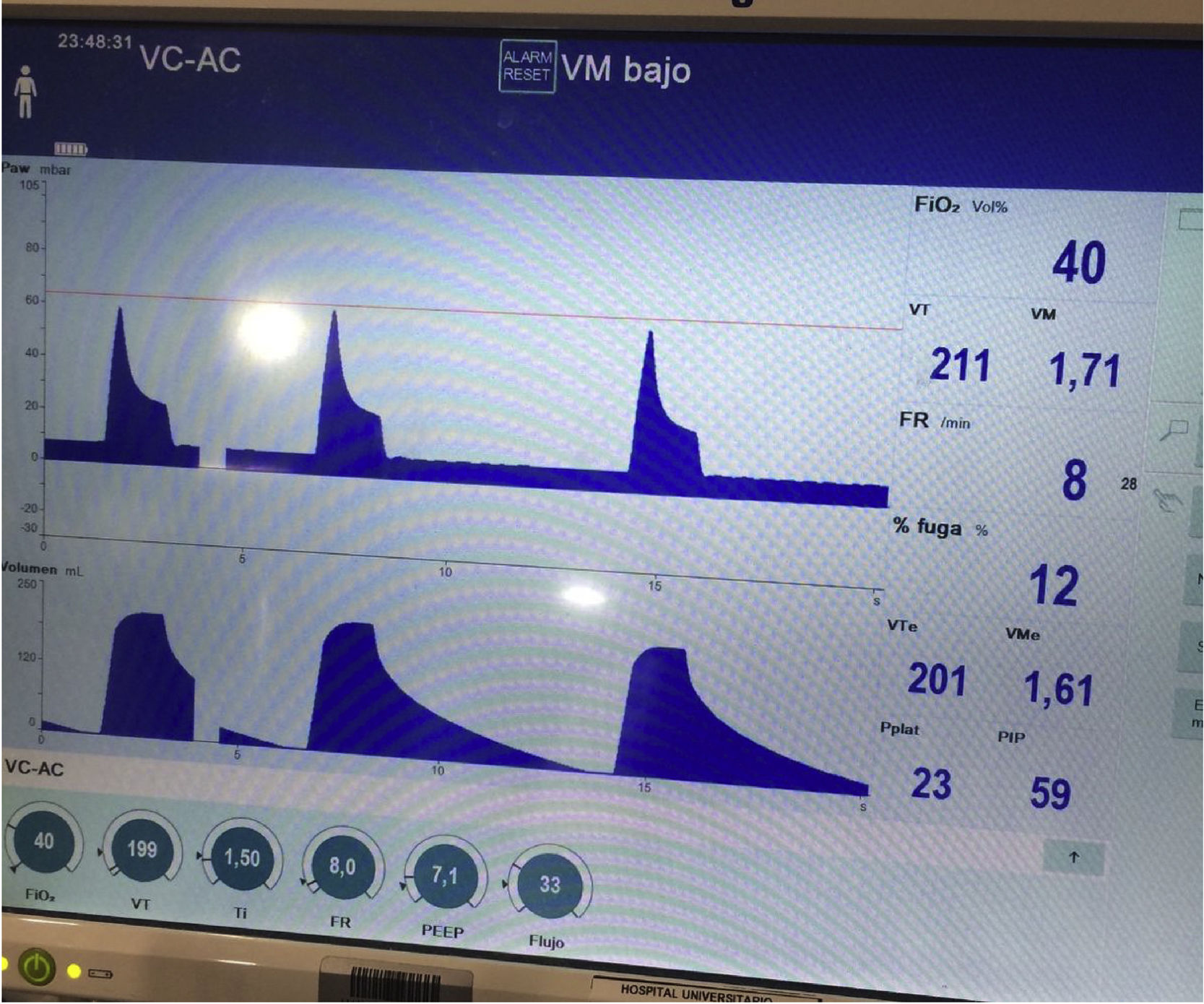
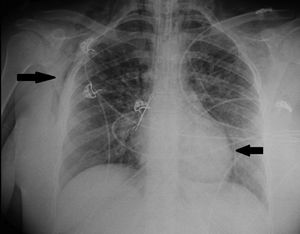
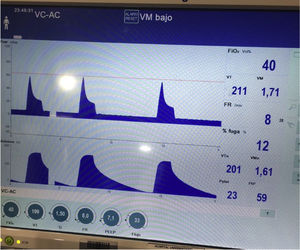
We present the case of a 21-year-old woman and smoker of tobacco and cannabis, with a history of extrinsic intermittent asthma since childhood. The patient was admitted to the Intensive Care Unit of our hospital due to a severe asthma attack that failed to respond to intensive drug treatment, and noninvasive mechanical ventilation (MV) proved necessary. Under conditions of muscle exhaustion with respiratory acidosis (pH 7.29, PaCO2 63.2mmHg), invasive MV was decided from the first three hours of admission to intensive care. During the first days of her stay, deep sedation-relaxation proved necessary (midazolam, fentanyl and cisatracurium), with very high-dose inhaled bronchodilators (salbutamol 5mg/h, ipratropium 0.5mg/h, adrenalin 1mg/2h), intravenous corticosteroids (6-methylprednisolone 80mg/8h) and intravenous rescue medication in the form of ketamine 60mg/kg/min in continuous perfusion, magnesium sulfate 12g/24h and theophylline 0.8mg/kg/h. Following these measures, severe airflow obstruction was seen to persist, however, with air trapping despite protective ventilation with permissive hypercapnia: under a tidal volume (Vt) of 440ml, an inspiration/expiration ratio of 1:4, a flow of 36l/min, PEEP 7mbar and FiO2 0.5, a peak pressure of 55–60mbar was generated, with Pp 35mbar, compliance 23ml/mbar and total PEEP 16mbar. After 72h of admission, the patient showed evidence of barotrauma (pneumopericardium, with interstitial and subcutaneous emphysema [Fig. 1]), associated to progressive severe respiratory acidosis (pH 7.14 and PaCO2 124mmHg). At this point we decided to start femoro-jugular veno-venous extracorporeal membrane oxygenation (VV-ECMO) in order to perform ultraprotective ventilation with lung rest. Over the next few days the respiratory obstruction persisted (Fig. 2), together with very poor lung distensibility (Vt<4ml/kg, inspiration/expiration ratio 1:4, compliance 28ml/mbar, intrinsic PEEP 9mbar), and with the continued need for high-dose drug treatment, as well as fibrobronchoscopic bronchoalveolar lavage (BAL) and lung voiding maneuvers. After five days of ECMO the obstruction subsided, allowing slow modification of the ventilatory parameters, and with weaning from ECMO by day 7. The subsequent clinical course was slow but favorable, and the patient was discharged after 25 days of admission. As associated complications, she suffered respiratory infection caused by methicillin-sensitive S. aureus and critical patient myopathy.
Veno-venous extracorporeal membrane oxygenation is a temporary respiratory assist strategy for the management of respiratory failure refractory to conventional treatment. It improves oxygenation, reduces the CO2 levels, and lessens the risk of ventilator associated lung injury thanks to the provision of protective (Vt 4–6ml/kg) or ultraprotective ventilation (Vt<4ml/kg).3
The use of VV-ECMO could improve the prognosis of patients with severe hypoxemic acute respiratory failure, as in acute respiratory distress syndrome (ARDS), though it is still regarded as a rescue treatment strategy lacking sufficient scientific evidence.4
The evidence on the use of VV-ECMO in adult near-fatal status asthmaticus is limited to a number of published case series.5,6 The Extracorporeal Life Support Organization described the evolution of patients with status asthmaticus administered supportive treatment with ECMO in its international registry between 1986–2006.7 Of the 1257 patients requiring respiratory support, only 24 corresponded to severe asthma cases – ECMO being performed with a mean pH of 7.14 (standard deviation [SD] 0.16) and a mean PaCO2 of 119.7mmHg (SD 58.1). This has been the largest series published to date, with a survival rate of 83.3%. However, despite the good results obtained, the technique was associated with significant complications: cannula-related or pulmonary bleeding in 37.5% of the cases, infections in 8.3%, circuit-related complications in 41.4%, and even intracranial bleeding and brain death in 12.5%. In a recent series, Di Lascio et al. studied 16 adults with near-fatal asthma during 5 years and subjected to ECMO support.8 Prior to the start of ECMO, the mean pH was 6.89 (SD 0.014), with a mean PaCO2 of 111mmHg (SD 4.24), and the patients underwent fibrobronchoscopic bronchoalveolar lavage on a daily basis. All the patients survived, and there were no relevant complications.
Specific devices have recently been developed for the extracorporeal elimination of CO2 that are technically less complicated than ECMO, and which could prove useful in patients requiring lung rest.9 The use of such devices has been considered in ARDS and in exacerbated chronic obstructive pulmonary disease (COPD), though experience in cases of status asthmaticus is still limited.10
On analyzing our case and the published series on status asthmaticus, in view of its reversible nature, ventilatory support with extracorporeal assist could be regarded as an early treatment option, particularly in the more aggressive or near-fatal presentations of the disorder. However, we still need to define the specific clinical or blood gas criteria for introducing ECMO in cases refractory to conventional management, as in the patients with ARDS. On the other hand, ECMO has frequent complications, and careful assessment of the risk–benefit balance of each individual patient is required, without being able to predict the moment in which bronchospasm will subside.
In our patient, assist was decided when we observed incipient signs of barotrauma, despite the maintenance of moderate permissive hypercapnia. Even so, 5 days of ECMO with Vt<4ml/kg were needed in order for bronchospasm to subside. Fortunately, we will never know the risk we would have had to accept had more conventional ventilatory management been used.
Please cite this article as: Corcia-Palomo Y, Martín-Villén L, Escalona-Rodríguez S, Roldán Reina A, Adsuar-Gómez A, Martín-Bermúdez R. Membrana de oxigenación extracorpórea (ECMO) y ventilación mecánica ultraprotectora en el estatus asmático casi fatal. Med Intensiva. 2018;42:556–558.