To determine mortality prognostic factors in elderly patients who are admitted to intensive care units (ICUs) due to acute critical illness.
DesignA prospective cohort study was carried out.
SettingA polyvalent intensive care unit at a University Hospital in Argentina.
Patients or participantsWe included 249 patients over 65years of age who were consecutively admitted to the ICU and required mechanical ventilation for more than 48h, between January 2011 and December 2012. Patients with degenerative neurological disease, limitation of therapeutic effort or on chronic mechanical ventilation were excluded.
Principal variables of interestIn-hospital mortality, comorbidity (Charlson index), APACHE II score, and pre-acute illness status were recorded: nutritional status (subjective global assessment), functionality (activities of daily living [ADL] and Barthel index), cognitive abilities (Short Reporting Questionnaire on Cognitive Decline in the Elderly [S_IQCODE]) and quality of life (EQ-5D).
ResultsThe in-hospital mortality rate was 52%. Logistic regression analysis, after adjusting for APACHE II score and age, identified the following independent variables associated to mortality: male gender (OR: 2.46, 95%CI: 1.37–4.42), moderate malnutrition (OR: 2.07, 95%CI: 1.09–3.94), severe malnutrition (OR: 2.20, 95%CI: 1.06–4.59), and ADL<6 (OR: 2.35, 95%CI: 1.16–4.75).
ConclusionsIn our study, chronological age was not associated to in-hospital mortality. However, loss of functional independence (assessed by ADL) and malnourishment were shown to be strong prognostic factors; knowing these baseline characteristics from ICU admission would be useful when making decisions regarding the intensity of treatment.
Determinar factores pronóstico para mortalidad en los pacientes ancianos que se internan en unidades de cuidados intensivos (UCI) por enfermedad crítica aguda.
DiseñoEstudio de cohorte prospectiva.
ÁmbitoUnidad de Terapia Intensiva polivalente en Hospital Universitario en Argentina.
Pacientes o participantesSe incluyeron 249 pacientes mayores de 65años que ingresaron en forma consecutiva a la UCI con requerimiento de asistencia ventilatoria mecánica durante más de 48h entre enero de 2011 y diciembre de 2012. Fueron excluidos pacientes con enfermedad neurológica degenerativa, limitación de esfuerzos terapéuticos y ventilación mecánica crónica.
Variables de interés principalesSe registró la mortalidad hospitalaria, la carga de comorbilidad (índice de Charlson), APACHEII y estado previo a la enfermedad aguda: estado nutricional (valoración global subjetiva), funcionalidad (actividades de la vida diaria [AVD] o índice de Katz y Barthel), habilidades cognitivas (Short Informant Questionnaire on Cognitive Decline in the Elderly [S_IQCODE]) y calidad de vida (EQ-5D).
ResultadosLa mortalidad hospitalaria fue del 52%. Por regresión logística, y después de ajustar por APACHEII y edad, resultaron variables independientes asociadas a mortalidad el sexo masculino, con un odds ratio (OR) de 2,46 (intervalo de confianza [IC] del 95%: 1,37–4,42), la desnutrición moderada (OR: 2,07, IC95%: 1,09–3,94), la desnutrición severa (OR: 2,20, IC95%: 1,06–4,59) y un AVD<6 (OR: 2,35, IC95%: 1,16–4,75).
ConclusionesEn nuestro estudio, la edad cronológica no resultó asociada a la mortalidad hospitalaria. Sin embargo, la pérdida de la independencia funcional (evaluada por AVD) y la desnutrición mostraron ser factores pronóstico. Conocer estos datos desde el ingreso por enfermedad crítica sería de utilidad a la hora de tomar decisiones relevantes respecto a la intensidad del tratamiento a instituir.
The impact of advanced age upon the prognosis of patients admitted to the intensive care unit (ICU) has been the subject of research and controversy for several decades.1 Studies involving adult patients in general have found advanced age to be an independent indicator of poor prognosis.2,3 The higher mortality rate in the elderly has been attributed to the presence of serious comorbidities such as cancer, or to a higher prevalence of complications associated to the main disease process.4 However, in those studies that have analyzed age as a prognostic factor specifically among populations of elderly patients of different ages, the results obtained are not so clear. In some investigations mortality was found to be higher among the oldest subjects,5 while other studies found mortality to be more closely related to the comorbidities and acute disease condition giving rise to admission than to patient age as such.6,7 Based on the above, it is not clear whether the unfavorable evolution of elderly patients admitted to the ICU is fundamentally attributable to the normal aging process, the previous health condition of the patient, or the acute disease condition itself.8,9
A potential source of bias that may have influenced the previous studies is the degree of therapeutic effort applied in caring for the elderly. In some cases, elderly patients are less often admitted to the ICU,10 and in others they receive less intensive care.6,11 Furthermore, the provision of limited care with the purpose of avoiding futile treatment is particularly common among such patients.12,13
In intensive care, the main elements of discussion referred to the limitation of therapeutic effort are patient age and the quality of life patients can be offered after overcoming the critical period, or their possibilities of surviving the acute event.14 However, many of these decisions are speculative, since it is difficult to accurately predict the outcome once the patient has received intensive care.15 Further studies are needed in this field, particularly addressing patient age, previous health condition and the evolution of elderly individuals treated in the ICU due to very serious acute disease.16
Considering the above, we conducted a prospective study of critically ill patients over 65 years of age not subjected to limitation of therapeutic effort, with the purpose of evaluating mortality risk factors, including functionality or performance status and cognitive function prior to the acute disease condition.
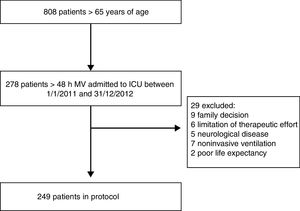
Patients and methodsA prospective cohort study was carried out in a polyvalent ICU with 38 beds belonging to a University Hospital in the city of Buenos Aires (Argentina). A total of 278 elderly patients consecutively admitted to the ICU between 1 January 2011 and 31 December 2012 were screened for inclusion in the study. Elderly patients were defined as those aged ≥65 years. As a surrogate of critical disease we used the need for mechanical ventilation (MV) for 48h or more, secondary to acute illness or complicated postoperative periods of major surgery. Patients with chronic degenerative diseases were excluded, as were those subjected to prolonged MV and those with limitation of therapeutic effort or a low probability of survival. Twenty-nine patients were excluded on the basis of these criteria, leaving a total of 249 patients for the final analysis (Fig. 1).
The study protocol was approved by the Research Ethics Committee of the Italian Hospital of Buenos Aires (protocol no. 1687), and informed consent was requested from the patient relatives upon admission to the ICU.
The primary study objective was to determine the variables associated to in-hospital mortality among these patients and to assess the impact of age upon this outcome. We recorded demographic characteristics, comorbidity burden based on the Charlson comorbidity index,17 severity upon admission based on the APACHE II score,18 nutritional condition, performance status, cognitive skills and quality of life before the acute disease giving rise to admission. Performance status was assessed using the Katz index of independence in activities of daily living (ADL)19 and the Barthel scale,20 while cognitive capacity was explored by means of the Short Informant Questionnaire on Cognitive Decline in the Elderly (S-IQCODE),21 and patient quality of life was evaluated with the EQ5D.22
The ADL score ranged from 0 to 6 (from minimum to maximum independence). Due to the large proportion of patients yielding the maximum score, the variable was transformed into a dichotomic parameter for analysis: “independent” (ADL=6 points) or “not independent” (ADL<6 points). The Barthel index (BI) was scored from 0 to 100 (from full dependence to independence). To the effects of analysis, the patients were classified as High Barthel (BI≥85) or Low Barthel (BI<85).23 In relation to quality of life, the EQ5D scores were adjusted to a continuous time trade-off (TTO) scale24 where 1 represented maximum quality of life and 0 corresponded to imminent death. This scale assesses the remaining life the patient is willing to exchange for perfect health condition. Nutritional condition in turn was evaluated in the first 48h of admission to the ICU, and the patients were classified by the Subjective Global Assessment (SGA)25 as A (well nourished), B (moderate malnutrition) or C (severe malnutrition).
We evaluated the appearance of complications during admission to the ICU, including ventilator associated pneumonia according to the definition of the American Thoracic Society of 2005 (ATS/IDSA), in effect at the time of patient enrollment26; acute renal failure as established by the AKIN criteria27; acquired muscle weakness as defined by a cumulative Medical Research Council (MRC) score of <48 points28; and septic shock according to the guidelines of the Surviving Sepsis Campaign 2008.29 We also recorded the ventilatory parameters of all the patients based on a ventilation monitoring sheet routinely used in the Unit, and which is entered in the electronic case history.
A mortality rate of close to 40% was considered, based on previous studies. By evaluating 250 patients, the number of events (defined as deaths during hospital stay) was calculated as being sufficient to enter 10 variables in the regression model, including patient age and the APACHE II score, assuming that 10 events are required for each variable.30
The descriptive study expressed categorical variables as frequencies and percentages, while numerical variables were reported as the mean and standard deviation (SD), or as the median and interquartile range (IQR), depending on the distribution of the data.
The differences between the groups of patients that died during admission and those discharged live were analyzed using the Student t-test for continuous variables with a normal distribution, while the Mann–Whitney U-test and Wilcoxon test were used for quantitative variables with a non-normal distribution but similar between levels or ordinals. The median test was used for quantitative variables with a different distribution, and the chi-squared test was applied in the case of categorical variables.
Logistic regression models were developed, using mortality during hospital stay as dependent variable. As predictive variables we used those yielding p<0.20 in the univariate analysis, as well as patient age and APACHE II score upon admission, due to their biological and theoretical importance. The variables were entered in the model using the forward selection method – entry firstly being determined by the Wald test and secondly by the likelihood ratio test. We then evaluated the presence of interactions between the entered variables, generating new variables as the product between them, and evaluating the statistical significance of their respective regression coefficients based on the Wald test. The goodness of fit was assessed using the Hosmer Lemeshow test. We only considered models for which the likelihood ratio test proved significant while the Hosmer Lemeshow test proved nonsignificant. The existence of multi-colinearity between the entered variables was discarded, evaluating the variance inflation factor and using 2.5 points as cut-off value. The IBM SPSS version 21 statistical package was used throughout. Statistical significance was considered for p<0.05.
ResultsDuring the two-year study period we included 249 patients ≥65 years of age requiring over 48h of MV in the ICU. The median age of the population was 77 years (IQR: 70–84), and the mean APACHE II score upon admission was 20±7 points. Of the 249 patients, 184 (74%) were admitted due to clinical reasons, 24 (9%) due to complicated elective surgery, 34 (14%) due to emergency surgery, and 7 (3%) due to polytraumatism. The median comorbidity score of the study population as assessed by the Charlson index was 6 points (IQR: 4–8). The mortality rate during admission to the ICU was 48% (n=119), and another 11 patients died in the course of hospital stay after discharge from the ICU.
All the patients were subjected to MV and were intubated via the orotracheal route at the start, with a median duration of MV of 10 days (IQR: 5–21). A total of 194 patients (78%) were successfully weaned. Tracheostomy was performed in 82 patients (33%), and the mean tracheostomy time from the start of MV was 8 days (IQR: 6–11). There was a greater proportion of tracheostomized patients among the survivors than among those who died (47% vs 25%, respectively; p<0.01).
Table 1 shows the results of the comparison of the baseline characteristics, performance status, quality of life and cognitive capacity before admission between the patients that died during hospital admission and those discharged alive from hospital. Statistical significance was only observed for the proportion of males and moderate or severe malnutrition among those who died during admission. The same analysis was performed with variables referred to clinical events during admission, including complications, ventilation parameters during the first 72h, and stays in the ICU and in hospital (Table 2). With regard to the baseline characteristics, we selected those yielding p<0.20 for entry in the model using the forward selection method: low ADL (<6 points), SGA B and C (category A being the reference), and the male gender. The presence of interactions between ADL<6 points and SGA B or C was discarded. Among the variables referred to outcomes during admission, only the need for hemodialysis was entered in the model, since the rest of the variables that reached statistical significance were not considered to be clinically relevant. Lastly, we entered the variables patient age and APACHE II score in the model due to their theoretical importance as mortality predictors in this population.
Baseline characteristics, cognitive function, performance status and quality of life before admission in the study cohort, and comparison between patients alive at discharge and patients who died during admission.
| Variables | Total (n=249) | Alive at discharge (n=119) | Died during admission (n=130) | p | |
|---|---|---|---|---|---|
| Age (years)a | 77 (70; 84) | 77 (70; 85) | 78 (70; 84) | 0.95 | |
| Maleb | 133 (53) | 52 (44) | 81 (62) | <0.01 | |
| APACHE IIc | 20±7 | 20±7 | 21±7 | 0.28 | |
| Reason for admissionb | Elective surgery | 23 (10) | 8 (7) | 15 (12) | 0.15 |
| Liver transplant | 6 (2) | 4 (3) | 2 (1) | ||
| Emergency surgery | 31 (13) | 19 (16) | 12 (9) | ||
| Nonsurgical acute abdomen | 16 (6) | 9 (8) | 7 (5) | ||
| Septic shock | 17 (7) | 6 (5) | 11 (9) | ||
| Pneumonia | 53 (21) | 24 (20) | 29 (23) | ||
| Exacerbated COPD | 11 (4) | 9 (8) | 2 (1) | ||
| Neurological | 46 (19) | 22 (18) | 24 (18) | ||
| Others | 33 (13) | 11 (9) | 22 (17) | ||
| Polytraumatism | 13 (5) | 7 (6) | 6 (5) | ||
| Charlson indexa | 6 (4; 8) | 6 (4; 8) | 6 (5; 8) | 0.24 | |
| SGAb | A | 97 (44) | 60 (54) | 37 (34) | <0.01 |
| B | 73 (33) | 33 (30) | 40 (37) | ||
| C | 51 (23) | 19 (17) | 32 (30) | ||
| S_IQCODEa | 3.19 (3.19; 3.38) | 3.19 (3.19; 3.31) | 3.19 (3.19; 3.39) | 0.96 | |
| ADL<6b | 61 (25) | 23 (19) | 38 (29) | 0.07 | |
| Barthel<85b | 54 (22) | 21 (18) | 33 (25) | 0.14 | |
| Quality of lifea (EQ5D/TTO) | 0.724 (0.604; 1) | 0.724 (0.612; 1) | 0.724 (0.519; 0.931) | 0.87 | |
ADL: activities of daily living (Katz); EQ5D: EuroQoL 5 dimensions expressed in time trade off (TTO); S_IQCODE: Short Form of the Informant Questionnaire on Cognitive Decline in the Elderly; SGA: Subjective Global Assessment.
Clinical outcomes during hospital admission.
| Variables | Total (n=249) | Alive at discharge (n=119) | Died during admission (n=130) | p |
|---|---|---|---|---|
| VAPa | 31 (12) | 14 (12) | 17 (13) | 0.75 |
| Septic shocka | 18 (7) | 6 (5) | 12 (9) | 0.20 |
| Acute renal failurea | 73 (29) | 36 (30) | 37 (29) | 0.76 |
| Renal replacement therapya | 52 (21) | 12 (10) | 40 (31) | <0.01 |
| Acquired muscle weaknessa | 95 (38) | 49 (41) | 46 (35) | 0.35 |
| Days on MVb | 10 (5; 21) | 9 (4; 27) | 11 (6; 19) | 0.45 |
| PEEPb | 6 (5; 7) | 6 (5; 7) | 6 (6; 7) | 0.86 |
| MAPb | 11 (10; 12) | 11 (9; 12) | 11 (10; 12) | 0.12 |
| Static complianceb | 43 (35; 55) | 42 (34; 48) | 46 (36; 60) | 0.06 |
| PaO2/FIO2b | 339 (273; 399) | 305 (241; 392) | 360 (294; 410) | 0.16 |
| SaO2/FIO2b | 248 (240; 263) | 250 (245; 278) | 246 (237; 250) | <0.01 |
| Driving pressureb | 12 (10; 15) | 13 (11; 15) | 11 (9; 14) | 0.03 |
| Tidal volume/kg theoretical weight in ml/kgb | 9 (8; 9) | 9 (8; 9) | 9 (8; 10) | <0.01 |
| Days of stay in ICUb | 16 (9; 29) | 18 (11; 36) | 14 (8; 22) | <0.01 |
| Days of hospital stayb | 26 (15; 48) | 37 (22; 64) | 19 (11; 33) | <0.01 |
| Death in ICUa | 109 (44) | 0 (0) | 109 (84) |
MAP: mean airway pressure in cmH2O; VAP: ventilator associated pneumonia; PEEP: positive end-expiratory pressure in cmH2O; ICU: intensive care unit; MV: mechanical ventilation.
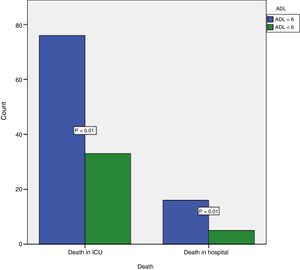
The final model with the raw and adjusted odds ratios (ORs) is shown in Table 3. An ADL score of <6 was associated to mortality in the ICU and in hospital (Fig. 2). The chi-squared statistic of the Hosmer–Lemeshow test was 9.99 (p=0.27) for the final regression model, and the existence of multi-colinearity between the independent variables was discarded. No significant −2 log likelihood ratio change was observed on removing the variable age from the model (268.9 in the model including age and 269 in the model excluding age).
In-hospital mortality odds ratios (raw and adjusted).
| Variable | Raw OR | Adjusted OR |
|---|---|---|
| Male gender | 2.13 (95%CI: 1.28–3.53; p<0.01) | 2.18 (95%CI: 1.19–3.99; p=0.01) |
| ADL<6 | 1.72 (95%CI: 0.95–3.11; p=0.07) | 2.47 (95%CI: 1.21–5.06; p=0.01) |
| SGA B | 1.97 (95%CI: 1.06–3.64; p=0.03) | 2.14 (95%CI: 1.10–4.15; p=0.02) |
| SGA C | 2.73 (95%CI: 1.36–5.51; p<0.01) | 2.12 (95%CI: 0.99–4.53; p=0.05) |
| RRT | 3.96 (95%CI: 1.96–8.01; p<0.01) | 3.97 (95%CI: 1.83–8.55; p<0.01) |
| Age | 1.01 (95%CI: 0.97–1.03; p=0.91) | 0.99 (95%CI: 0.96–1.03; p=0.79) |
| APACHE II | 1.02 (95%CI: 0.95–1.05; p=0.28) | 1.01 (95%CI: 0.97–1.05; p=0.77) |
ADL: activities of daily living; CI: confidence interval; RRT: renal replacement therapy; SGA: Subjective Global Assessment.
This prospective cohort study of critically ill elderly patients explored factors associated to in-hospital mortality capable of predicting outcomes and of facilitating optimum decision making referred to the required intensity of patient care.
The definition of elderly patient is very controversial, and the important heterogeneity of the existing definitions underscores the complexity of the problem. In many cases only the retirement age is reflected. We understand aging as an intrinsic and unavoidable degradation process affecting biological function, and which accumulates over time at all levels of biological organization – from molecules to populations. In this context, the limits between aging and disease are poorly defined, and although diseases have multiple defined causes, the effect of passing time appears to be one of them.31
In our study we arbitrarily defined “elderly” as a person over 65 years of age, since this is the retirement age in our country and also in most other South American countries. Furthermore, the Argentinean Ministry of Health, in the same way as the World Health Organization and the United Nations, uses 60 years as the cut-off point defining the elderly population.
Our study sought to evaluate the impact of chronological age upon in-hospital mortality among patients admitted due to acute critical illness. No influence was detected either in the univariate analysis or after adjusting for possible confounders. Many studies have reported an association between advancing age and increased mortality in the elderly population. The Study to Understand Prognosis and Preferences for Outcomes and Risks of Treatment (SUPPORT)2 found the probability of death to increase 2% with each additional year among patients over 70 years of age. Despite this, and although aging is inevitable, it is a multifactorial process in which the number of years as an absolute value would have different effects in different organisms.32
The observed in-hospital mortality rate was 52%, which is consistent with the rate expected from the APACHE II score but higher than in most retrospective studies that do not only include patients subjected to MV.33–35 Prospective studies that only assess elderly patients requiring MV offer similar results.36,37 On the other hand, most of the patients included in the study (74%) were admitted due to medical reasons, with a predominance of severe community-acquired pneumonia (21%), which is one of the infectious diseases with the highest associated mortality in this particular age group38 – a fact that also explains the severity and mortality in this population.
In searching for mortality predictors, and in addition to patient age, comorbidities, severity of the acute disease process and complications during admission, we also considered parameters referred to performance status, nutritional condition, cognitive function and previous quality of life. The male gender, worsened performance status (ADL<6), the presence of denutrition (SGA B or C) and the need for renal replacement therapy were identified as mortality predictors.
Seventy-five percent of the patients in our cohort had an ADL score of 6 points, indicating full independence for activities of daily living, and 78% presented a Barthel index score of >85 points, indicating an acceptable level of independence. Only 5% (n=12) presented ADL≤1, and 2% (n=5) presented a Barthel index score of <20 points (complete dependence according to both indices). In our study, the loss of functional independence (regarded as a loss of at least one point in the ADL score) showed a strong correlation to in-hospital mortality (OR=2.5), in coincidence with the limited data published on this population to date.1,6 However, this could not be demonstrated with the Barthel index (OR=0.99 for each one-point increase in the scale; p=0.12), possibly due to greater specificity and predictive value of the Katz index described in previous publications.39 We interpret worsening of the performance status as a sign of the degree of aging of these patients, and this could explain the correlation to mortality in critical disease. Other factors associated to aging, such as cognitive status and quality of life showed no correlation to in-hospital mortality in our study. Severe cognitive deterioration has been described by other authors as a mortality risk factor in elderly patients admitted to the ICU.1 This was not seen in our study (p=0.51), possibly because of the low proportion of included individuals with dementia (S-IQCODE>4)40: only 9% (n=22), with a median score of 3.2 points (IQR: 3.2–3.4).
The relationship between malnutrition and mortality in critically ill elderly patients has been described in the literature, and in this respect the body mass index (BMI) is a known in-hospital mortality risk factor.1,2,41 Our SGA-based evaluation of malnutrition upon admission yielded conclusive data, with 50% of the patients presenting some degree of malnutrition. Moderate and severe malnutrition were identified as mortality predictors in our cohort (OR: 2.14 and 2.12, respectively).
Kidney failure with a need for renal replacement therapy is an established mortality risk factor in critical patients of all ages.42 The male gender likewise has been identified in recent studies as a predictor of mortality in elderly patients.7
With regard to the ventilation parameters recorded in the first days of admission to the ICU, some of them showed statistically significant results (such as SaO2/FiO2 and static compliance), but we do not consider these minimum differences in absolute values to be significant from the clinical perspective. On the other hand, although the recorded tidal volumes (9ml/kg theoretical body weight) were higher than those recommended in the current literature, they reflect the first 72h of MV, which include assist/control ventilation periods (volume control – continuous mandatory ventilation [VC-CMV] and pressure control – continuous mandatory ventilation [PC-CMV]) and periods of spontaneous ventilation (pressure control – continuous spontaneous ventilation [PC-CSV]). Considering the data in Table 2, it can be assumed that our study population was not in serious condition from the respiratory functional point of view. The data referred to performance status and cognitive function of the patients prior to the acute disease condition leading to admission were collected by questioning the relatives or accompanying persons. Although this could be regarded as a weakness in the case of ADL, due to the risk of bias, a recent study has found the correlation for ADL between such relatives or accompanying persons and the patients to be good.43 A weak point in our study is the possible under-representation of elderly subjects with more comorbidities that are never admitted to the ICU due to limitation of therapeutic effort – thus giving only a partial view of the effect of age upon the outcomes of admission to intensive care. Nevertheless, our study population had an important presence of comorbidities, with a median Charlson index score of 6 points. Another weak point is that the study was carried out in a single high complexity center treating patients with an important disease burden and with many elderly individuals – a situation that could affect its external validity.
In addition to its prospective design, sample size and lack of losses over follow-up, a strength of our study was the fact that it excluded patients subjected to some degree of limitation of therapeutic effort – this being corroborated by the prolonged stay both in the ICU and in hospital. This circumstance avoided confounders in detecting factors associated to mortality.
In conclusion, we understand that chronological age as such does not constitute a predictor of in-hospital mortality in critically ill elderly patients. In contrast, a history of malnutrition and impaired performance status referred to activities of daily living (both related to the aging process) are strong outcome predictors during hospital stay. Taking these aspects into account upon admission to the ICU could help in decision making and improve the prognosis of critically ill elderly subjects.
Financial supportThis study has received no financial support.
AuthorshipSergio Eduardo Giannasi: Study design, writing of the manuscript, critical review and final approval.
Maria Sofia Venuti: Data compilation, analysis and interpretation, writing of the manuscript and final approval.
Alejandro Midley: Study design, critical review and final approval.
Nicolas G. Roux: Study design, critical review and final approval.
Claudia Kecskes: Writing of the manuscript, critical review and final approval.
Eduardo San Roman: Critical review and final approval.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Thanks are due to the Department of Adult Intensive Care of the Italian Hospital of Buenos Aires, and to Sergio Terrasa.
Please cite this article as: Giannasi SE, Venuti MS, Midley AD, Roux N, Kecskes C, San Román E. Factores de riesgo de mortalidad de los pacientes ancianos en cuidados intensivos sin limitación del esfuerzo de tratamiento. Med Intensiva. 2018;42:482–489.